5
External Root Resorption and Orthodontic Treatment—Assessment of the Evidence
Despite the extensive amount of research that has been conducted, it is unknown how orthodontic treatment influences root resorption. The etiological factors are complex and multifactorial, but it appears that apical root resorption results from a combination of individual biological variability and the effect of mechanical stimulus.
The aim of this chapter is to review the existing literature on orthodontically induced inflammatory root resorption (OIIRR). It will begin by providing background information on the etiology of external apical root resorption, followed by an examination of the prevalence of OIIRR and a discussion of the limitations with our various detection methods for root resorption. Evidence examining the association between root resorption and orthodontic treatment will be based on a systematic review of the literature organized by patient-related risk factors and orthodontic treatment-related risk factors. Results from randomized controlled trials will be highlighted in the review, and any conclusions presented will be based on following the research findings through the hierarchy of evidence. Prevention and treatment options for OIIRR based on the existing evidence will be discussed. A general summary with recommendations for future research will be provided.
Background and Etiology of External Apical Root Resorption
Root resorption (RR) is defined as microscopic areas of resorption lacunae visualized with histological techniques (Hartsfield et al. 2004).
The etiology of root resorption requires two phases: injury to the external surface of the root yielding denuded mineralized tissue and extended stimulation of multinucleated cells (Trope 1998). Denuded, mineralized tissue is colonized by multinucleated cells that initiate the resorption process. Without further stimulation, spontaneous repair will occur with cementum-like material within 2–3 weeks (surface resorption). However, if the stimuli remain, the inflammatory process on the denuded root will persist, and when it involves the deeper root dentin, it can then be detected radiographically (Fuss et al. 2003).
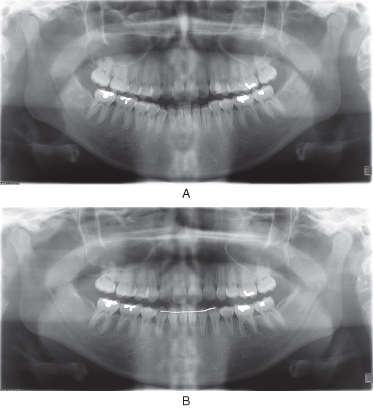
Apical root resorption may be observed during or at the end of treatment. Root resorption begins approximately 2–5 weeks into treatment, but it takes 3–4 months before it can be detected radiographically (Figure 5.1). It is a complex biological process for which many aspects remain unclear but that occurs when forces created at the root apex exceed the resistance and reparative ability of the periapical tissues (Parker & Harris 1998).
Figure 5.1 (a) Pretreatment radiograph of a 22-year-old female with crowding and protrusion necessitating orthodontic treatment with extraction of four first premolars. (b) Active root resorption can be seen in this immediate posttreatment radiograph. Shortening of the root has occurred as a result of resorptive cavities coalescing at the apex, cutting off peninsulas of root structure.
There are three types of orthodontically induced inflammatory root resorption (OIIRR), and severity is determined by the extent to which root tissue is involved (Trope 1998; Brezniak & Wasserstein 2002a).
- Surface resorption: Only the outer cemental layers are resorbed and are then fully regenerated or remodeled once the etiologic factor has been removed.
- Deep resorption: The cementum and outer layers of dentin are resorbed and are usually repaired with cementum material. The final shape of the root after this resorption and formation process may or may not be identical to the original form.
- Circumferential apical root resorption: Tridimensional resorption of the hard tissue components of the root apex occurs, and root shortening is evident. When the root looses apical material beneath the cementum, no regeneration is possible and the resorption is irreversible, although sharp edges may remodel.
Orthodontic forces create pressure and tension zones within the periodontal ligament, resulting in an infiltration of osteoclasts into the area of compression, which then resorb bone. The necrotic tissue must be removed before tooth movement can progress. It accounts for the initial delay in orthodontic treatment and always precedes the root resorption process; it has been seen close to areas of root resorption coinciding with the pressure zones of the periodontal ligament (Kurol & Owman-Moll 1998).
