4
Setting the Stage: Adjunctive Surgical Procedures, Restorative Options, and Treatment Planning
Take the necessary steps to gain an understanding of adjunctive procedures, the surgical/restorative options for implants, and what evaluation steps are needed to develop a comprehensive implant treatment plan.
A new frontier of implant dentistry is interdisciplinary, with coordination of therapy: auxiliaries, dentists, lab technicians, and specialists all work together to provide regeneration dentistry! The restorative dentist is the “quarterback of the team” and the hygienist plays the key role of helping to identify the wants and needs of the patient for replacing hopeless or missing teeth with implants. The specialist is usually the member who has the extra training in either the surgical aspects or advanced restorative techniques that are very often necessary in more challenging cases.
Hygienists can take the lead to evaluate necessary adjunctive procedures and to discuss wants, needs, and restorative options with the patients. Treatment planning is essential and most of all the patient’s questions, financial concerns, and final expectations must all be addressed prior to the first surgical procedure. Talk with your patients on what to expect with implant therapy.
To place implants you need a proper foundation of bone. Gingival tissue follows the biologic type and anatomy of the bone. To achieve optimal treatment results, the dentist needs to “set the stage.” Implant dentistry and regenerative adjunct procedures are no longer something for the future; they are a predictable and effective way to assure successful implant dentistry.
As hygienists, how can we effectively educate our patients on the pros and cons of a restorative option or adjunctive procedure without understanding the process ourselves? There are many facets to implant placement that might require additional procedures before or after placement to ensure long-term implant success. Some are necessary to support a proper foundation and others are indicated to provide optimal esthetic results.
Adjunctive surgical procedures
Contributed by Robert Horowitz, DDS
Adjunctive procedures in implant dentistry are on both the surgical and restorative sides. To have an ideal, esthetic, implant-supported restoration, the implant must be placed in as ideal a location as possible. There needs to be sufficient alveolar bone to support the gingival margin and papillae that will frame the proximal surfaces of the implant-supported fixed restoration. An optimal volume of bone is required to obtain sufficient osseointegrated support for facilitation of occlusion and force transfer from the restoration to the alveolar bone. To achieve these “simple” goals, there is a multitude of surgical and restorative options to obtain the necessary osseointegrated support or retention for the planned prosthetic device.
Surgical therapy
Implant dentistry begins with a tooth that has to be extracted or has already been lost. Improper extraction socket therapy is a major concern related to either the inability to place implants at all or functional and/or esthetic issues related to the resulting implant-restorative complex. Case reports, studies and meta-analysis papers have all reinforced what is seen clinically on a daily basis.
When a tooth is extracted, if no socket preservation or augmentation strategies are performed, there is concomitant loss of both hard and soft tissue. More than 45 years ago,. Pietrakovsky, Massler, and their coworkers performed a dried skull study (1) that verified where teeth had been lost on the “patients,” plus identified there was bone loss. The resulting alveolar ridge had collapsed from both the buccal side and occlusally. What bone remained in the alveolar ridge was shorter and more lingually located than when the teeth had been present.
Yes, leaving the socket with no graft material does lead to a “fill” with vital alveolar bone as described by Amler in the 1960s (2). This was one of a number of papers that validated the conclusion that leaving a blood clot to heal in an undisturbed manner will leave vital bone suitable for osseointegration in the area. However, if we follow the work by Massler and others, this may not leave sufficient bone nor in the ideal location to either place a dental implant or a conventional prosthetic replacement for the missing tooth or teeth.
The most critical component of extraction therapy is the removal of the affected tooth with minimal destruction to either the alveolar bone or surrounding gingival tissues. The group of techniques incorporated into the achievement of this goal is known as atraumatic extraction (3). As the periodontal ligament is what holds the nonankylosed tooth in place, that component of the periodontal apparatus must be stretched and broken to get out the root or roots with maximal preservation of the structures adjacent to the tooth. It is the surgeon’s responsibility to have the appropriate instrumentation required to remove the tooth and root, leaving the bone and gingival region as intact as possible. The first instrument utilized in this process is the periotome.
The periotome (see Figure 4.1) is a single- or double-ended instrument that is used manually or with a mallet in the PDL space. The goal is to widen the potential space of the PDL into a wider area that can accept larger instruments. In some cases, the tooth or root can be extracted using only this type of instrument.
