Management of Secondary Jaw Deformities after Maxillofacial Trauma
Posttraumatic Temporomandibular Joint Ankylosis in the Pediatric Population
Background
The treatment of TMJ ankylosis poses challenges to the maxillofacial surgeon as a result of technical difficulties associated with access to the joint, the currently available autogenous and prosthetic TMJ replacement options, and the high incidence of ankylosis recurrence. Failure to restore adequate mandibular opening is likely to result in speech and swallowing impairment; difficulty with mastication; poor oral hygiene and dental neglect; continued facial growth disturbances; and the potential for airway compromise. The surgical management of bony ankylosis of the TMJ requires the complete excision of the involved osseous mass with intraoperative achievement of satisfactory passive mouth opening. The immediate reconstruction with a costochondral rib graft is often carried out, and this is followed by a postoperative physiotherapy regimen to maintain mouth opening. A number of authors have critically evaluated the treatment of TMJ ankylosis in adults, with less emphasis placed on addressing this problem exclusively in the pediatric population.*
In a study by Posnick and colleagues, a consecutive series of nine pediatric patients (mean age, 7.7 years) who underwent a standardized treatment protocol for 13 affected ankylosed temporomandibular joints was reviewed.88 Four patients had unilateral TMJ ankylosis, and five had bilateral ankylosis. One child required bilateral release but only unilateral reconstruction. Radiographic evidence demonstrated bony ankylosis in all 13 operated joints. Two patients had previously undergone surgical intervention of the TMJ. The cause of ankylosis within the study group was primarily traumatic or congenital. The protocol that was followed included complete excision of the involved ankylotic structures with mobilization through coronal scalp and Risdon neck incisions as well as the achievement of wide mouth opening. This was followed by immediate costochondral grafting. Fixation with mini-plates and screws allowed for early mobilization with the rapid institution of a physiotherapy program (Fig. 35-1).
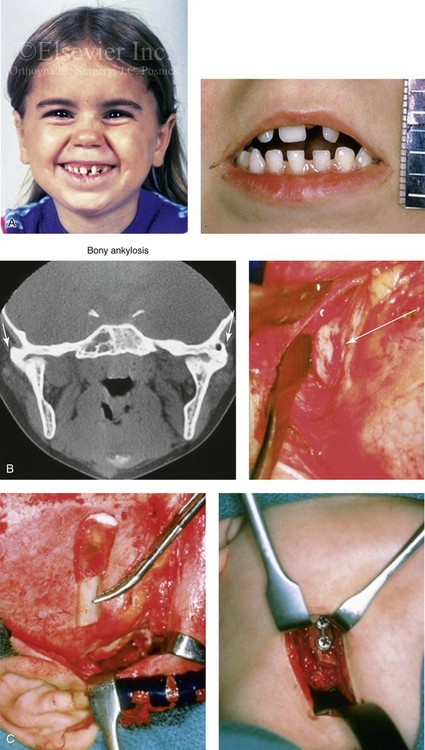
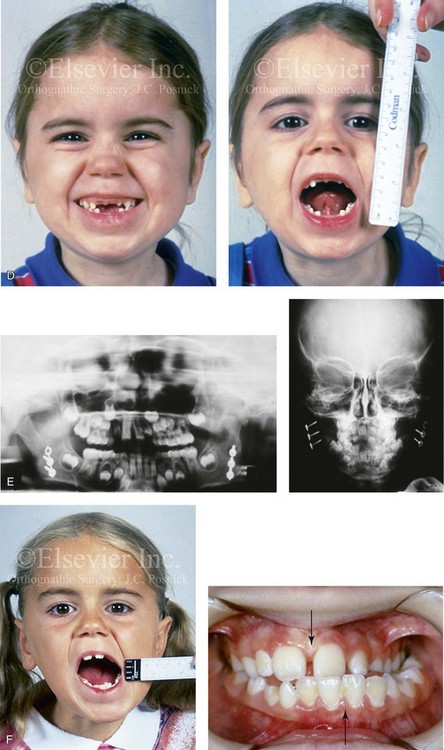
Figure 35-1 A 5-year-old girl was referred to this surgeon after intracapsular condyle fractures of the mandible resulted in bilateral temporomandibular joint ankylosis. These fractures were unrecognized and went untreated at the time of injury 1 year earlier. A, Preoperative frontal views showing maximal opening limited to 4 mm. B, Preoperative computed tomography scan that demonstrates the bony ankylosis of each condyle to its zygomatic arch. Intraoperative view demonstrates ankylosis to the zygomatic arch to the condyle as viewed through the coronal scalp incision. C, Intraoperative view of costochondral graft placement through the coronal scalp incision. Stabilization of the rib graft through Risdon neck incision with the use of a mini-plate and screws. D, Facial views 1 year after reconstruction showing good cranial nerve VII function and satisfactory vertical mouth opening being maintained. E, Postoperative Panorex and anteroposterior facial radiographs. F, Facial view with maximum mouth opening maintained 3 years after reconstruction but with asymmetrical overgrowth of the costochondral grafts. Occlusal view 3 years after reconstruction that shows an asymmetrical Angle class III malocclusion as a result of the overgrowth.