These forces also act on cementum, which is more resistant to resorption than bone (Brezniak & Wasserstein 1993). In OIIRR the injury results from the pressure applied to the root during tooth movement. This pressure produces ischemic necrosis of the periodontal ligament (PDL) localized in the pressure area (Trope 1998; Brezniak & Wasserstein 2002a). Macrophage-like and multinucleated cells are activated by biochemical signals derived from the sterile necrotic tissue. The initial elimination of necrotic tissue takes place at the periphery of the hyaline zone. During removal of the hyaline zone, the nearby outer surface of the root can be damaged (Brezniak & Wasserstein 1993; Brudvik & Rygh 1994; Brezniak & Wasserstein 2002a). In severe cases the orthodontic pressure may directly damage the outer root surface, including cementoblasts and precementum layers. Macrophage and multinucleated cell activity continues until no hyaline tissue is present and/or the force level decreases. Resorption lacunae expand the root surfaces involved and thereby indirectly decrease the pressure exerted through force application. Thus, the decompression allows the process to reverse and the cementum to be repaired (Trope 1998; Brezniak & Wasserstein 2002a).
If the orthodontic forces are applied for several hours, multinucleated positive cells develop odontoclastic-like morphology and functions and begin a complete cementum resorption, denuding mineralized dentin areas (Brudvik & Rygh 1994; Brezniak & Wasserstein 2002a). At this stage the resorption is still reversible provided that the pressure exerted by the orthodontic force stops and the extension of the root-damaged area is limited.
In severe injuries, in which the damaged area is large or the time of the force application is long, multinucleated cells continue the resorptive process, also including the exposed dentin. Odontoclasts, which were activated by the persistence of the inflammation, produce a tridimensional, nonreversible, and radiographically evident root resorption (Figure 5.2) (Brezniak & Wasserstein 1993; Brudvik & Rygh 1994; Brezniak & Wasserstein 2002a).
Figure 5.2 Varying severity of root resorption in intruded teeth (15×). (a) Minor root resorption (see the arrows). (b) Severe root resorption (see the arrows).
From Han, G. et al., 2005. Root resorption after orthodontic intrusion and extrusion: an intraindividual study. Angle Orthodontist, 75, 912–918. With permission from the E. H. Angle Education and Research Foundation.

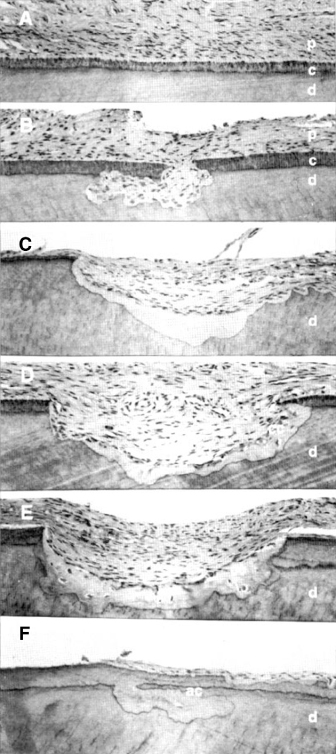
Following termination of orthodontic forces, root resorption typically ceases, and a progressive remodeling and reparative process of the root surface is evident. According to Schwartz (1932), this occurs when pressure falls below the optimal forces of 20–26 g/cm2. The resorption lacunae are filled in with new secondary cementum during periods of relative quiescence. Different spatial repair patterns have been reported, proceeding either from the periphery of the resorption cavity or from within the center (Owman-Moll & Kurol 1998). Repair areas also exhibit many combinations of cellular (fast process trapping cells) and acellular (slow process) cementum (Figure 5.3). However, if an island of cementum or dentine becomes separated from the root surface, it will be resorbed, and repair of the damaged root will be impossible (Proffit et al. 2006).
Figure 5.3 Histological illustration of varying degrees of repair in orthodontically induced root resorption in maxillary premolars after 1 to 8 weeks in retention. (A) Root surface in the cervical third of the root with normal dentin, acellular root cementum, and periodontal ligament. (B) Undermined root resorption in the cervical third of the root with no repair. (C) Partial repair with acellular cementum in the cervical third of the root. (D) Partial repair with cellular cementum in the cervical third of the root. (E) Total repair of the root surface with cellular cementum, but root contour has been altered. (F) Total repair of the root surface with acellular cementum, and original root contour was reestablished. Abbreviations: acellular root cementum, ac; cellular cementum, cc; cementum, c; dentin, d; periodontal ligament, p.
From Owman-Moll, P., Kurol, J. & Lundgren, D, 1995b. Repair of orthodontically induced root resorption. Angle Orthodontist, 65(6), pp. 403–408. With permission from the E. H. Angle Education and Research Foundation.