Figure 4.1 Example of serrated periotome. Courtesy of PDT, Inc.
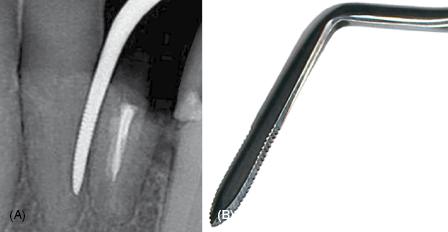
Other teeth require more “aggressive” instrumentation to remove them. Multi-rooted teeth are sectioned so that individual roots can be removed. Fewer macro and micro fractures will affect the facial and lingual plates of bone when single roots can be taken out of the jaw. Piezosurgery (4) and high-speed rotary instruments can be used when teeth are severely broken down and have no mobility whatsoever. Depending on the cross-sectional form and longitudinal shape of each root, many can be rotated to increase their individual mobility, facilitating atraumatic removal. Removing individual roots should also decrease the chance of fracture and flap elevation.
As Fickl and coworkers have shown in animal studies (5), surgical access at the time of extraction increases volumetric bone loss. In their study, the lingual portion of the socket lost 0.5 mm of horizontal width when no grafting was performed, regardless of facial flap elevation. The buccal plate resorbed 1.5 and 1.8 mm at the two treated sites 2 months after extraction in the nontreated sockets. By 4 months after extraction, those measurements increased to 1.5 and 2.1 mm of resorption. Where flap elevation and repositioning was performed, the site collapse was 1.9 and 2.5 mm 2 months after the procedure and 2.0 and 2.5 mm 4 months later. As the numbers demonstrate, there is significantly less socket resorption when a flapless approach can be accomplished at the time of extraction.
Socket preservation
Extraction socket preservation is essential to minimize the amount of bone and gingival loss following tooth removal. Studies in animals and humans have documented a multitude of strategies that have been attempted over the years to achieve certain goals. Our patients want the fewest number of procedures in which maximal results can be obtained. It is up to the dentists who are treating the patient to come up with their objectives for each and every individual site.
There are short- and long-term goals to be realized after a tooth is removed. We wish the patient to be free from dental disease and discomfort as quickly as possible. The maximal volume of bone and keratinized tissue should be preserved to support or esthetically frame the final prosthesis inserted in the site. True regeneration of vital bone with keratinized tissue in the area is the ideal result.
Many classes of bone replacement graft materials and barriers have been used while trying to improve the simplicity and predictability of reaching our socket preservation goals. Analysis of the results of these procedures has to be framed in light of the desired outcomes. Case reports, human and animal controlled studies, and meta-analyses have all been done to assist the surgeon in choosing the materials he/she will use to place in and/or over the socket immediately after tooth extraction.
Barriers and grafts of varied types have been used with significantly different outcomes depending on the endpoint desired by the surgical/restorative team to be working on the patient. A number of these materials will be described here for illustrative purposes.
A barrier membrane is inserted over an untreated or grafted socket. The goals are to graft or for blood clot protection and enhancement of guided bone regeneration by preventing migration of epithelium or connective tissue into the area.
Bone replacement grafts can be synthetic or can come from the same patient or from another human. Once they are placed in the body, they can remain for a short time, long time, or may never be removed. These materials can have some or no mineral content and can degrade quickly, slowly, or not at all. The specific formation or processing of the material will determine the amount and time of restorability, which can affect the amount of vital bone formation. It is the formation of vital bone that is critical for osseointegration, as osteocytes must be present in the grafted or treated site to enable bone-to-implant contact. That light microscopic contact with vital bone will enable force transfer from the prosthesis to the jaw and ensure stability of the bone–implant interface over time.
For proper physiology, the bone must resorb and remodel as occlusal stresses are transferred through it. Having osteoclasts acting on the vital bone and any residual graft material is important for this process to maintain homeostasis. An analysis of the time of resorption/replacement of the graft has to be weighed against the amount of bone formation desired and volume preservation expected. All of this has to fit into the discussions with the patient as to when, after extraction, an implant should be placed and then safely, predictably loaded with a prosthetic device.
