Aphthae (recurrent aphthous stomatitis)
INTRODUCTION
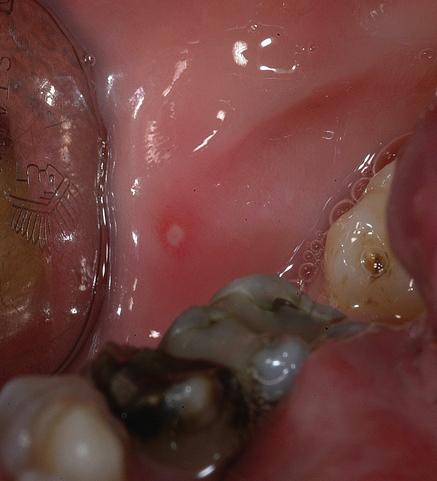
 With multiple recurrent, round or ovoid ulcers known as aphthae or canker sores, which have circumscribed margins, erythematous haloes, and yellow or grey floors (Fig. 34.1):
With multiple recurrent, round or ovoid ulcers known as aphthae or canker sores, which have circumscribed margins, erythematous haloes, and yellow or grey floors (Fig. 34.1):
It is important to note that individual aphthae last only a limited period of time, before they heal spontaneously. This is quite a different history from ulcers that persist without healing, such as malignant ulcers and those associated with vesiculobullous disorders such as pemphigoid and pemphigus.
PREDISPOSING FACTORS
 Stress underlies RAS in some cases and ulcers appear to exacerbate during school or university examination times.
Stress underlies RAS in some cases and ulcers appear to exacerbate during school or university examination times.
 Trauma from biting the mucosa or from dental appliances may lead to aphthae in some people.
Trauma from biting the mucosa or from dental appliances may lead to aphthae in some people.
 Cessation of smoking may precipitate or exacerbate RAS in some cases, but the reason is unclear.
Cessation of smoking may precipitate or exacerbate RAS in some cases, but the reason is unclear.
 Haematinic deficiency may be relevant in a minority. In up to 20% of patients, deficiencies of iron, folic acid (folate) or vitamin B are found and sometimes the correction of this may relieve the ulceration. Iron deficiency is usually due to chronic haemorrhage (e.g. from the gastrointestinal or genitourinary tract). Folic acid is found in green leafy vegetables especially, and body stores are small; deficiencies may be dietary, or related to malabsorption or drugs (alcohol, anticonvulsants, carbamazepine, and some cytotoxic drugs). Vitamin B12 is found especially in meat, is absorbed via intrinsic factor from the gastric parietal cells in the ileum and stored in the liver for about 3 years. Dietary B12 deficiency can arise particularly in vegans, in pernicious anaemia and after gastrectomy, and in ileal disease (e.g. Crohn disease). Histamine H2 receptor antagonists (cimetidine, ranitidine, omeprazole) can also impede vitamin B12 absorption.
Haematinic deficiency may be relevant in a minority. In up to 20% of patients, deficiencies of iron, folic acid (folate) or vitamin B are found and sometimes the correction of this may relieve the ulceration. Iron deficiency is usually due to chronic haemorrhage (e.g. from the gastrointestinal or genitourinary tract). Folic acid is found in green leafy vegetables especially, and body stores are small; deficiencies may be dietary, or related to malabsorption or drugs (alcohol, anticonvulsants, carbamazepine, and some cytotoxic drugs). Vitamin B12 is found especially in meat, is absorbed via intrinsic factor from the gastric parietal cells in the ileum and stored in the liver for about 3 years. Dietary B12 deficiency can arise particularly in vegans, in pernicious anaemia and after gastrectomy, and in ileal disease (e.g. Crohn disease). Histamine H2 receptor antagonists (cimetidine, ranitidine, omeprazole) can also impede vitamin B12 absorption.
 Endocrine factors are relevant in some women where RAS are related to the fall in progestogen level in the luteal phase of the menstrual cycle, or to the contraceptive pill, and then RAS may regress temporarily in pregnancy.
Endocrine factors are relevant in some women where RAS are related to the fall in progestogen level in the luteal phase of the menstrual cycle, or to the contraceptive pill, and then RAS may regress temporarily in pregnancy.
 Allergies to food occasionally underlie RAS, and there is a high incidence of atopy.
Allergies to food occasionally underlie RAS, and there is a high incidence of atopy.
 Sodium lauryl sulphate (SLS), a detergent in some toothpastes and other oral healthcare products, may produce oral ulceration.