Prevalence and Detection of Orthodontically Induced Inflammatory Root Resorption
Histological studies report a greater than 90% occurrence of RR in orthodontically treated teeth (McLaughlin 1964; Stenvik & Mjor 1970; Harry & Sims 1982). In most cases the loss of root structure is minimal and clinically insignificant. Lower percentages are reported when diagnostic radiographic techniques are used. Lupi et al. (1996) reported the incidence of external apical root resorption (EARR) before treatment is 15% and after treatment, 73%.
Using panoramic or periapical radiographs, the average OIIRR is usually less than 2.5 mm (Linge & Linge 1983; Linge & Linge 1991; Mirabella & Artun 1995; Mavragani et al. 2000; Sameshima & Sinclair 2001a), or varies from 6 to 13% for different teeth (Blake et al. 1995).
Using graded scales, OIIRR is usually classified as minor or moderate in most orthodontic patients (Levander & Malmgren 1988; Janson et al. 1999; McNab et al. 1999; Brin et al. 2003). Severe resorption defined as exceeding 4 mm or one-third of the original root length is seen in 1–5% of teeth (Linge & Linge 1983; Levander & Malmgren 1988; Lupi et al. 1996; Taithongchai et al. 1996; Levander, Malmgren & Stenback 1998; Janson et al. 1999; McNab et al. 1999; Killiany 2002).
Regardless of genetic or treatment-related factors, the maxillary incisors consistently average more apical root resorption than any other tooth (Figure 5.4), followed by mandibular incisors and mandibular first molars (Newman 1975; Goldson & Henrikson 1975; Malmgren et al. 1982; Kennedy et al. 1983; Linge & Linge 1991; Kaley & Phillips 1991; Harris et al. 1997; Janson et al. 1999; Sameshima & Sinclair 2001a; Brin et al. 2003; Kook et al. 2003).
Figure 5.4 Pretreatment (a) and posttreatment (b) panoramic radiographs of a 12-year-old female with bimaxillary protrusion who experienced moderate root resorption of her maxillary incisors during orthodontic treatment.
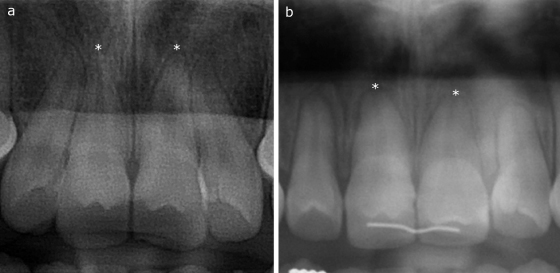
This may be because the extent of orthodontic movement of these teeth is usually greater than in the rest of the dentition, and the characteristic root structure of the incisors, along with their relationship to bone and the periodontal membrane, tends to transfer most of the forces to the apex (English 2001).
Adequacy of 2-D Radiographs to Analyze Root Resorption
The detection of RR has been mainly through radiographs, light microscopes, scanning electron microscopes, and micro-CT. Panoramic radiographs have been found to overestimate the amount of root loss by 20% or more when compared with periapical radiographs (PAs) (Sameshima & Sinclair 2001a). Although the paralleling technique with PAs has been described as the technique of choice for detecting root shortening, it has also been shown to be geometrically inaccurate (Katona 2006).
Periapical radiographs have been criticized as inaccurate in the assessment of root resorption (Katona 2006). It is believed that the compensatory algorithms are intrinsically ambiguous and incapable of reliably identifying constant length roots (nonresorbing and nongrowing), even in explicitly defined “perfect” teeth, and are further compromised by landmark identification.
Lateral skull cephalometric radiographs have been shown to yield an accurate and reproducible view of upper incisor length, but is subjected to a 5–12% enlargement factor as a result of the radiographic set up (Chan & Darendeliler 2004b). Also, overlapping of the right and left side make the individual images unclear. While radiography is a valuable diagnostic tool in detecting RR, quantitative measurements are relatively poor and should be avoided (Chan & Darendeliler 2004b).