Graft and/or barrier protection of the socket
As aforementioned studies have taught us, leaving a socket untreated will enable it to fill with vital bone. That is at the cost of 35% or greater loss of height on the facial and up to 50% of the width, primarily from the facial as well. There are concerns if graft particles are placed alone in an extraction socket. Particle loss or infection can lead to loss of the preservation and/or increase attempted. With no barrier to keep out the quicker migrating epithelial or connective tissue cells, there is potential for more connective tissue encapsulation of the graft particles and less vital bone formation. Refer to Figure 4.2.
Figure 4.2 Socket preservation case. Courtesy of Dr. Robert Horowitz. (A) Initial tooth and abscess. (B) Socket grafted with barrier by MIS. (C) Cone beam scan implant planned. (D) Screw-retained provisional crown. (E) Implant placed and site healed. (F) Final restoration.
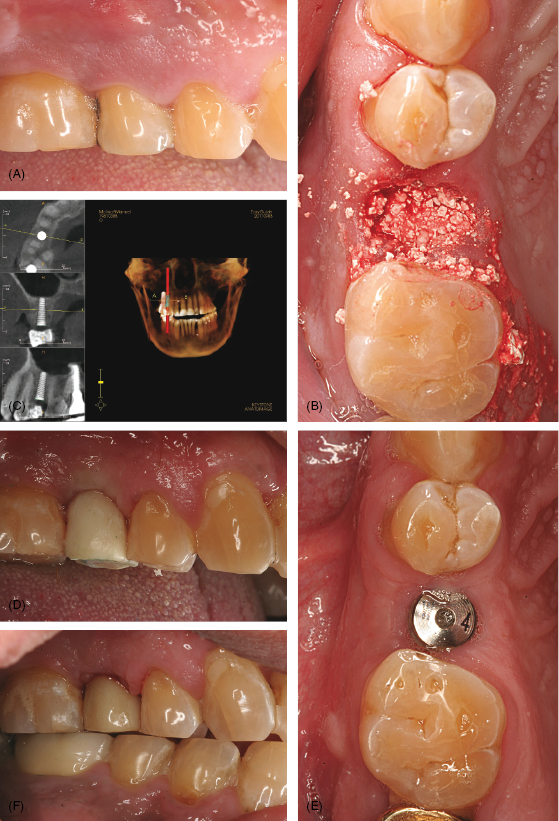
Tal and coworkers (6) described this in a study. They grafted extraction sockets in humans with anorganic bovine bone mineral. Fifteen patients requiring extraction and bone preservation of nonmolar teeth were included in the study. After extraction and flap elevation, sockets were debrided and filled to ideal contour with anorganic bovine bone mineral. As a containment barrier, a rotated split palatal connective tissue flap was used. Nine months later, the areas were exposed for placement of implants. The buccal plate resorption (in all but three cases where none was evident) ranged from 2 to 12 mm, with an average of 5.64 mm.
There was almost 20% resorption of the buccal bony walls. Some of the cases exhibited lingual or palatal resorption of between 3 and 6 mm, as well as the significant buccal bone loss. Cores of material were retrieved at the time of implant placement from the central portion of the sockets. These were demineralized and processed for histologic analysis for residual graft, vital bone ingrowth, and amount of connective tissue in the site. Apically, there was up to 60% vital bone, compared with only 16% coronally. Clinically, the anorganic bovine bone mineral (ABBM) particles were evident at the crest in all cases. Histologically, there was 30% residual graft present throughout the cores.
The authors concluded that resorption of the graft material was extremely slow from this data. They also stated that “the ultimate bone substitute should induce/conduct new bone formation and eventually completely resorb and be replaced by bone.” Other authors (7) similarly agreed with the conclusion that anorganic bovine bone mineral did not contribute to bone formation in an extraction socket and did not advise the use of this material to enhance vital bone-to-implant contact.
Removable barriers can work well when primary closure is obtained and maintained over them. In a dog model, Stavropoulis (8) studied grafted defects that had barrier protection. Two different types of resorbable barriers were tested in intra-alveolar defects that were created in healed sites. Both of the resorbable barriers led to vital bone formation underneath them. There were variations between the collagen and synthetic barriers with respect to the amount of vital bone formation and connective tissue in the histologic analysis. The sites protected by the synthetic barrier retained more of the ideal shape of the alveolar ridge and more vital bone was formed. It is possible that the collagen barrier (Bio-Gide, Geistlich AG, Switzerland) had a few properties less desirable for a GBR membrane. As a porcine collagen, it may have not retained its shape as well as the other barrier (Gore Resolut Adapt, WL Gore and Associates, Flagstaff, AZ).