Controversies and Unresolved Issues
Knowing the cause of TMJ ankylosis helps with the understanding of its pathophysiology. In developed countries, the most common etiology is trauma, followed by infection.33,48,121,125,126 Topazian found an association with trauma in 39% and with infection in 43% of patients with TMJ ankylosis (N = 229).119 Since the late 1970s, authors have found trauma to be a more frequent cause than infection, probably because of the use of antibiotics to combat the latter. In a report of a series of patients with TMJ ankylosis, Rajgopal and colleagues concluded that 80% of cases were traumatic in origin.96 Unfortunately, these reviews lumped both pediatric and adult patients together.
Surgical attempts to release TMJ ankylosis have been described dating back to 1850.5 Since then, ankylosis release followed by reconstruction using a variety of alloplastic and autogenous materials has been repoted.9,15,34,46,50,53,62,68,72,79,81,101,114,124,129 Although Gillies first described costochondral grafting for TMJ reconstruction in 1920,30 it was Poswillo93,94 and MacIntosh and Henny53 who popularized its use during the 1970s. Poswillo attempted to demonstrate the histologic and physiologic similarities between the mandibular condyle and rib cartilage in humans.93 MacIntosh and Henny described 26 cases of costochondral grafting for mandibular condyle replacement, with 6 of them being for ankylosis.53 Their approach included graft stabilization with interosseous wires and intermaxillary fixation (IMF) for 6 to 8 weeks. They claimed subsequent growth of the grafts in their pediatric patients, but they failed to provide any objective data.
In 1973, Kennett reported on two cases of unilateral TMJ ankylosis in young patients who underwent the combination of condylectomy, coronoidectomy, and reconstruction with a wired rib graft.45 He concluded that ankylosis in children should be treated as soon as possible and that recurrence is the most frequent complication. In 1986, Munro and colleagues reviewed their series of 18 adult and pediatric patients with TMJ ankylosis and facial deformity.67 Their reconstruction included interosseous wiring of the rib graft and 8 weeks of IMF followed by physiotherapy. They demonstrated better results in unilateral ankylosis cases than in bilateral cases. The next year, Lindqvist and associates described 27 patients of varying ages with TMJ ankylosis; 25 of them underwent costochondral graft reconstruction. Fixation was also by direct interosseous wires, but IMF was reduced to 3.5 weeks to encourage early mobilization and to limit recurrence. The maximum incisor opening improved from 16 mm to 31 mm. In 1987, Politis and others reported satisfactory results in six patients (five adults) with ankylosis.81 They employed a preauricular approach that involved the use of costochondral grafts and either wire or plate and screw fixation followed by 2 or 6 weeks of IMF, respectively. However, 50% of their patients developed seventh cranial nerve palsies. In 1990, Kaban and colleagues described their experience in 14 adult and pediatric patients with TMJ ankylosis and indicated satisfactory results.41,42 The protocol reported by Posnick and colleagues was similar to that of Kaban and colleagues except that the former found that the use of a mini-plate with screw fixation improved stability (as compared with interosseous wire fixation), thereby allowing for the minimal use of IMF and the early initiation of mandibular range of motion with active physiotherapy.88
In 2009, Kaban and colleagues described an updated protocol for the management of TMJ ankylosis exclusively in children.78,79,122,123 They restated their observation that the most common cause of treatment failure was inadequate resection of the ankylotic mass followed by the failure to achieve adequate passive maximum opening in the operating room. Clearly, unless adequate passive mandibular vertical opening is achieved in the operating room, failure will inevitably occur. Unfortunately, this is no guarantee that mouth opening will be maintained. Their seven-step protocol consisted of the following:
1. Complete excision of the fibrous and/or bony ankylotic mass
2. Coronoidectomy of the affected side
3. Coronoidectomy of the contralateral side if steps 1 and 2 do not result in a maximum incisal opening (>35 mm) or opening to the point of dislocation of the unaffected TMJ
4. Lining of the TMJ with either a temporalis myofascial flap or the native disc, if it can be salvaged
5. Reconstruction of the ramus condyle unit with either proximal segment osteotomy and distraction osteogenesis or costochondral graft and plate and screw fixation
6. Early mobilization of the jaws: If distraction osteogenesis is used to reconstruct the ramus condyle unit, mobilization begins the day of operation. In patients who are undergoing costochondral graft reconstruction, mobilization begins after 10 days of IMF.
