5
The periodontium, tooth deposits and periodontal diseases
- Periodontal health as the key to maintaining lifelong tooth support
- Dental plaque as the principal cause of inflammatory diseases leading to destruction of tooth-supporting bone
- Cigarette smoking, diabetes and increasing age as the main risk factors in destructive periodontitis
- Effective treatment protocols
- Antibiotics in periodontal therapy
- The control of periodontal diseases in the long term
Introduction
Periodontics is the fundamental dental science. All restorative dentistry relies on healthy periodontal support for its longevity so it is therefore essential that all practitioners of dentistry understand the structure of the periodontal tissues, know how problems develop and are adept at managing periodontal problems.
The normal periodontium
The periodontium (periodontal tissues) hold the teeth in the mouth. The normal periodontium consists of:
- The gingiva.
- Bone.
- Cementum.
- The periodontal ligament.
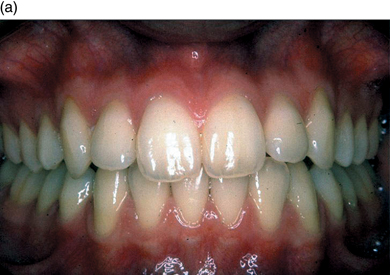
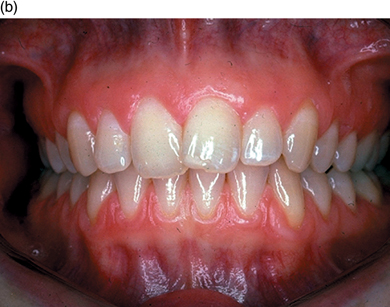
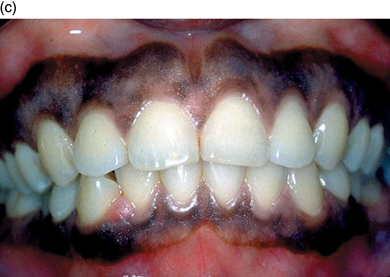
The appearance of healthy tissue varies between patients, and ‘normal’ healthy tissue can look quite different in different individuals (Figure 5.1a, b, c).
Figure 5.1 (a,b,c) Three very different, but all normal healthy gingival types.
The gingiva
There are two components to the gingival tissues:
- The attached gingiva.
- The alveolar mucosa.
Attached gingiva
The normal attached gingiva frames the crowns of the teeth on the buccal and lingual aspects and forms a strong protective cuff at the necks of the teeth, keeping food away from the delicate epithelial attachment. Covering the bony attachment to the teeth, it varies in colour, texture and architecture. Most Caucasians have a coral pink gingiva with a light stippling reminiscent of orange peel. This is because the tissue is a true mucoperiosteum and the stippling corresponds to a fibrous attachment of the epithelium to the periosteum. Therefore, the attached gingiva does not move (Figure 5.2).
Figure 5.2 Gingival anatomy. (Reproduced with permission from Clinical Periodontology and Implant Dentistry, 4th edition (eds J. Lindhe, T. Karring, and N. Lang). Blackwell Munksgaard; p. 6, Figure 1.6.)
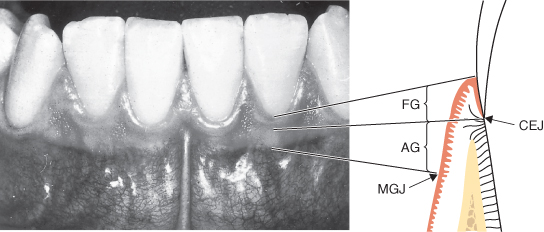
Gingival tissue is normally 2–3 mm thick and 1–5 mm wide, increasing in width with age. It has a dull surface and a firm consistency. At its coronal aspect the gingiva terminates in the free gingival margin, which is scalloped in outline to follow the contour of the teeth. At the apical aspect of the attached gingiva is the mucogingival junction, which separates it from the looser, more flexible, alveolar mucosa.
The biologic width
The ‘free’ gingival margin is free because it is not attached to the tooth on its inner (crevicular) aspect. There is a gingival crevice, on the inner aspect of the free gingiva, anything from 1–3 mm deep, usually deeper interproximally than labially or lingually. At the base of the crevice the gingiva is attached to the tooth by means of a 1 mm wide layer of epithelial cells attached to the cementum surface in the same way as they are attached to each other. This band is known as the junctional epithelium. One millimetre beyond the gingival crevice, the junctional epithelium gives way to a band of connective tissue, approximately 1mm wide, containing collagen fibres arranged in groups (Figure 5.3).