Another possibility is that as a barrier, the porcine collagen is very soft and has little memory. It may not have had the same space-maintaining characteristics as the synthetic barrier, no matter the pore size used. Compared with the two barrier protected sites, the control demineralized freeze-dried bone allograft in a thermoplastic gelatin matrix (Regeneration Technologies Inc., Alachua, FL) was observed to have significant volumetric deformation compared with the barrier protected sites.
Through collapse of both the buccal and lingual sides of the grafted region, a knife-edge shape was often realized after healing. The synthetic barrier groups consistently had less soft tissue and a greater volume of bone formed than the collagen-protected grafted sites. These same sites also had more vital bone than either the control or the collagen barrier sites. Had these barriers become exposed and resorbed prior to epithelialization, the results for all regions could have been worse. Results like this were demonstrated by numerous authors, including Becker and Becker (9). In their case reports, sites that had guided tissue regenerative barriers became exposed during healing and formed less bone. These sites were more prone to infection and some even lost native bone around the defects that were treated.
Retrievable barriers alone have been used successfully in a number of studies. There are significant differences between dense and expanded polytetrafluorethylene (PFTE) barriers. Studies such as those by Becker (9), Zitzmann (10), and others have shown bone loss with exposed expanded PTFE (ePTFE) barriers. The same occurrence is not true with the dense PTFE barriers. Building on the research of others, in 2005 Horowitz (11) published a series of cases showing the use of dPTFE alone for socket volume preservation and bone regeneration. Techniques for atraumatic extraction were reviewed to ensure extraction is performed with no or minimal flap elevation. After debridement of the socket, no graft was placed; the volume was allowed to fill with a blood clot.
Clinically, though width measurements were not given, a significant width of the original socket was preserved. Histologically, as no graft was placed in the socket, there was no residual material that could potentially interfere with osseointegration of the implant inserted into the healed area. Vital bone was formed throughout the core to the region where the barrier was placed. Despite an average vital bone percentage of 28%, there were very thick and dense trabeculae at the apical portion of the socket with a more cancellous nature coronally.
Expanding on this study, Hoffmann and coworkers (13) treated patients in a similar manner and evaluated 276 sockets. No graft material was placed in the socket under the barrier membrane. As in the other studies, primary closure was not obtained due to the design of the material. Unlike expanded PTFE, which has been shown to have bacterial penetration through the barrier in 30 days when left exposed, this membrane is nonporous and does not have to be buried. It is removed approximately 3 weeks postoperatively or when one edge of the barrier becomes exposed.
A small percentage of the sockets was evaluated histologically after 12 months of healing. They appeared full of vital bone and having a consistent preservation of socket volume. These and other studies confirmed Amler’s original work (2), stating that a blood clot left undisturbed in an extraction socket will predictably form bone in a 3-month time frame. Adding a dense PTFE barrier to the treatment protocol increased the amount of the original socket volume that was preserved. In the absence of both graft material and the need for flap surgery, the extraction socket was regenerated and filled with vital bone. This is an ideal result from both the patient’s and surgeon’s standpoint. Only one surgical visit is required, with maximal physical and biologic results in terms of bone preservation and keratinized tissue augmentation.
Bone graft materials are categorized depending on their source. Autogenous bone is retrieved from the same patient, usually in a different site. Intraorally, cortical shavings are taken from the ramus. Cancellous bone is removed from osteotomies or using trephines from anywhere in the mouth. Monocortical blocks and corticocancellous blocks that are primarily used for ridge augmentation are removed from the anterior mandible or the ramus. Extraoral donor sites from which a patient’s own bone can be removed include the calvarium, iliac crest, and tibia.
Allograft or donor bone is processed from another member of the same species and can come in any of four forms. The material can be retrieved from either a cancellous or cortical location and can either be obtained as either mineralized or demineralized freeze-dried bone graft substitute.
