CHAPTER 5
Onlay Bone Grafting
Michael Grau, Jr.1 and Christopher J. Haggerty2
1Department of Oral and Maxillofacial Surgery, Naval Medical Center San Diego, California, USA
2Private Practice, Lakewood Oral and Maxillofacial Surgery Specialists, Lees Summit; and Department of Oral and Maxillofacial Surgery, University of Missouri–Kansas City, Kansas City, Missouri, USA
Procedure: Onlay Bone Grafting
A method of augmenting the hard tissue of the mandible and/or maxilla to facilitate the placement of endosseous implants.
Indications
- Inadequate available bone width and/or height for the placement of dental implants in an ideal restorative position
- Adequate space in relation to vital structures allowing for the placement of fixation screws
Contraindications
- Inadequate restorative space for the fabrication of the final prosthesis
- Intravenous bisphosphonate therapy
- Immunosuppressed patients
- Previous radiation therapy to the site of grafting and implant placement
- Relative contraindications include uncontrolled diabetes mellitus, cigarette smoking, alcoholism, active periodontal disease, and chronic corticosteroid therapy
Intraoral Alveolar Ridge Augmentation Technique
- Preoperative models and radiographs are used to estimate the amount of bone augmentation required.
- The risks, benefits, and alternatives of the procedure and the type of graft material utilized are discussed with the patient. The graft material (autogenous or allogenous) is selected based on defect size, defect location, patient desires, and the surgeon’s preference.
- The procedure may be performed under local anesthesia, intravenous sedation, or general endotracheal sedation depending on the amount of bone augmentation required, the donor site, the patient’s anxiety level, and the patient’s medical history.
- Antibiotic prophylaxis is administered 30 minutes preoperatively. The oral cavity is prepped with chlorohexidine and/or betadine solution.
- Local anesthesia is administered within the augmentation site, utilizing nerve blocks for the mandible and local infiltration for the maxilla. Local anesthesia is also injected within the attached and unattached tissue of the recipient site to aid in hemostasis and to hydro-dissect the periosteum from the underlying bone.
- A #15 blade is utilized to bisect the keratinized tissue overlying the atrophic alveolar ridge. Vertical releasing incisions are frequently necessary to obtain adequate exposure of the surgical site and to allow for a tension-free closure over the augmented area.
-
A full-thickness mucoperiosteal flap is elevated to expose the atrophic alveolar site (see Figures 5.1 and 5.8 in the Case Reports) and the osseus defect is measured. The defect size determines the amount of allograft required or the amount of autogenous bone to be extracted from the harvest site.
- For block grafting, the recipient site is contoured (Figure 5.2, Case Report 5.1) with a round bur to allow for passive and even contact of the graft to the underlying alveolus.
- A 701 bur may be utilized to create perforations within the buccal cortex to stimulate blood flow to the area. The periosteum is released with a #15 blade (Figure 5.8, Case Report 5.2) in order to allow for added mobility of the mucoperiosteal flap and a tension-free closure.
-
The graft (block or particulate) is placed with the recipient site. For block grafts, the graft should be flush with the underlying bone, should have a passive fit, should completely fill the area to be grafted, and should have no sharp protruding edges or corners. Block grafts are fixated to the recipient site with a minimum of two positional screws (see Figures 5.4 and 5.23 in the Case Reports). Particulate grafting may be utilized to fill any residual defects between the graft and the recipient bed (Figure 5.23, Case Report 5.3).
- The full-thickness mucoperiosteal flap is advanced and checked for passivity. Membranes are placed (Figure 5.11, Case Report 5.2) based on the surgeon’s preference, the ability to achieve a tension-free primary closure of the augmented surgical site, and the presence of tears within the overlying mucoperiosteal flap.
- The surgical site is closed in a tension-free manner (Figure 5.12, Case Report 5.2) with 3-0 or 4-0 sutures.
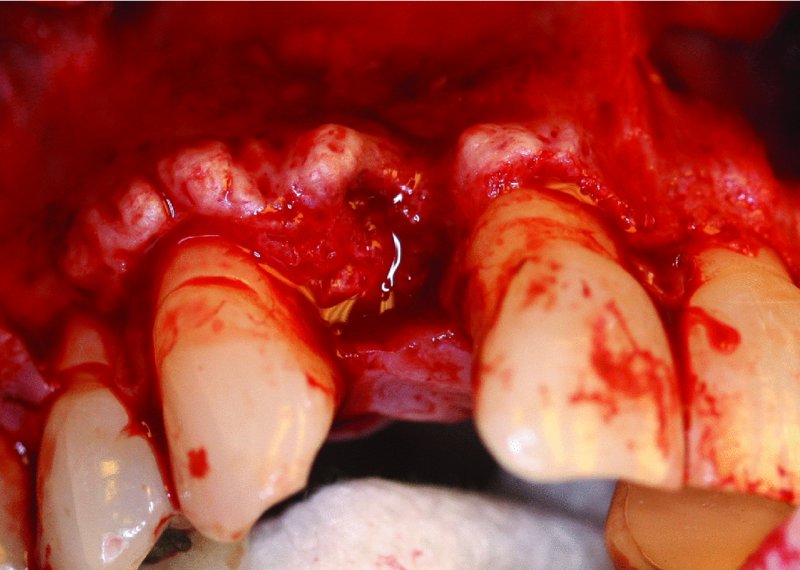
Figure 5.1. Mucoperiosteal flap reflection revealed alveolar ridge bony exostosis and a combined vertical and horizontal bone deficiency.
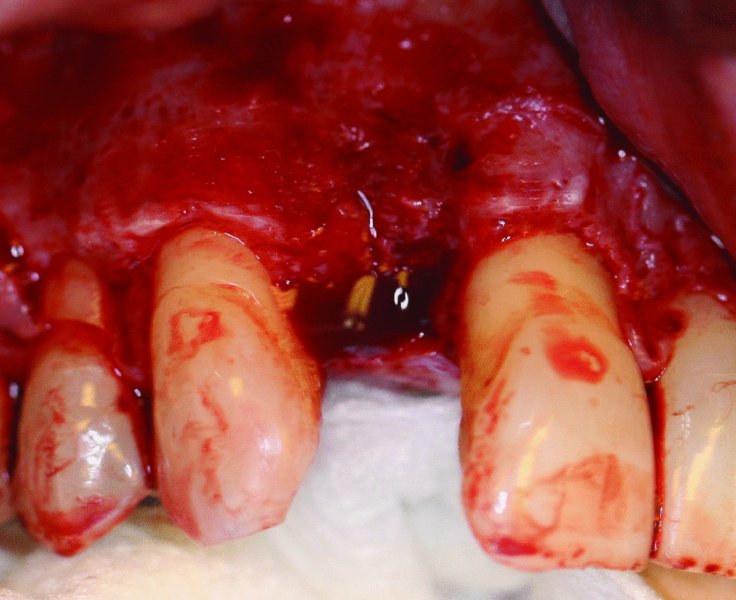
Figure 5.2. The recipient site is recontoured to remove areas of bony exostosis and to allow for even contact of the onlay graft.
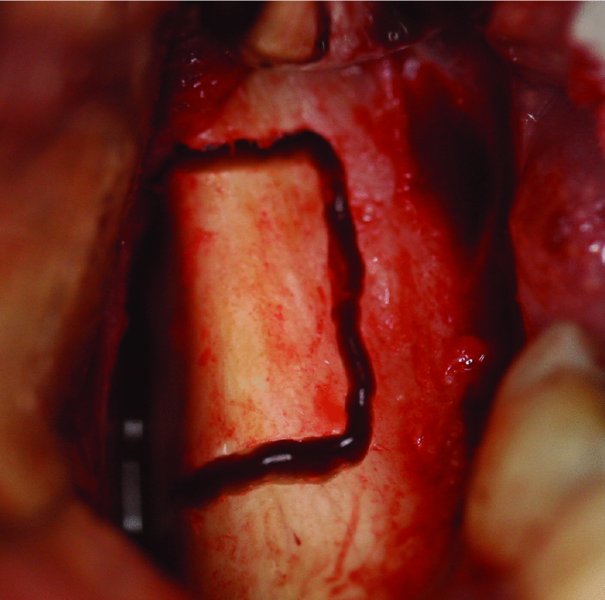
Figure 5.3. The lateral ramus is osteotomized posterior to the dentition.
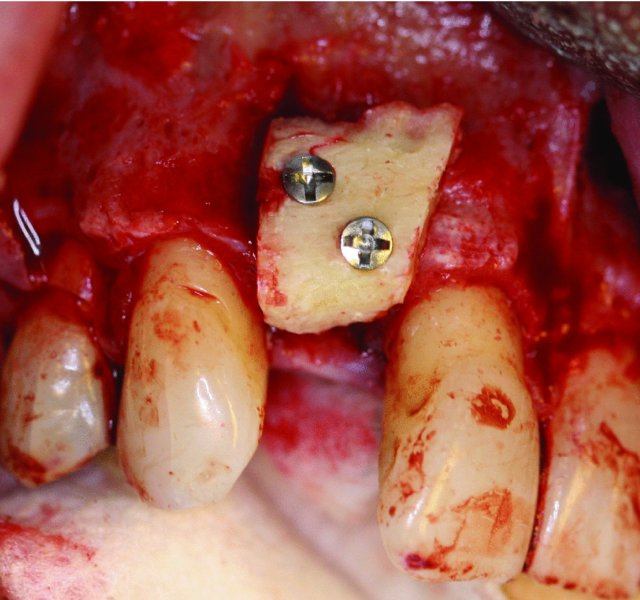
Figure 5.4. The ramus block onlay graft is secured to the donor site with positional screws. All sharp edges of the graft will be removed, and particulate bone will be packed around the periphery of the graft prior to closing the site.
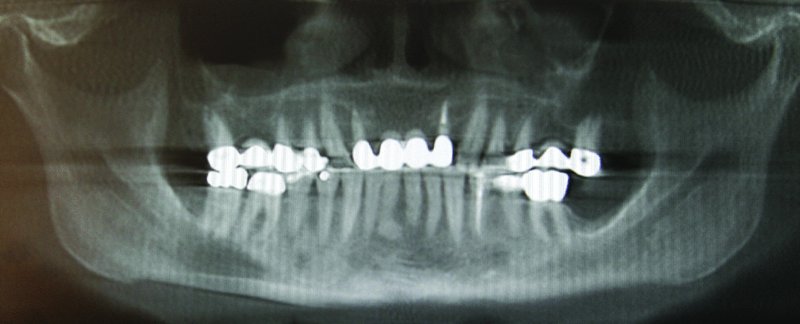
Figure 5.5. Orthopantomogram depicting failing teeth #7 and #10 with associated four-unit bridge.
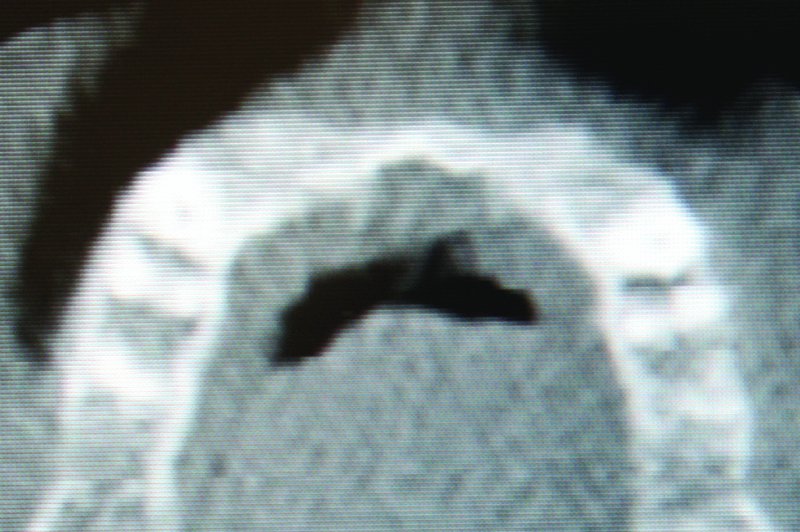
Figure 5.6. Axial cone beam computed tomography image depicting anterior maxillary alveolar resorption.
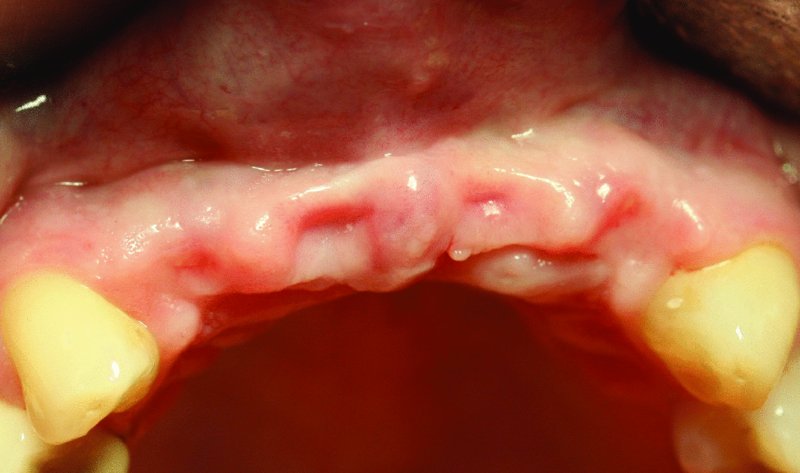
Figure 5.7. Patient with sufficient vertical bone height, but insufficient bone width for the placement of dental implants.
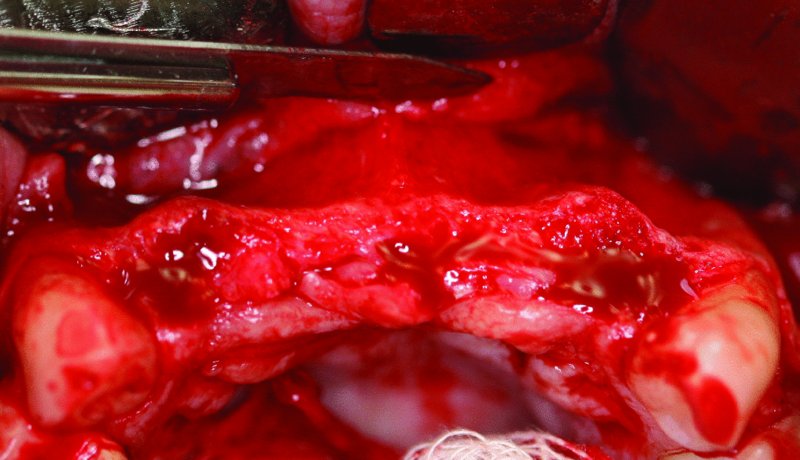
Figure 5.8. A mucoperiosteal tissue flap is elevated with vertical releases posterior to the canines. The periosteum is released with a #15 blade to allow for a tension-free closure.
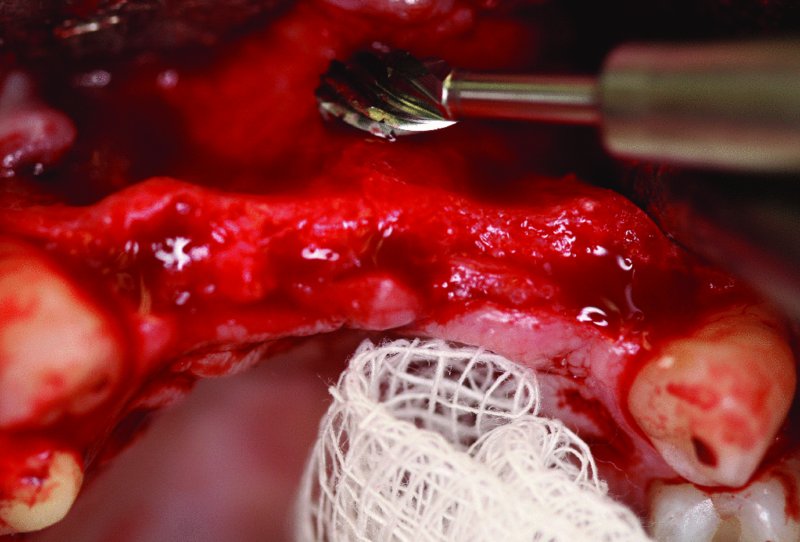
Figure 5.9. A round bur is used to stimulate bleeding of the rec/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