Clinically, radiographs are an important diagnostic tool in detecting OIIRR, but the varying degrees of magnification, and the limitations involved with 2-D measurement of a 3-D phenomenon make the quantitative value of radiographs questionable and geometrically inaccurate (Sameshima & Sinclair 2001a; Chan & Darendeliler 2004b; Katona 2006). Quantitative 3-D volumetric evaluation of root resorption craters has been found to be a feasible alternative with a high level of accuracy and repeatability (Chan et al. 2004a; Chan & Darendeliler 2004b; Darendeliler et al. 2004; Chan & Darendeliler 2006). A mathematical computer-based reconstruction of pre- and posttreatment dental images have also been shown to be a reliable technique for measuring root resorption (Reukers et al. 1998). It is important that future studies use accurate, reliable, and valid measurement tools so meaningful comparisons can be made.
Evidence for the Association Between Root Resorption and Orthodontic Treatment
Even in the absence of orthodontic treatment, root resorption can and does occur (Al-Nazhan 1991; Counts & Widlak 1993; Rivera & Walton 1994; Snelgrove 1995).
Patient-Related Risk Factors
Individual susceptibility is considered a major factor in determining root resorption potential with or without orthodontic treatment (Segal et al. 2004; Hartsfield et al. 2004; Pizzo et al. 2007).
History of Root Resorption
There is a high correlation between the amount (number of teeth involved) and the severity of root resorption present at the start of treatment to the root resorption present when the orthodontic appliance is removed (Brezniak & Wasserstein 1993, Drysdale et al. 1996; Hamilton & Gutmann 1999; Hartsfield et al. 2004).
Tooth Root Morphology
Through mostly observational studies, abnormal root shape and other dental anomalies have been reported as risk factors for OIIRR (Levander & Malmgren 1988; Kjaer 1995; Thongudomporn & Freer 1998; Sameshima & Sinclair 2001a; Sameshima & Sinclair 2004; Smale et al. 2005). However, other investigators have found no significant correlation between tooth anomalies and OIIRR (Lee et al. 1999; Brin et al. 2003) or significant correlations between peg-shaped roots or microdontia of lateral incisors and OIIRR (Kook et al. 2003).
There is a common belief that short roots undergo more resorption (Newman 1975; McFadden et al. 1989). A more recent study supports the opposite view; the tendency for resorption increases with increasing tooth length (Mirabella & Artun 1995). A possible explanation may be that long teeth need stronger forces to be moved, so the actual displacement of the root apex is greater during torquing movements (English 2001).
Genetic Influences
Several studies support the idea that there is a genetic influence on OIIRR (Harris et al. 1997; Hartsfield et al. 2004; Ngan et al. 2004). A study of 123 sibling pairs treated with the same technique by a single operator showed that there were significantly greater variances among than within sibling pairs (Harris et al. 1997).
More recently, OIIRR has been linked to the IL-1B gene, substantiating an important genetic predisposition to this problem. It has been observed that the presence of IL-1B allele 1 is associated with a high risk of OIIRR (homozygous has 5.6 times increased risk than nonhomozygous [Al-Qawasmi et al. 2003a]).
Another candidate gene for EARR is TNFRSF11A, or another genetic locus found at the D18s64 polymorphic marker level, that encodes a TNF receptor that mediates signaling loading to osteoclastogenesis contributing to OIIRR during orthodontic treatment (Al-Qawasmi et al. 2003b).
Sameshima and Sinclair (2001a) reported that Asian patients have significantly less root resorption than Caucasian or Hispanic patients. However, the statistical analysis on these data does not clearly show whether this is due to genetic factors or to differences in orthodontic treatment approaches (Bollen 2002).
Systemic Factors
It has been demonstrated that root resorption can be reduced with the use of drugs and hormones.
Drug-Related Resorption
The main drug employed for resorption is bisphosphonate, which demonstrate a dose-dependent reduction of root resorption when administered in rats (Adachi et al. 1994; Igarashi et al. 1996). However, it has also been demonstrated that these drugs produce cemental surface alterations by inhibiting acellular cementum formation, increasing the vulnerability of the dental root to the resorptive process (Attati & Hammarstrom 1996a; Attati et al. 1996b).
Nabumetone, a drug belonging to the NSAID group, reduces the amount of root resorption along with the control of pain from intrusive orthodontic forces without affecting the pace of tooth movement (Villa et al. 2005).
Hormone Deficiency
It is thought that endocrine disturbances including hypothyroidism and hypopituitarism may play a role in the occurrence of root resorption.
In rats it has been demonstrated that administration of L-thyroxine increases the resistance of cementum and dentin to osteoclastic activity (Poumpros et al. 1994). Shirazi et al. (1999) found that the administration of increased doses of L-thyroxine to rats resulted in the reduction of the extent of root resorption.