Figure 5.3 The oral epithelium. CEJ, cemento–enamel junction; CT, connective tissue; E, enamel; OE, oral epithelium; OSE, oral sulcular (crevicular) epithelium; JE, junctional epithelium. (Reproduced with permission from Clinical Periodontology and Implant Dentistry, 4th edition (eds J. Lindhe, T. Karring, and N. Lang). Blackwell Munksgaard; p. 8, Figure 1.14a & b.)
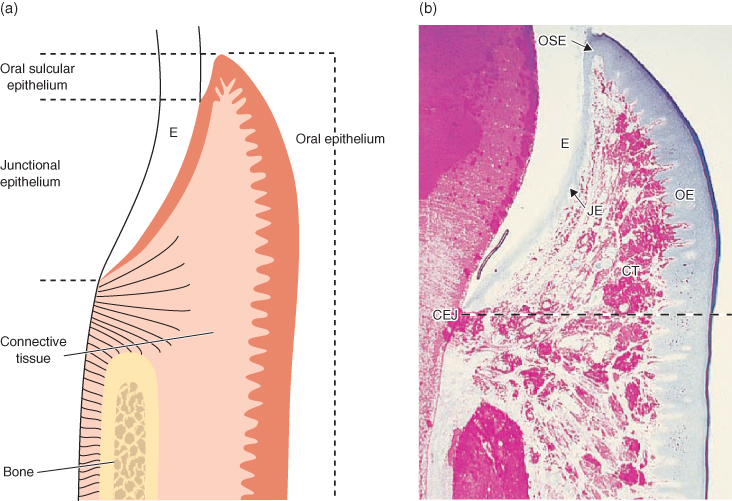
These three elements, the gingival crevice, junctional epithelium and connective tissue attachment, make up the biologic width of the gingiva. It will always re-establish itself after any damage to the gingiva and must not be violated beyond the gingival crevice by restorative dentistry. A restoration margin located beyond the crevice, in the area attached by junctional epithelium or connective tissue, will cause persistent inflammation and this may be both uncomfortable, due to pain and bleeding, and unsightly due to swollen and discoloured tissue. The restorative dentist or therapist, therefore, must respect the biologic width. Errors in tooth preparation can sometimes only be corrected by crown-lengthening surgery.
The gingival papilla
Between the teeth, the attached gingiva forms a triangular papilla on both buccal and lingual aspects. If it could be viewed transversely (i.e. from in between the teeth), the papillae would resemble two peaks and a valley and the contact areas between the teeth determine the shape of the valleys. Between the anterior teeth the papillae have the shape of a pyramid, and between the posterior teeth the papillae are more flattened. The valley between the two peaks of the papillae is known as the interdental col.
Microscopically, the attached gingiva has a layer of keratin (a tough fibrous insoluble protein) on the surface, covering a layer of connective tissue known as the lamina propria. The basal layer of the epithelium is in contact with the periosteum, which is in direct contact with the underlying bone. The periosteum is a richly vascular layer of dense connective tissue, containing osteoblasts in the deeper part and with fibres embedded in the bone surface making it very difficult to separate the periosteum from the bone. The gingiva derives much of its blood supply from the periosteum.
Gingival periodontal fibres
Although the gingival tissue contains randomly arranged collagen fibres everywhere, there are four groups of recognisable fibres closely associated with the attachment of the gingiva to the tooth: Dentogingival fibres, Dentoperiosteal fibres, circular fibres, and trans-septal fibres.
- Dentogingival fibres are embedded in the cementum in the connective tissue attachment area, and fan out in a coronal direction towards the free gingival margin.
- Dentoperiosteal fibres are embedded in the same area as dentogingival fibres, but turn in an apical direction towards the root of the tooth.
- Circular fibres encircle the tooth like a ring.
- Transeptal fibres are embedded in the cementum of adjacent teeth and run above the bone crest.
These gingival collagen fibres are responsible for the stippling seen in healthy gingival tissue, and for maintaining a tightly attached gingival cuff around the tooth. They are damaged or destroyed in gingivitis and periodontitis, and severed by the raising of a surgical flap. This explains why:
- The gingivae become glazed when the tissues are inflamed, changing the surface to a smooth texture.
- Teeth become looser due to gingivitis and periodontitis, and become tighter after successful treatment as the fibres regrow.
- Teeth become looser when a surgical flap is raised, severing the transeptal and circular fibres, and tighter after surgery when the tissues heal; healing takes approximately 3–4 months.
Alveolar mucosa
Alveolar mucosa is a softer and more flexible tissue than attached gingiva. It has a much thinner keratin layer on the surface and is continuous with the mucosa of the buccal and lingual sulci. It is therefore a darker red than the attached gingivae and has a smooth shiny surface.
Bone
The bone that supports the teeth, the alveolar bone, grows with the teeth as they erupt into the mouth forming the alveolus of both maxilla and mandible. The alveolar bone supports the teeth by absorbing and distributing occlusal forces. The wall of each tooth socket is formed from a thin layer of dense compact bone. The areas between the tooth sockets are composed of spongy cancellous bone, which contains many more spaces than compact (cortical) bone. The ends of the periodontal ligament fibres, which are also known as Sharpey’s fibres, are embedded into the cementum and the alveolar bone of the socket walls.
Bone is a constantly changing dynamic tissue, always remodelling in response to functional change and needs.
Cementum
The cementum is the calcified surface tissue covering the dentine of the root of the tooth. It has a similar structure to bone, but it has no blood supply, no nerves, does not remodel and is more resistant to resorption than bone. This resistance to resorption is important in orthodontic treatment, allowing the tooth to move by alveolar bone resorption in response to controlled applied pressure, without root resorption. The cementum attaches the periodontal ligament (Sharpey’s) fibres to the root of the tooth and contributes to the process of repair after the root surface has been damaged. The cementum protects the underlying dentine and compensates for occlusal attrition by increasing in thickness in the apical area (cementosis). Cementum therefore does not remodel in response to functional changes, but instead gradually increases in thickness with age by the deposition of new layers; where this increase is excessive it is known as hypercementosis.
Connective tissue
Connective tissue consists of a ground substance containing fibroblasts, macrophages and neutrophils (defence cells) and lymphocytes (active in the immune response process) and fibrous tissue. The fibrous tissue is made up of elastic elastin fibres and non-elastic collagen fibres.
Periodontal ligament
The periodontal ligament (PDL) surrounds the tooth. It consists of a dense network of collagen fibres (Sharpey’s fibres) arranged in oblique rows, inserted at one end into the bone and at the other into the cementum. This is the means by which the tooth is attached to the bone. The fibres are elastic in nature, giving them the ability to extend in response to occlusal loading (biting pressure), and this extension acts as a cushion for the tooth, allowing it to absorb functional forces thus reducing the likelihood of damage to the supporting structures and making chewing more comfortable.
Connective tissue fills the spaces between the fibres, through which run the blood vessels that supply the tooth attachment apparatus with blood, and the small nerve fibres that make the PDL so sensitive. Whereas pain from a dental pulp is poorly localised – the patient usually cannot tell which tooth is hurting – pain from the PDL is very accurately localised. The patient can describe exactly which tooth is the source of pain.
Blood supply
Blood is brought to the gingival tissues by small branches of the superior and inferior dental arteries. The gingiva receives its blood mainly from blood vessels on the surface of the periosteum arising from various facial arteries. A fine network of small vessels traverses the tissue and can often be seen with the naked eye.
Nerve supply
The gingivae are supplied with nerves from the various branches of the trigeminal nerve, the same nerves that supply the teeth themselves with fibres carrying sensory input (sensation) back to the brain.
The classification of periodontal disease
A classification of diseases is useful so that we can make sense of the clinical information we collect and to help us to take a rational approach to planning treatment for our patients. The classification presented here is based on the American Academy of Periodontology (AAP) Classification of 1999. A simplified version of this is described in Table 5.1. It has been simplified for the purposes of this book, however the full classification can be obtained from the AAP website (www.perio.org/).
Table 5.1 The classification of periodontal disease.

Chronic gingivitis
The term gingivitis means inflammation of the gingiva. The most common cause is the accumulation of dental bacterial plaque at the gingival margins if not effectively removed by the patient. Unlike periodontitis, gingivitis results in reversible damage to the gingival tissues. The effect of the plaque on the tissue varies between different individuals and may be modified by various factors, as listed in the AAP classification (Table 5.1).
Clinical features
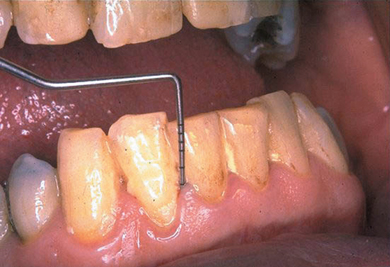
The signs of gingivitis are the same signs as can be found in any other inflammatory condition, and, in particular, erythema (redness), oedema (swelling), and pain or tenderness can be identified by close examination of the gingival tissues. The symptoms are that the patient may notice bleeding, and perhaps some tenderness of the gingivae when brushing, although pain is not normally a clinical feature of gingivitis. Inspection of the gingivae with a good white light may show a red line at the margin with swelling and redness of the papillae initially and later the margins, with a tendency to bleed on gentle probing with a blunt periodontal probe. The probing depth does not exceed 3 mm in gingivitis unless the tissue is so swollen that the gingival tissue is larger than normal. The level of the attachment of the gingivae to the tooth remains unchanged. Plaque can frequently be seen at the gingival margins corresponding to the signs of inflammation. It takes 2–3 days for unnoticed plaque to result in the development of gingivitis. Some patients brush vigorously just before attending for treatment and may have removed the plaque, but signs of the tissue having bled on brushing will then be seen. The examiner should not be deceived when looking closely! (Figure 5.4).
Figure 5.4 Chronic marginal gingivitis in a 72-year-old man; probing depth does not exceed 3 mm.
Chronic periodontitis
Susceptibility
For most patients with gingivitis, the inflammation does not progress beyond the gingival margins; however 10–15% of patients are susceptible to a more destructive process known as chronic periodontitis. In the most recent UK Adult Dental Health Survey (1998) it was found that 54% of dentate adults had some periodontal pocketing of 4 mm or more and 5% had deep pocketing (6 mm or more); 43% had some loss of attachment of 4 mm or more and 8% had loss of attachment of 6 mm or more. The prevalence of pocketing and loss of attachment increased with age. For example, the proportion of dentate adults with some loss of attachment increased from 14% among those aged 16–24 years to 85% of those aged 65 and over.
The initiation of periodontitis in an individual is thought to be due to a gene polymorphism, which causes a change in the behaviour of cytokines, substances that regulate the movement of immune system defending cells. This behaviour change leads to the destruction of bone and connective tissue, which usually takes place very slowly, and is mostly asymptomatic, so that affected teeth may lose their attachment to the bone over a period of 30–50 years. The genetic element explains why chronic periodontitis frequently affects several members of the same family.
The technology exists to identify the genetic abnormality (which in the future may well turn out to be one of many) that is thought to determine the susceptibility to destructive disease and this knowledge would be very helpful in deciding about the long-term prognosis of the affected teeth and therefore in treatment planning. There is no chair-side test currently available in the UK to assess genetic susceptibility or the presence of periodontal pathogens.
It follows that susceptibility cannot be ‘treated’. Once susceptible, the patient will always be so because this characteristic is genetically determined. Treatment of periodontitis is directed towards elimination of the substance to which the patient is susceptible (i.e. the plaque). Susceptible patients, once treated, must therefore be monitored for the rest of their lives to ensure that periodontitis does not become re-established.
General risk factors
Cigarette smoking
The most important known risk factor for periodontitis is cigarette smoking. Smokers have more periodontal problems than non-smokers, and the results of treatment are poorer in smokers. This is thought to be due to:
- A reduction in gingival blood circulation (which results in a reduction in bleeding on both brushing and probing in smokers).
- Impaired white blood cell mobility and function.
- Impaired wound healing.
- An increased production of inflammatory substances (cytokines), particularly due to nicotine, which results in an increased production of collagenase.
Many studies (Palmer et al., 2005; Preshaw et al., 2005) have shown that treatment outcomes are not as good for smokers as for non-smokers, and recurrence of disease is more likely.
Diabetes
Diabetes increases the risk of periodontal diseases and because the way in which diabetic complications develop is a similar pathological process to periodontitis, it is not surprising that there may be a close relationship between the two diseases. It has even been reported that treatment of periodontitis may improve diabetic control (Grossi et al., 1997). Wound healing is adversely affected by diabetes, especially if poorly controlled, and this can make treatment of diabetic patients more difficult.
Other systemic conditions
Hormonal changes can affect the response of the gingival tissues to plaque and to therapy. Puberty, pregnancy, and the menopause are all known to cause changes in gingival response. The increase in the severity of gingivitis during pregnancy has been attributed to the increased circulating levels of progesterone causing increased permeability of gingival vessels and a consequent increase in bleeding and swelling.
An enlargement of a gingival papilla, known as a pregnancy epulis, is frequently seen during pregnancy. These lesions can be up to 2 cm in diameter; removal is best deferred until after parturition if possible although they can be quite large and troublesome. Thorough plaque control is essential and the lesions tend to resolve spontaneously at the end of the pregnancy.
Leukaemias, resulting in altered leukocyte function, are likely to increase the incidence of periodontal disease.
Age
Older patients are more likely to have periodontitis than younger patients due to many years of exposure to plaque and to the various risk factors known to be associated with periodontitis.
Stress
Many clinicians believe that stress is a factor modifying the host response to plaque. The evidence is inconclusive, but it has been suggested that stress can have a direct effect on immune responses to plaque. Equally, stress may exert an influence by way of behavioural change, reducing the thoroughness and therefore the effectiveness of oral hygiene.
Periodontal diseases and general health
There is a growing body of evidence suggesting that there is an association between periodontitis and various systemic conditions, in particular the incidence of premature low birth weight babies and coronary heart disease. Various studies have been published suggesting that there appears to be a benefit in improving the periodontal health of expectant mothers expressed as a reduced incidence of adverse pregnancy outcomes.
The association between periodontitis and cardiovascular disease is thought to be mediated by inflammatory markers, in particular C-reactive protein. Periodontitis is also thought to be associated with raised levels of serum cholesterol and further research is in progress to elucidate this.
Local risk factors
Anything that makes it easier for plaque to accumulate at the gingival margins is a local risk factor for periodontitis. This includes:
- Calculus.
- Enamel pearls (a small, focal excessive mass of enamel on the surface of the tooth. They occur most frequently in the bifurcation or trifurcation of the tooth and are occasionally supported by dentine and very rarely contain pulp tissue).
- Root grooves and concavities.
- Malpositioned teeth (Figure 5.5).
- Overhanging, poorly fitting or contoured restorations.
- Removable partial dentures.
- Weak or malpositioned contact areas between the teeth.
- A deep overbite causing direct gingival trauma.
Figure 5.5 Palatally placed upper canine.
Clinical features
Gingivitis becomes periodontitis when the junctional epithelium is damaged and migrates apically. The clinical result of this is an increase in the depth of the gingival crevice beyond 3 mm. The gingival crevice now has become a periodontal pocket, and the implication of this is that the patient can no longer remove the plaque that is located at the base of the pocket, thereby allowing the condition to progress towards the apex of the tooth. Periodontal probing will now show depths deeper than 3 mm and it is well established that the inflammation quickly progresses to involve the connective tissue that is situated between the junctional epithelium and the periodontal ligament (PDL).
The same clinical features seen in gingivitis are also commonly seen in periodontitis i.e. oedematous (swollen), erythematous (red) and discoloured gingival margins and papillae, with bleeding on gentle probing with a blunt periodontal probe. The most important diagnostic information is obtained by the use of the periodontal probe. Inserted into the gingival crevice, aligned with the long axis of the tooth, the probe is moved apically until resistance is felt to gentle probing. In some cases this will be uncomfortable for the patient, and will always cause bleeding if the subgingival tissues are inflamed. There will be no bleeding to gentle probing if the tissues are not inflamed or if the blood supply to the gingivae has been reduced by cigarette smoking, disguising the true condition of the tissues.
Periodontal measurements are recorded on a chart and can then be used to assess the severity of the condition, and to compare measurements taken at previous visits. The progress of the condition can thereby be quantified.
Other clinical features that may be observed are:
- Detachment of papillae.
- Swelling of papillae.
- Gingival sinuses.
- Hyperplasia of papillae or gingival margins.
- Exudation of pus on digital pressure or probing.
- Mobility.
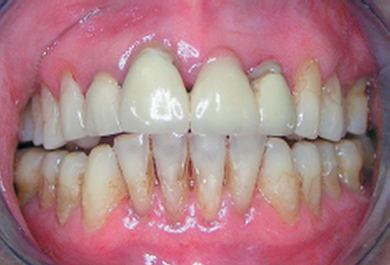
- Gingival recession: this may be associated with destructive periodontitis because the gingiva derives much of its blood supply from the underlying periosteum. If bone is destroyed by periodontitis the gingiva will tend to follow the bone crest in an apical direction; the thinner the gingival tissue, the more likely this will be to happen (Figure 5.6).
- Clinical evidence of furcation involvement by means of probing into the inter-radicular space.
Figure 5.6 Atrophic periodontitis where gingival tissue is thin; pocketing is predominantly shallow.
Measurement of tooth mobility
Tooth mobility is assessed using two handles of two hand instruments positioned buccally and lingually. The tooth is moved buccolingually with the instruments and the degree of movement recorded (Table 5.2).
Table 5.2 Measuring tooth mobility.
| Grade | Mobility |
| 1 | Horizontal movement of the crown 0.2–1 mm |
| 2 | Horizontal movement of the crown >1 mm |
| 3 | Horizontal movement of the crown >1 mm and additional movability in a vertical direction |
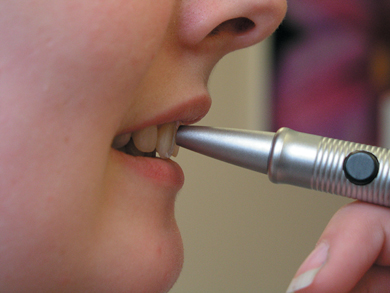
This crude system is very subjective but remains the only method in universal use. Recent research has resulted in the development of an electronic device for measuring tooth mobility, the Periotest, which uses an accelerometer to measure the resistance of the tooth to a force applied 16 times over a four second period by a small metal cylinder. This method employs a 60-point numerical scale ranging from −10 (ankylosed teeth or osseointegration e.g. implants) to +50 (extreme mobility) and has the advantage of providing results that are reproducible and comparable with previous readings. Increasing or decreasing trends in mobility of individual teeth can thereby be more accurately assessed (Figure 5.7).
Figure 5.7 The Periotest, for assessment of tooth mobility using an accelerometer.
Aggressive periodontitis
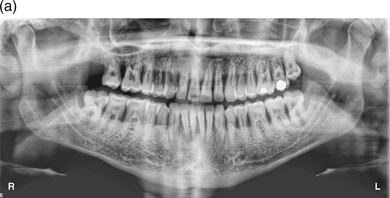
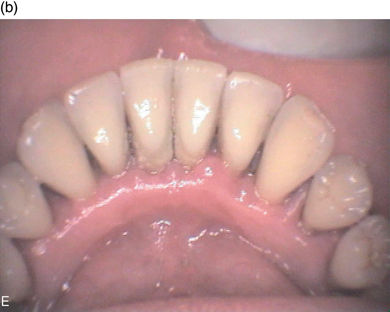
Among the relatively small proportion of the population who are susceptible to periodontitis, a small subgroup suffers more rapid bone destruction, so that by the age of approximately 35, they may have lost half of their total tooth attachment; this can be clearly seen on radiographs. This condition is known as aggressive periodontitis and it may affect all the teeth equally, only incisors and molars, or even just a small number of isolated teeth. It is essential to identify this group of patients as early as possible in the progress of the condition, because early treatment can be effective and long lasting, whereas delayed diagnosis can be disastrous for the survival of the teeth (Figure 5.8a and b).
Figure 5.8 (a and b) Aggressive periodontitis in a man aged 22.
This highly destructive form of periodontitis is characterised by rapid attachment loss with destruction of the periodontal ligament and supporting bone in an otherwise healthy young adult usually below the age of 35, but older patients may be affected. There is often a familial tendency (other members of the family have been affected) and this condition is thought to be associated with high levels of specific bacteria, notably Aggregatibacter actinomycetemcomitans and Porphyromonas gingivalis. There is some evidence of altered leucocyte function and also increased production of some immune system mediators.
Clinical features
Aggressive periodontitis, formerly known as early onset periodontitis, frequently presents with a lack of overt gingival inflammation, relatively good oral hygiene and low levels of visible plaque, which is inconsistent with the aggressive nature of the disease, and the diagnosis can therefore easily be missed. Clear signs of supporting bone loss are to be found on radiographs, however, and these should therefore be scrutinised in all cases. Vigilance is required on the part of the dentist, therapist and hygienist to ensure that aggressive cases are diagnosed as early as possible.
Aggressive periodontitis is rarely seen affecting children below the age of puberty. When it does occur at this age it is known as prepubertal periodontitis.
Localised and generalised forms of aggressive periodontitis are recognised corresponding to the older terms rapidly progressive periodontitis and localised juvenile periodontitis. Localised aggressive periodontitis (LAP) involves the incisors and molars, with onset around or soon after puberty, and is characterised by interproximal attachment loss on at least two permanent teeth, one of which is a first molar, and involving no more that two teeth other than first molars and incisors.
Generalised aggressive periodontitis (GAP) usually has a later onset, before the age of 30, and is characterised by generalised attachment loss involving at least three permanent teeth other than first molars and incisors. There is a more episodic nature to the progress of GAP.
Most studies put the prevalence of aggressive periodontitis among 13–20-year-olds at well below 1%; however, this still represents a large number of people who may be affected and the disease is so destructive that early detection, by means of periodontal screening for all teenagers, is essential in the protection of those individuals who may develop this condition.
Vigorous treatment, including mechanical plaque removal and systemic antibiotics, can be just as successful in controlling these conditions, as it is in controlling the more common forms of periodontitis
Histopathology of periodontal diseases
Five histopathological states are recognised in the gingiva (Lindhe et al., 2003) namely:
- Pristine gingiva (only really found in experimental animals) has an intact layer of epithelium lining the gingival crevice, and no inflammatory cells in the connective tissue. There is a continuous sparse migration of neutrophil leucocytes into the coronal part of the junctional epithelium and gingival crevice.
- Normal healthy gingiva has a small number of inflammatory cells in the junctional epithelium and connective tissue. Even though gingivitis at this stage is not clinically detectable, inflammatory changes can be found microscopically.
- Early gingivitis is seen after 10–20 days of plaque accumulation. There are an increased number of inflammatory cells in the tissue and an increase in the number of neutrophils emigrating into the gingival crevice. The junctional epithelium becomes thicker. The gingival connective tissue becomes more heavily infiltrated with inflammatory cells and dilated blood vessels.
- Established gingivitis has a more dense infiltration of inflammatory cells in the connective tissue, with plasma cells becoming much more evident (10–30%). Collagen loss increases due to an outpouring of the enzyme collagenase and the epithelium lining the gingival crevice continues to increase in thickness.
- Periodontitis is characterised by the apical migration of the junctional epithelium – the first stage of attachment loss. The same dense infiltration of inflammatory cells can be seen with plasma cells now dominant, (>50%). Bone loss now begins to occur.
Gingival recession
Gingival recession, which is a lack of complete coverage of the tooth root exposing the root surface, is very common, and is a cause of concern to many patients. The main causes are:
- Disproportion in sizes of teeth and alveolar bone – the tooth is too wide for the supporting alveolar bone; this situation may be exacerbated by abrasion from a toothbrush, especially if the patient is using an over-aggressive technique.
- Attachment loss due to chronic periodontitis.
Recession is often associated with a lack of keratinised mucosa and a highly placed muscle attachment is often found aggravating the situation. This can cause discomfort, and may lead to difficulty in maintaining plaque control, which results in inflammation, infection and a deterioration of the situation in relation to the tooth involved. Mostly, however, gingival health can be maintained in spite of recession, and with little or no attached gingiva. Where there is an adequate zone of keratinised mucosa associated with the defect, the problem is usually self-limiting and stops when the gingiva reaches a level at which it is in harmony with the underlying bone.
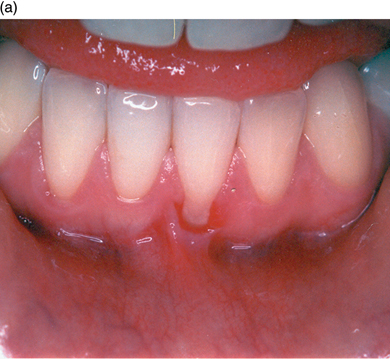
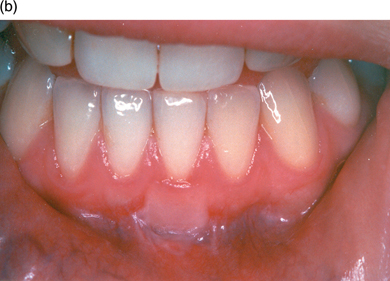
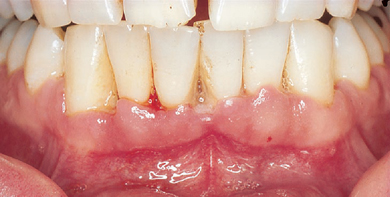
Recession may also cause aesthetic problems if it affects the anterior teeth, and may also result in sensitivity of the root surface to hot, cold or acidic foods and fluids. Where the patient experiences such problems, there are several predictable surgical techniques available to alleviate symptoms and sometimes achieve coverage of denuded root surfaces (Figure 5.9a and b).