7. All patients receive rigorous physiotherapy after surgery to maintain mouth opening, generally for at least 3 to 6 months
Observations and Recommendations
Several generalizations can be made about pediatric patients with TMJ ankylosis. First, in first-world countries as in the adult population, trauma remains a prevalent cause, and this is followed closely by congenital issues. In underdeveloped countries, infection continues to be an important causative factor.92 Second, although patients with unilateral ankylosis present with a severely limited preoperative opening, they are more likely to achieve satisfactory long-term functional results. Third, children with bilateral congenital TMJ ankylosis generally attain poor long-term results, despite adequate intraoperative release. This is likely the result of associated masticatory muscle anomalies, neuromuscular discoordination, or longstanding muscular disuse atrophy. Fourth, the use of mini-plate and screw fixation of the graft diminishes the need for immobilization, thereby allowing for the early institution of physiotherapy. Unfortunately, this does not guarantee the maintenance of a satisfactory long-term opening. Fifth, when a costochondral graft is used in children, only several millimeters of cartilage should remain. This is to limit the frequent complication of overgrowth and its complex secondary deformities. In clinical practice, overgrowth after costochondral grafting remains a frequent occurrence (see Chapter 28 and Fig. 35-1). Sixth, for most patients, reconstruction to achieve long-term maxillomandibular harmony must wait until they have achieved skeletal maturity (i.e., 14 to 18 years of age).
Posttraumatic Saddle-Nose Deformity
Background
Nasal injuries are often recognized but then ignored as being unimportant.11,82,86 When a depressed nasal fracture occurs in a child, injury to the growth center leaves the nose prone to a so-called “saddle deformity” with flattening of the osseocartilaginous vault.63 In general, the overlying soft tissues, the upper and lower lateral cartilages, and the nasal lining are distorted but remain intact. Reconstruction of a saddle deformity that involves the bony and cartilaginous dorsum is generally carried out with an autogenous graft (e.g., chondral, costochondral, iliac, split- or full-thickness cranial) (Fig. 35-2).27,28,32,38,60,74,91,117,118,131 The use of an allogenic graft (e.g., porous polypropylene) is always a second choice as a result of a higher incidence of associated infection and extrusion.43 The type of graft material selected (e.g., bone, cartilage, allograft), the method of fixation, the incisions required for access, the extent of dorsum that requires reconstruction, and the timing of treatment are all important details and will vary according to the age of the patient, the extent of the deformity, and the surgeon’s personal preferences (see Chapter 38).
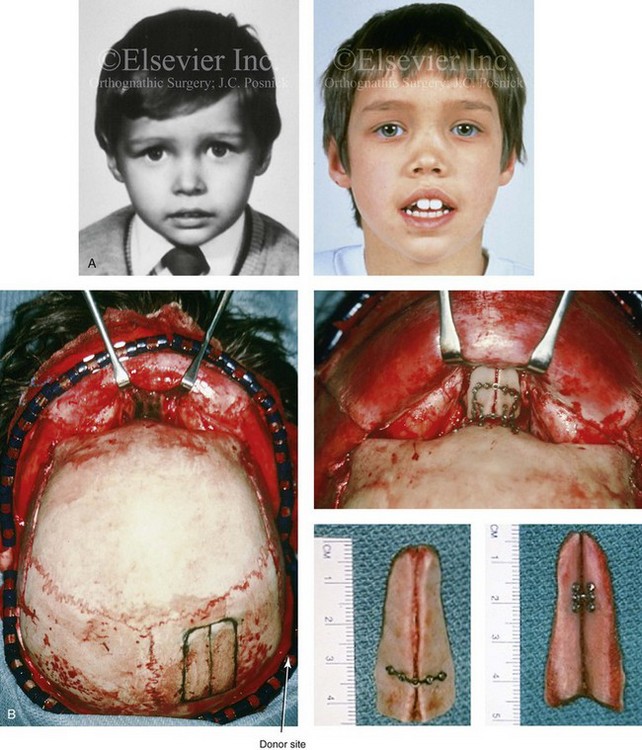
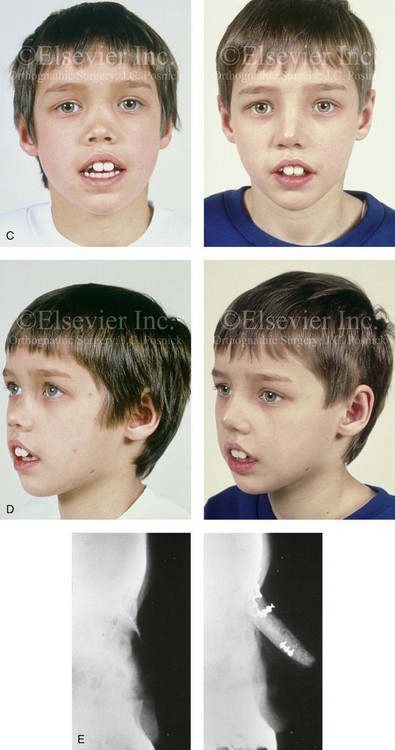
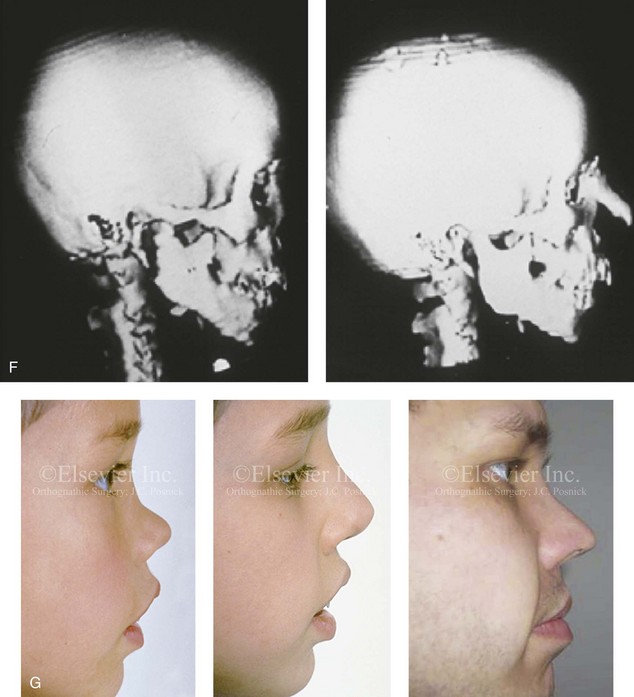
Figure 35-2 A 4-year-old boy fell and hit the bridge of his nose on a hard tabletop, which resulted in a saddle-nose deformity (i.e. osseous and cartilagenous vault). He arrived for evaluation when he was 8 years old and then underwent reconstruction through a coronal scalp incision with nasal osteotomies (in-fracture) and the placement of fixed crafted full-thickness cranial grafts. A, Frontal view at 3 years of age just before injury and at 8 years of age just before surgery. B, Intraoperative views through the coronal scalp incision that show the frontonasal region and the proposed right calvarial donor site. Crafted full-thickness cranial bone grafts are shown before placement for nasal reconstruction. Stabilization is achieved with microplates and screws. A close-up view of the frontonasal region shows the full-thickness cranial bone grafts in place and stabilized with microplates and screws. C, Frontal views before and 6 months after reconstruction. D, Oblique views before and 6 months after reconstruction. E, Lateral radiographs of the nose before and 6 months after reconstruction. F, Computed tomography scan views before and 6 months after reconstruction. G, Close-up lateral view just before surgery, 6 months after treatment, and then 16 years after reconstruction. A, B, C, E, F, from Posnick JC: The role of plate and screw fixation in the management of pediatric facial fractures. In Gruss JS, Manson PM, Yaremchuk MJ, eds: Rigid fixation of the craniomaxillofacial skeleton, Stoneham, Mass, 1992, Butterworth-Heinemann, p 1412.
Involvement of Both the Bone and the Cartilage Vault
The autogenous graft used (e.g., chondral, costochondral, rib, iliac, split- or full-thickness cranial) is contoured to provide reasonable nasal dorsum morphology. When the whole dorsum requires reconstruction (i.e., the radix to the nasal tip), the lower lateral cartilages are sutured over the top of the graft to provide a more natural tip contour and feel (see Fig. 35-2). When a bone graft extends from the radix to the tip, minor degrees of graft resorption at the tip should be anticipated. When correcting the saddle deformity with bone graft, freshening the base of the nasal bones with a rotary drill before graft placement is carried out; the onlay graft will then rest on a bleeding base. The accurately shaped graft is either dovetailed into a bony groove at the nasofrontal process or abutted to the frontal bones while resting evenly on the contoured and freshened)dorsal base to establish the correct nasofrontal angle. Stabilization of the bone graft is either with microplates and screws or transcutaneous Kirschner wires, depending on the access provided by the incisions that are used for the reconstruction. If a coronal scalp incision or a direct vertical nasal incision is used, plate and screw fixation is easily accommodated. An open approach (i.e., a columella splitting incision) should be combined when a coronal scalp incision is used to best manage the lower lateral cartilages and the nasal tip (see Chapter 38 and Fig. 35-2).
Involvement of the Cartilage Vault Only
When the saddle deformity causes collapse of the septal cartilage without injury to the nasal bones, then a cartilage graft reconstruction is preferred. A rib cartilage dorsal strut graft is crafted and placed flush with the nasal bones. The graft then extends caudal to the tip. The tip of the dorsal strut graft is joined to a second rib cartilage graft (caudal strut) that extends from the base of the maxilla to the nasal tip. The caudal strut is stabilized with a short buried Kirschner wire (no. 35 threaded) that is secured to the base of the maxilla/anterior nasal spine region and then pierced into the graft (Fig. 35-3). There is no advantage to harvesting the L-shaped rib cartilage graft as one unit. The separately crafted dorsal strut and caudal strut grafts are then joined together at the tip with non-resorbable suture. The lower lateral cartilages are sutured together and over the grafts to form the new nasal tip. This technique is efficient and results in a “soft” nasal tip that looks and feels natural (see Chapter 38).
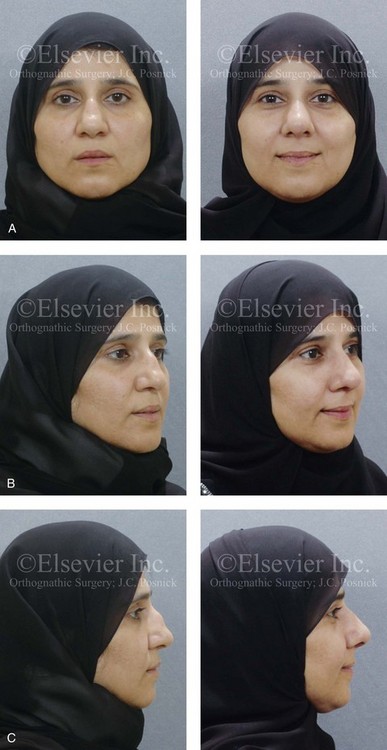
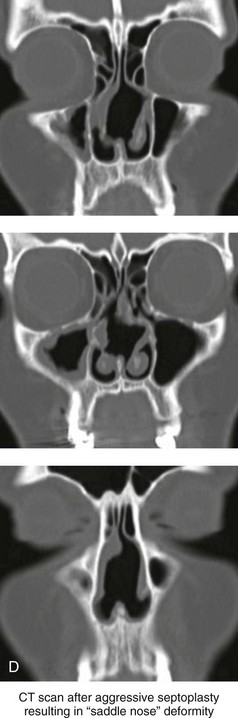
Figure 35-3 A middle-aged woman of Arabic origin with a postsurgical (iatrogenic) saddle-nose deformity. Two years earlier, she underwent an aggressive septoplasty procedure at another institution. This resulted in a large septal perforation with collapse of the cartilaginous vault. She arrived for evaluation and then underwent nasal reconstruction that included rib cartilage grafting (caudal and dorsal struts). A, Frontal views before and after reconstruction. B, Left oblique view before and after reconstruction. C, Profile view before and after reconstruction. D, Computed tomography scan views (coronal cut) of nasal septum confirming the extent of the septal deficits before reconstruction.