Sodium lauryl sulphate (SLS), a detergent in some toothpastes and other oral healthcare products, may produce oral ulceration.
APHTHOUS-LIKE ULCERS (ALU) (see Table 34.3)
 Immune deficiencies such as HIV (Ch. 53), cyclical neutropenia and other immune defects.
Immune deficiencies such as HIV (Ch. 53), cyclical neutropenia and other immune defects.
 Behçet syndrome, where mouth ulcers are seen along with ulcers of the genitals (Ch. 36).
Behçet syndrome, where mouth ulcers are seen along with ulcers of the genitals (Ch. 36).
 Coeliac disease: in about 3% of patients with recurrent mouth ulcers, coeliac disease (gluten-sensitive enteropathy) – an allergic reaction to gluten in wheat – is seen.
Coeliac disease: in about 3% of patients with recurrent mouth ulcers, coeliac disease (gluten-sensitive enteropathy) – an allergic reaction to gluten in wheat – is seen.
 Crohn disease, where ulcers are seen with an enteropathy (Ch. 46).
Crohn disease, where ulcers are seen with an enteropathy (Ch. 46).
 Autoinflammatory conditions such as periodic fever, aphthous stomatitis, pharyngitis and cervical adenitis (PFAPA) syndrome, which is seen in children, appears related to a disorder of innate immunity with complement activation and IL-1β/-18, resolves spontaneously and rarely has long-term sequelae. Corticosteroids are highly effective symptomatically; tonsillectomy and cimetidine treatment have been effective in a few patients.
Autoinflammatory conditions such as periodic fever, aphthous stomatitis, pharyngitis and cervical adenitis (PFAPA) syndrome, which is seen in children, appears related to a disorder of innate immunity with complement activation and IL-1β/-18, resolves spontaneously and rarely has long-term sequelae. Corticosteroids are highly effective symptomatically; tonsillectomy and cimetidine treatment have been effective in a few patients.
 Sweet syndrome, in which mouth ulcers are found with conjunctivitis, episcleritis and inflamed tender skin papules or nodules.
Sweet syndrome, in which mouth ulcers are found with conjunctivitis, episcleritis and inflamed tender skin papules or nodules.
AETIOLOGY AND PATHOGENESIS
Immune mechanisms that appear to play a role in a people with a genetic predisposition to oral ulceration include the following (Fig. 34.2):
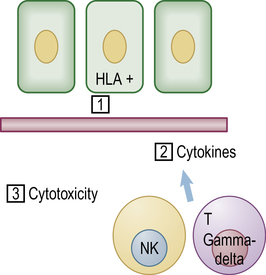
 A local cell-mediated immune response involving cytotoxic CD8+ T-cells, natural killer (NK) cells, macrophages and mast cells.
A local cell-mediated immune response involving cytotoxic CD8+ T-cells, natural killer (NK) cells, macrophages and mast cells.
 T-helper cells (gamma-delta cells), predominate in the early RAS lesions, along with some natural killer (NK) cells.
T-helper cells (gamma-delta cells), predominate in the early RAS lesions, along with some natural killer (NK) cells.
 Cytotoxic cells then appear in the lesions and there is evidence for an antibody-dependent cellular cytotoxicity (ADCC) reaction, and neutrophils and NK cells may be involved.
Cytotoxic cells then appear in the lesions and there is evidence for an antibody-dependent cellular cytotoxicity (ADCC) reaction, and neutrophils and NK cells may be involved.
 HSP can block the production of pro-inflammatory cytokines (e.g. tumour necrosis factors (TNFs) and interleukins (IL-1β, IL-6 and IL-8)) through inhibition of NF-κB and mitogen-activated protein kinase (MAPK) pathways, or activate antiinflammatory cytokines (e.g. transforming growth factor-β1), and therefore control the magnitude of the immune response.
HSP can block the production of pro-inflammatory cytokines (e.g. tumour necrosis factors (TNFs) and interleukins (IL-1β, IL-6 and IL-8)) through inhibition of NF-κB and mitogen-activated protein kinase (MAPK) pathways, or activate antiinflammatory cytokines (e.g. transforming growth factor-β1), and therefore control the magnitude of the immune response.
 IL-1β and IL-6 gene polymorphisms are associated with an increased risk for RAS. Systemic immunological abnormalities in RAS include increased plasma levels of IL-8 and IL-6. Serum IL-6 (via IL-1β over-production) typically fluctuates in auto-inflammatory syndromes, which also manifest with recurrent ulceration.