Figure 5.9 (a and b) Gingival recession corrected with a free gingival graft.
Acute periodontal conditions
Necrotising periodontal diseases
The incidence of these conditions has declined in the UK over the last 30 years except for human immunodeficiency virus (HIV)-positive patients. There are three types of necrotising periodontal diseases (NPD) defined according to the extent of tissue destruction:
- Necrotising gingivitis, which is confined to the gingivae.
- Necrotising periodontitis, where there is attachment loss.
- Necrotising stomatitis, where the surrounding tissues are involved.
The predisposing factors are:
- Poor oral hygiene; associated with all the necrotising diseases.
- Cigarette smoking; commonly seen in all NPDs.
- Raised stress levels, e.g. students at exam time or soldiers in action.
- Malnutrition; associated with poor protein intake in developing countries.
- Fatigue.
- Immune dysfunction or suppression of the immune system.
- Pre-existing gingivitis.
- Systemic conditions such as leukaemia or HIV.
Clinical features
There is a visible change in gingival contour accompanied by bleeding, pain, and the loss of the apex of the papilla producing a punched out appearance. A grey slough, known as a pseudomembrane is frequently seen on the surface and there is tissue necrosis that causes an unpleasant but typically unforgettable odour (Figure 5.10). Patients may present with swollen lymph nodes (lymphadenopathy) or more rarely pyrexia. Destruction of tissue may progress to necrosis of the periodontal ligament and bone, which may be rapid and extend to the interproximal or facial bone, forming a fragment of dead bone known as a sequestrum.
Figure 5.10 NUG. Note the loss of papillae and the grey pseudomembrane. (Reproduced with permission from Clinical Periodontology and Implant Dentistry, 4th edition (eds J. Lindhe, T. Karring, and N. Lang). Blackwell Munksgaard; p. 245, Figure 10.3.)
Histopathology
Ulceration of the epithelium can be seen under the microscope, with necrosis (tissue death) and many inflammatory cells. Infecting organisms (spirochetes and fusiform bacilli) can be found in the tissues (not just on the surface) and in the necrotic slough.
Treatment of necrotising diseases
The most important therapeutic measure is debridement, (i.e. the removal of the soft and hard plaque, slough and calculus deposits), as soon as possible. This should be accomplished, or at least started, at the first visit as soon as the diagnosis has been made. The use of ultrasonic scalers makes this process much quicker, gentler and therefore more tolerable for the patient.
Hydrogen peroxide as a mouth rinse or applied directly to the ulcerated tissue is beneficial due to its mechanical cleansing properties and its ability to release oxygen into the area, damaging the environment for the predominantly anaerobic infecting organisms.
Chlorhexidine mouth rinse (0.2%) is an effective way of reducing plaque growth; it can be very helpful because the patient may not be able to brush thoroughly due to the pain of the infection.
Systemic antibiotics may be required if the patient shows signs of being generally ill, with fever, lassitude or severe lymph gland involvement. Metronidazole (200 mg, three times daily for 3–5 days) has been found to be very useful in NPD as well as penicillin and tetracyclines. Topical antibiotics have no place in the treatment of NPD because of the presence of bacteria within the tissues where there is no penetration of the antibiotic.
Patients with NPD should be seen frequently, perhaps daily, until the acute phase is over. Thereafter, completion of debridement and maintenance treatment as required should be arranged. In view of the association between NPD and other systemic conditions, consideration should be given to advising the patient’s medical practitioner so that further investigations can be considered.
Acute herpetic gingivostomatitis
Patients with this condition can present with pyrexia, lymph gland involvement, flu-like symptoms, stomatitis, oral ulceration, gingivitis and pain. The causative organism is the herpes simplex virus. The condition, which has an incubation period of about 7 days, is highly contagious and is spread from the oral lesions. Treatment is by maintaining adequate fluid intake and by the use of antipyretics and topical antiseptics. Antiviral drugs such as Aciclovir are not n/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