Posttraumatic Orthognathic Deformities
Background
Posttraumatic secondary maxillomandibular deformities may occur as a result of inadequate initial anatomic reduction or stabilization and fixation of a jaw fracture or the late resorption and remodeling of the bones resulting in malunion.23–25,35,101,105,111,115,132 The segmental loss of maxillary or mandibular dentoalveolar components may also have occurred, as discussed later in this chapter. In the child, limited growth of the bones after the initial injury (e.g., condylar fracture) or as a result of the surgical intervention carried out to reduce and stabilize the fracture may also cause secondary deformities.58,108,113
Malunion after Midface (Le Fort I) Fracture
An elongated face with an anterior open-bite malocclusion (counterclockwise rotated maxilla and clockwise rotated mandible) can occur when a Le Fort fracture is allowed to heal with inadequate reduction or stabilization at the time of initial management.30,40,87,132 The displaced maxilla often heals inferiorly with counterclockwise rotation. The mandible is forced into a clockwise-rotated position that involves a retrusive pogonion seen in profile. Another common reason for malocclusion after midface fracture is an unrecognized palatal split with secondary arch-form deformity.25,132 Reconstruction requires osteotomies to recreate the midface fractures followed by the surgical repositioning of the maxilla into the correct (preinjury) anatomic location. The uninjured mandible will then counterclockwise rotate to close the anterior open bite, to improve the horizontal projection at the pogonion, and to reduce the vertical height of the lower face.43,87 Another frequent scenario is when a midface fracture occurs during childhood and then results in growth disturbance. At the time of facial growth maturity (i.e., 14 to 18 years of age), maxillary hypoplasia with a skeletal class III malocclusion will be evident (Fig. 35-4).
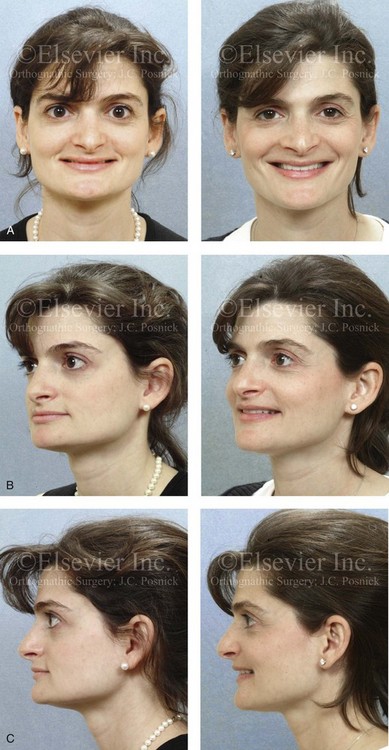
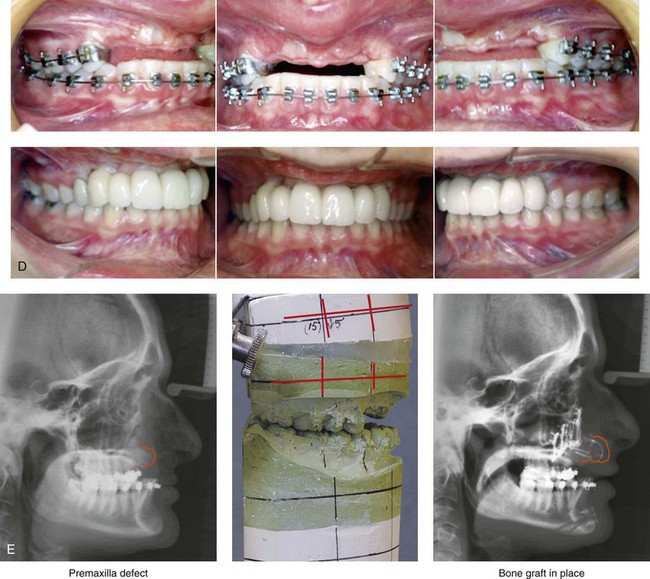
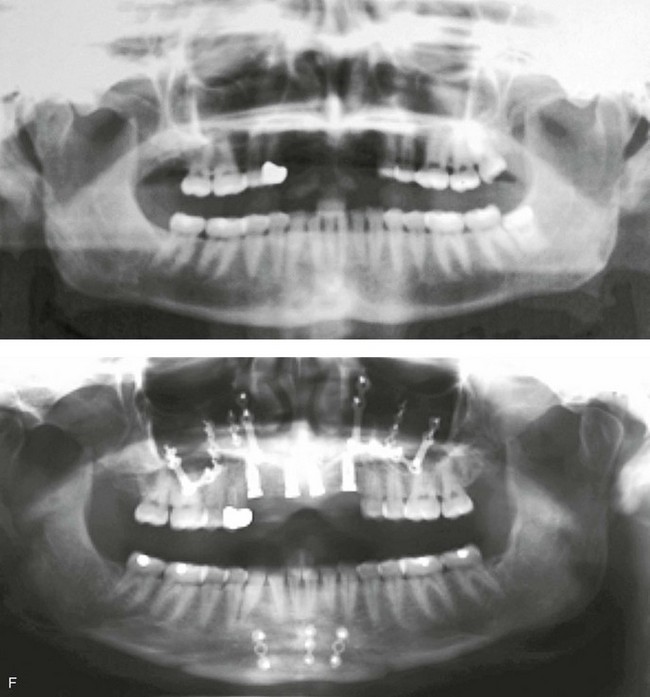
Figure 35-4 In this patient, midface trauma occurred during childhood and included the loss of the anterior teeth and the alveolar bone component. She presented during adulthood with maxillary hypoplasia, Class III malocclusion, and an anterior segmental dentoalveolar defect. Comprehensive reconstruction and dental rehabilitation involved a surgeon, a periodontist, an orthodontist, and a restorative dentist. The patient underwent Le Fort I osteotomy (horizontal advancement and vertical lengthening) with interpositional corticocancellous iliac bone grafting. A corticocancellous (iliac) bloc graft was simultaneously crafted to match the anterior maxillary defect; it was inset and secured with microplates and screws. This was followed 4 months later by the placement of four implants and then final restorations 6 months later. A, Frontal views with smile before and after reconstruction and rehabilitation. B, Oblique views before and after reconstruction and rehabilitation. C, Profile views before and after reconstruction and rehabilitation. D, Occlusal views before and after reconstruction and rehabilitation. E, Lateral cephalometric radiographs and analytic model planning before and after reconstruction. F, Panorex radiographs before and after reconstruction and rehabilitation.
Late Secondary Consequences of a Condylar Fracture
There are many published opposing treatment protocols for the primary management of mandibular condyle fractures.2,10,21,22,31,59,63–66,112,127,133 Despite much discussion, controversy remains regarding which type of condyle fracture should be treated open rather than closed and, if operated, what approach (i.e., incisions for access, extent of condylar fragment degloving, method of fixation, postoperative immobilization and remobilization protocol) is optimal for each specific fracture. In addition, when a condyle injury or fracture occurs in a child, it can easily be overlooked or misdiagnosed, especially if the individual has no other apparent injuries. The child may not even arrive for evaluation; if evaluation does occur, the patient may be seen by a general practitioner rather than a specialist. Radiographs may be limited as a result of the need for sedation or concern by the parents about ionized radiation exposure.49,83,84
Regardless of the primary treatment rendered, a frequently seen pattern of late secondary deformity results when the condylar injury occurs prior the completion of mandibular growth (Figs. 35-5 through 35-10).26,29,69,83–85,87,89,90,95,109,110 Often there is asymmetrical growth of the mandible from that point forward, with ipsilateral mandibular hypoplasia (see Chapter 4). Canting of the mandible (up on the ipsilateral side), shift of the dental midline (toward the fracture), and malocclusion (ipsilateral Class II) are frequently seen. The upper jaw is secondarily affected, growing with vertical asymmetry (canting) and with a shift of the maxillary dental midline toward the side of the fracture.
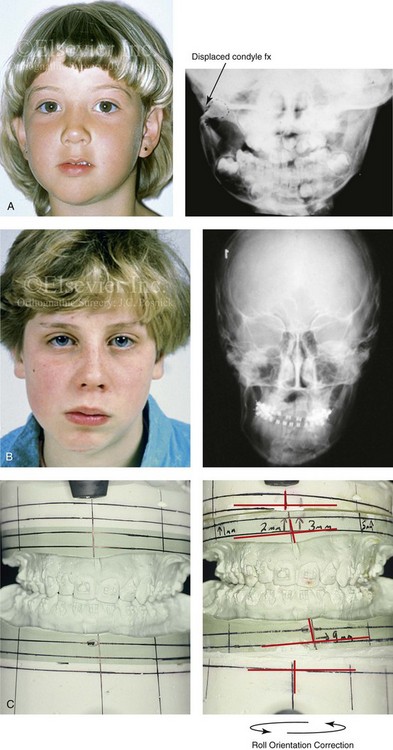
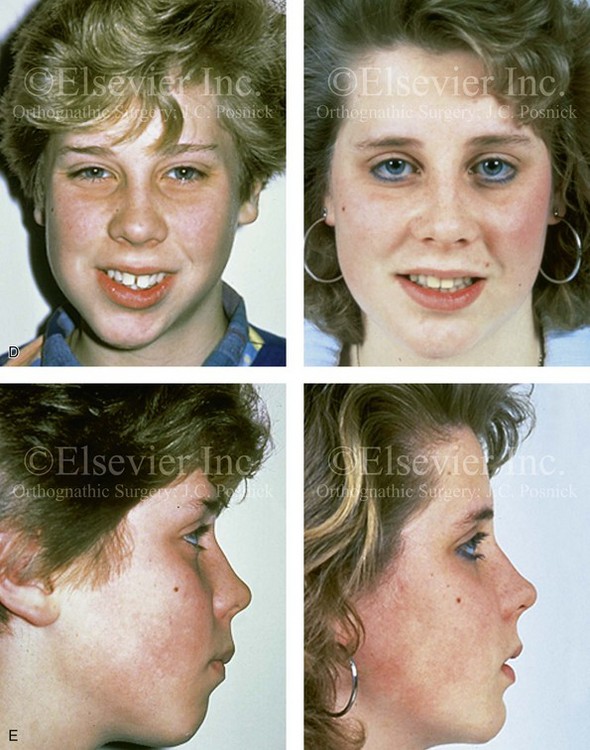
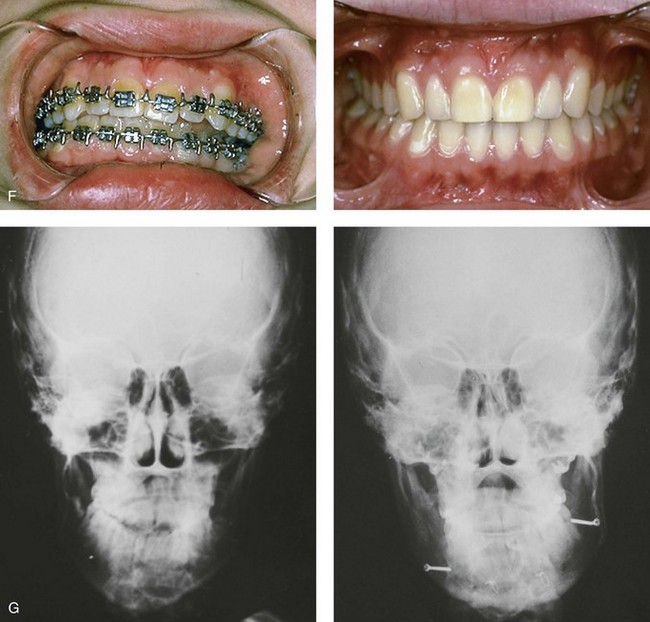
Figure 35-5 A 5-year-old girl sustained a right condyle fracture of the mandible and then presented as a teenager with resulting facial asymmetry that involved the maxilla and the mandible. She then underwent a comprehensive orthodontic and surgical approach that included a Le Fort I osteotomy; bilateral sagittal split ramus osteotomies; and an osseous genioplasty.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