Xenograft is retrieved from a member of another species and is processed in a steam or sintered manner to remove some, most, or all of the proteins. The primary sources of these materials are cows, pigs, and horses. These materials have been shown in some studies (6) to be practically nonresorbable and act primarily as a scaffold on which new bone is deposited.
Alloplastic materials are synthetic in nature. These are calcium-containing “salts” in the families of hydroxyapatite, calcium sulfate, or tricalcium phosphate. Particle size, three-dimensional structure, and chemical composition all play a role in the resorption or dissolution rate of the graft material.
Earlier in this chapter, studies on xenografts were mentioned, indicating they were not ideal to use where regeneration is desired in extraction socket therapy (6). That paper reinforced what had been shown in other papers in that resorption of anorganic bovine bone was not clinically nor histologically seen in the time frame ideal for placement of dental implants. Similar results were seen in a paper by Carmagnola in 2003 (14). In 21 patients, 31 sockets were grafted with either anorganic bovine bone, covered with a resorbable barrier, or left untreated. Timing for re-entry and histologic specimen retrieval was not standardized. The sockets that were covered with the resorbable barrier had, at approximately 4 months postoperatively, 52% vital bone. There was a very high standard of deviation of 16%, leading to the conclusion that results were not consistent.
Where the sockets were untreated, re-entry was performed between 1 and 15 years postoperatively with a resulting 56% vital bone, with almost a 20% standard deviation in results. The poorest histologic result was when anorganic bovine bone mineral was inserted into the socket at the time of extraction. The remaining ABBM particles were 21% with only 8% vital bone about 7 months after extraction. This is significantly less vital bone than shown by Horowitz (11), where a dPTFE barrier was used over no graft material, only a blood clot.
A clinical case report looked at the healing of sockets filled with demineralized, freeze-dried bone compared with autogenous bone in extraction sockets (14). Using rongeurs to test the most coronal portion of the healed area, Becker and his colleagues saw more vital bone formation when autogenous bone was used. Increasing the amount of vital bone formed around allograft particles has been shown by Sottosanti and others (15). In their randomized, controlled clinical trial on two types of graft and barrier protection of extraction sockets, in 24 patients, clinical and histologic analysis was taken of anorganic bovine bone mineral covered with a resorbable barrier or demineralized, freeze-dried bone allograft mixed with calcium sulfate in putty and covered with a calcium sulfate barrier (16). There was similar ridge width preservation for both groups and slightly better height maintained with the ABBM and barrier.
The differences in vital bone formation were significant. In the ABBM-treated sites, there was only 26% vital bone compared with 61% in the sites grafted with demineralized freeze-dried bone allograft (DFDBA) and calcium sulfate. Additionally, in the allograft-treated sites, there was only 3% residual graft material compared with 16% remaining bovine bone material. This study demonstrated that the use of an allograft combined with calcium sulfate led to significant preservation of socket volume with a high percentage of regeneration of alveolar bone in the socket, significantly more than when ABBM was placed.
Along with its uses as a graft enhancer and barrier, calcium sulfate has also been used alone as a graft material in extraction socket therapy. In addition to the works by Pecora and others from Italy, there is a recent paper documenting the clinical and biologic advantages of a nanocrystalline calcium sulfate graft material. Mazor et al. (17) showed clinical preservation of volume after extraction of a severely infected mandibular molar tooth. The site was grafted with this new formulation of calcium sulfate hydrated in sterile saline and covered with a dense PTFE barrier. The barrier was removed in a nonsurgical manner 3 weeks postoperatively. Two-dimensional radiographs and the cone beam volumetric tomography confirmed a similar radiation density in the grafted site and the adjacent bone 4 months after the extraction at the time of implant placement.
Histologically, the area was filled with densely woven bone that amounted to 47% of the volume of the analyzed specimen. There was no residual graft material at that time. Furthermore, a 10-month post-placement radiograph demonstrated full preservation of the regenerated alveolar bone at the crest. All of those are ideal results to achieve in such a short time period.
Another alloplastic material that has received extensive evaluation is tricalcium phosphate. It is distributed in many forms, compositions and particle sizes. The alpha and beta forms resorb at different rates. Some of the manufacturers make their particles in a round shape, others in a more open lattice pattern that leaves more exposed surface area for dissolution when exposed to the extracellular matrix. T/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