IL-1β and IL-6 gene polymorphisms are associated with an increased risk for RAS. Systemic immunological abnormalities in RAS include increased plasma levels of IL-8 and IL-6. Serum IL-6 (via IL-1β over-production) typically fluctuates in auto-inflammatory syndromes, which also manifest with recurrent ulceration.
 There is increased intra-lesional expression of TNF-α and IL-2. Systemic immunological abnormalities in RAS include increased plasma levels of TNF-α and IL-2,
There is increased intra-lesional expression of TNF-α and IL-2. Systemic immunological abnormalities in RAS include increased plasma levels of TNF-α and IL-2,
 There is an overly exuberant inflammation reaction which may be caused by derangement of toll-like receptor (TLR) gene expression.
There is an overly exuberant inflammation reaction which may be caused by derangement of toll-like receptor (TLR) gene expression.
 Regulatory T cells (TReg) CD4(+)CD25(+), crucial in regulating immune responses, are both functionally and quantitatively compromised in RAS.
Regulatory T cells (TReg) CD4(+)CD25(+), crucial in regulating immune responses, are both functionally and quantitatively compromised in RAS.
 There is decreased constitutive expression of indoleamine 2,3-dioxygenase (IDO) in the oral mucosa in RAS which may lead to the loss of local immune tolerance.
There is decreased constitutive expression of indoleamine 2,3-dioxygenase (IDO) in the oral mucosa in RAS which may lead to the loss of local immune tolerance.
 NOS2 (nitric oxide synthase) gene polymorphisms implicate a role of inducible nitric oxide synthase.
NOS2 (nitric oxide synthase) gene polymorphisms implicate a role of inducible nitric oxide synthase.
CLINICAL FEATURES
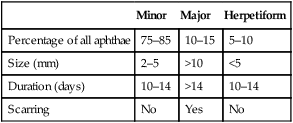
There are three main clinical types of RAS (Table 34.1), though any significance of these distinctions is unclear (they could be three distinct disorders):
Table 34.1
Clinical characteristics of the different clinical types of aphthae
| Minor | Major | Herpetiform | |
| Percentage of all aphthae | 75–85 | 10–15 | 5–10 |
| Size (mm) | 2–5 | >10 | <5 |
| Duration (days) | 10–14 | >14 | 10–14 |
| Scarring | No | Yes | No |
MINOR APHTHOUS ULCERS (MiAU; MIKULICZ ULCER)
 occurs mainly in the 10–40 year age group
occurs mainly in the 10–40 year age group
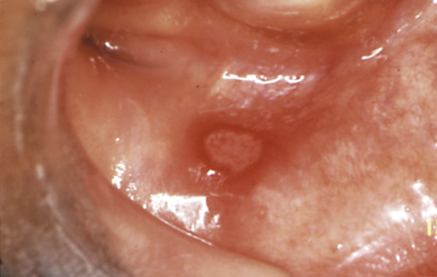
 consists of small round or ovoid ulcers 2–4 mm in diameter (Fig. 34.3), in groups of only a few ulcers (1–6) at a time, with initially yellowish floors surrounded by an erythematous halo and some oedema, but the floors assume a greyish hue as healing and epithelialization proceeds
consists of small round or ovoid ulcers 2–4 mm in diameter (Fig. 34.3), in groups of only a few ulcers (1–6) at a time, with initially yellowish floors surrounded by an erythematous halo and some oedema, but the floors assume a greyish hue as healing and epithelialization proceeds
 affects mainly the non-keratinized mobile mucosae of the lips, cheeks, floor of the mouth, sulci or ventrum of the tongue
affects mainly the non-keratinized mobile mucosae of the lips, cheeks, floor of the mouth, sulci or ventrum of the tongue
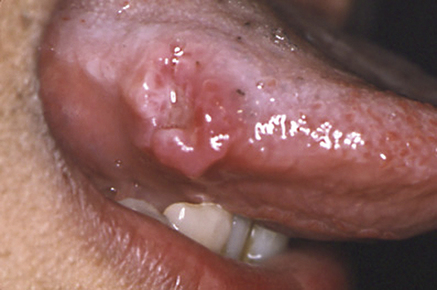
MAJOR APHTHOUS ULCERS (MjAU)
This type of RAS, also known as Sutton’s ulcers or periadenitis mucosa necrotica recurrens (PMNR):
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses