CHAPTER 58
Microvascular Principles
Christopher M. Harris,1 Allen O. Mitchell,2 and Robert M. Laughlin3
1Department of Oral and Maxillofacial Surgery, Naval Medical Center Portsmouth, Portsmouth, Virginia, USA
2Otolaryngology—Head and Neck Surgery, Naval Medical Center Portsmouth, Portsmouth, Virginia, USA
3Department of Oral and Maxillofacial Surgery, Naval Medical Center San Diego, San Diego, California, USA
Indications for Microvascular Reconstruction of Maxillofacial Defects
- Composite mandibular reconstruction
- Moderate to large soft tissue reconstruction in malignancy (i.e., hemiglossectomy and buccal carcinomas)
- Reconstruction of stage III osteoradionecrosis (bone and/or composite defects)
- Reconstruction of large soft tissue defects and soft tissue radionecrosis
- Large maxillectomy defects where obturation is unsuitable reconstruction
- Orbitomaxillary defects
- Benign, anterior mandibular defects with or without soft tissue deficiency
Contraindications for Microvascular Reconstruction of Maxillofacial Defects
- Medical status unsuitable for prolonged surgical procedure (advanced age alone is not a disqualifier)
- Medical history of hypercoaguable state (e.g., sickle cell anemia, Factor V Leiden, antiphospholipid syndrome, myeloproliferative disorders, or morbid obesity)
- Significant peripheral vascular disease
Microvascular Principles and Technique
A thorough preoperative evaluation is undertaken prior to performing any vascularized tissue flap. A complete medical history and physical exam are undertaken, including laboratory and imaging studies as indicated. Many patients presenting with oral cancers have significant medical comorbidities (e.g., chronic obstructive pulmonary disorder, polysubstance abuse, diabetes, and cardiovascular disease) that require evaluation and optimization prior to surgery.
Accurate assessment of the planned ablative defect and its consequences on flap selection, as well as the final prosthetic rehabilitation planning, must be performed prior to surgical therapy. Choosing the donor site is based on the types of tissues to be replaced, the prosthodontic requirements, and the quality of the donor site. The surgeon’s familiarity and success with the flap choice are also considered. For example, in our practice, most oral cancer defects are reconstructed with radial forearm, anterolateral thigh, and fibula free flaps. The reliability with these flaps is likely to be much higher, and the complication rate much lower, than a flap that is rarely utilized. When treatment planning any microvascular reconstruction, a “backup” reconstructive plan is also devised in the event that the primary flap choice is unusable or inadequate.
Specialized equipment is required to perform microvascular procedures. Equipment typically includes microvascular surgical instruments, microsurgical anastomotic couplers, surgical loupes, an operating microscope, microsurgical sutures and vascular clamps, Doppler ultrasound (external and internal), and warmed, heparinized irrigation fluids. Microsurgical instruments should be lightweight and made of titanium, measure 10–18 cm in length, and have low closing pressures. Magnetization of nontitanium instruments can increase the difficulty of positioning microsurgical needles. All instruments should be carefully inspected prior to the start of any case, and a reserve set should be available. It is a common occurrence to discover a bent instrument that needs to be replaced. A typical microsurgical instrument set consists of multiple jeweler’s forceps (curved and straight), microsurgical adventitial scissors (curved and straight), microsurgical suture scissors, vein dilator forceps, vascular clamp applying forceps, and microsurgical needle holders. The vascular clamp set contains multiple arterial and venous, single and double approximating clamps with or without frames of varying size. Clamps can be disposable or reusable, depending on the surgeon’s preference. Requirements for clamp pressures depend on vessel size and type; the pressure should be light enough to prevent leakage and damage to the vessel walls.
Venous anastomotic coupler devices are being used routinely in many institutions for venous anastomosis (Figures 58.1, 58.10, and 58.11). Studies have demonstrated an equivalent patency rate when compared to hand-sewn anastomoses, with significantly shorter anastomosis times and higher anastomosis strength. The SynovisTM anastomotic coupler (Birmingham, Alabama, USA) (Figure 58.1) is routinely used at our institution. The device can be used with vessel sizes from 1.0 mm to 4.0 mm. Arterial coupling is also possible with this device. The anastomotic coupler consists of two polyethylene rings in which several stainless-steel pins are embedded. The vessels are measured for the appropriate size, the vessels are passed through the rings, and the walls are everted over the steel pins. The coupler is closed, and the venous walls are sealed together by piercing the opposing ring.
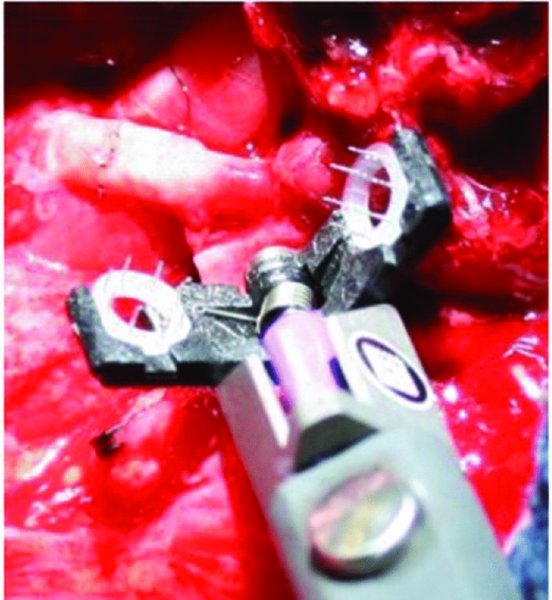
Figure 58.1. SynovisTM microvascular anastomotic coupling device.
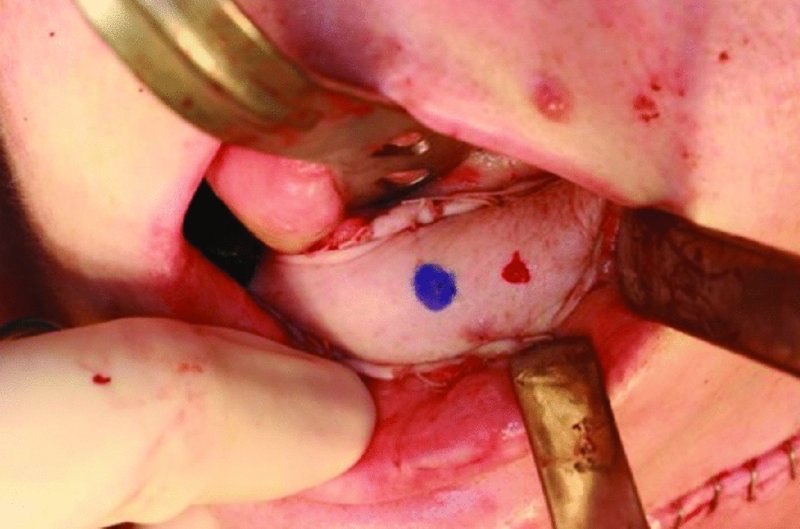
Figure 58.2. Radial forearm free flap covering a large soft tissue defect resulting from resection of a T4N2bM0—stage IVa gingival carcinoma. The flap demonstrates good vascularity, and the perforator is marked and bleeding from a 27-gauge needle stick.
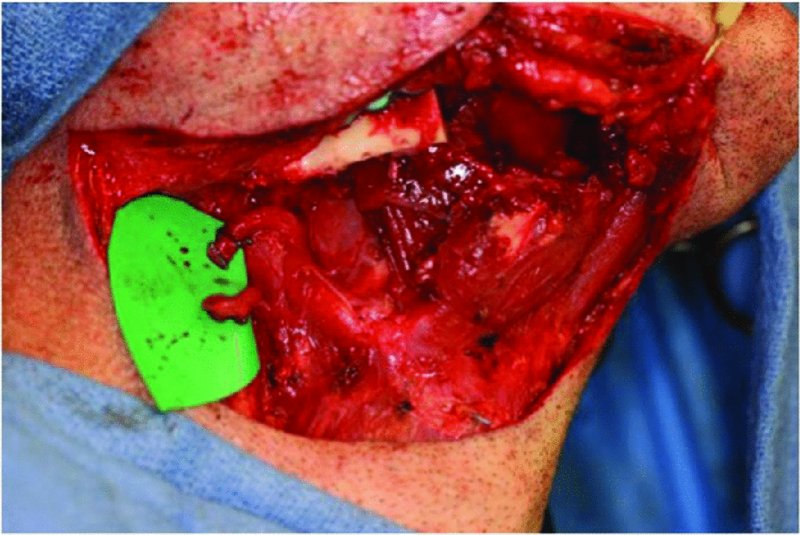
Figure 58.3. After resection of the amleloblastoma, the recipient facial vessels are identified, isolated and prepared.