A reduction of root resorption in humans was reported after the administration of the thyroid hormone thyroxine in three patients with no thyroid conditions (Loberg & Engstrom 1994; Christiansen 1994). These results must be interpreted with caution when considering thyroxine administration to orthodontic patients as no randomized clinical human trials looking at L-thyroxine have been reported.
Asthma
It was recently reported that patients with chronic asthma have an increased incidence of EARR that is confined to the maxillary molars. This finding may result from the close proximity of the roots to the inflamed maxillary sinus and the presence of inflammatory mediators in these patients (McNab et al. 1999; Brezniak & Wasserstein 2002b). The inflammatory mediators produced in asthma may enter the periodontal ligament and act synergistically to increase susceptibility to resorption. Although the asthma group had elevated incidence of EARR, both asthmatics and healthy patients exhibited similar amounts of moderate and severe resorption (McNab et al. 1999).
Root Proximity to Cortical Bone
It is unclear if proximity to cortical bone is related to root resorption. Kaley and Phillips (1991) reported that the risk of root resorption was 20 times greater when the maxillary incisors were in close proximity to the cortical plate, but roots were assessed with panoramic radiographs and cortical plates were assessed with lateral skull cephalograms. A similar study that used only cephalometric radiographs reported a much weaker correlation between the proximity of the root and cortex with the degree of OIIRR (Horiuchi et al. 1998). Another study using only cephalometric analysis failed to show any association (Otis et al. 2004).
Alveolar Bone Density
It had been postulated that tooth movement in dense bone requires greater or longer force application and consequently results in more root resorption (Rygh & Reitan 1972). In a rodent model, teeth were shown to move faster and still develop OIIRR in animals that had an induced decrease in bone density (Goldie & King 1984). A similar study in beagle dogs showed that tooth movement was slower in denser dentoalveolar complexes (Midgett et al. 1981).
In a human study, the amount of alveolar bone around the root, thickness of cortical bone, density of the trabecular network, and fractal dimension showed no significant correlation with the extent of the OIIRR (Otis et al. 2004).
Chronic Alcoholism
Chronic alcoholics receiving orthodontic treatment are at a high risk of developing severe root resorption during the course of orthodontic treatment (Davidovitch et al. 1996).
Previous Trauma
Historically, there has been a belief, based mostly upon observational data and animal studies, that all teeth with a previous history of trauma are more susceptible to OIIRR than healthy control teeth (Linge & Linge 1983; Linge & Linge 1991; Andreasen 1985).
Other investigators have reported that teeth with slight to moderate injuries may not have any greater tendency for root resorption during orthodontic treatment than do uninjured teeth (Malmgren et al. 1982). Data from a recent systematic review included three randomized clinical trials (RCTs) that met inclusion criteria. Incisors with clinical signs or patient reports of trauma (but no signs of EARR) had essentially the same prevalence of moderate to severe OIIRR as those without trauma (Levander, Malmgren & Eliasson 1994; Brin et al. 2003; Mandall et al. 2006).
There is a lack of RCTs that examine patients with previously traumatized teeth that have exhibited root resorption before orthodontic treatment. Observational data indicate orthodontic tooth movement will contribute to the resorptive process. (Brezniak & Wasserstein 1993; Drysdale et al. 1996; Hamilton & Gutmann 1999; Hartsfield et al. 2004).
Endodontic Treatment
Some investigators have reported an increased frequency of resorption in endodontically treated teeth (Wickwire et al. 1974).
It has been demonstrated that the endodontic treatment does not represent a risk factor for OIIRR. Teeth with previous root canal treatment exhibit a lesser propensity for apical root resorption during orthodontic tooth movement (Mirabella & Artun 1995; Drysdale et al. 1996; Hamilton & Gutmann 1999; Brezniak & Wasserstein 2002b). It has been suggested that endodontically treated teeth are more resistant because of an increased dentine hardness and density (Remington et al. 1989).
Severity and Type of Malocclusion
The amount of orthodontic tooth movement is associated with the resulting EARR (Mirabella & Artun 1995; Parker & Harris 1998; Hartsfield et al. 2004; Segal et al. 2004; Fox 2005). In turn, the required amount of tooth movement is a function of the severity of the malocclusion, which makes the presence of a severe malocclusion a risk factor for EARR.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


