3
What Lies Beneath the Surface? Natural Teeth, Bone, and Implant Placement
As dental hygienists we sculpt root anatomy while being blindfolded. The goal is not to alter the root surface, but to uncover the pre-existing root anatomy which lies beneath.
—Catherine Fairfield, RDH
For hygienists, uncovering the underlying anatomy is critical to understanding how to access, monitor, and maintain implants. Being able to visualize the physical characteristics of natural roots and implants, as well as the differences of the types of tissue surrounding implants, will allow hygienists to effectively maintain implants. Having an understanding of bone is also key to understanding the differences an implant can make versus the choice of a bridge for patients. As well as a fundamental knowledge of the biomechanics and component parts of an implant and the many varied restorative options.
Natural teeth versus implants
The physical differences between natural teeth and implants are often compared to the roots of teeth. Replacing the root of a tooth helps to maintain the bone, but there are fundamental differences. It starts with the surface of a natural tooth (i.e., cementum) and the implant surface of titanium alloy or zirconia, rough, porous, or smooth, sometimes with coated surfaces to create a more biocompatible surface for the bone to osseointegrate with. Figure 3.1 shows how an implant attaches to bone.
Figure 3.1 How an implant attaches to bone. Courtesy of Keystone Dental.
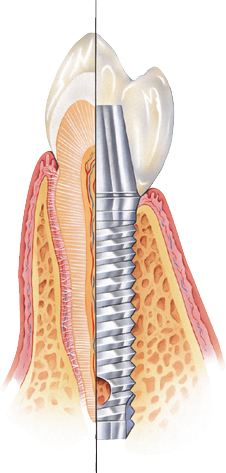
The key physical difference between a natural root and an implant is that the natural root is sensitive to heat and a pulpitis is possible, whereas an implant has no sense of temperature. Also, implants are not susceptible to decay and natural teeth are, which is one reason implants are very good restorative choice for patients with controlled diabetes, xerostomia, or autoimmune disease. Xerostomia patients who suffer from decreased saliva go from an increase in decay to broken teeth and eventually dentures without much success. If an implant is placed when the first tooth is lost, this cycle can be broken and the quality of life for these patients greatly improved. Autoimmune diseases (e.g., AIDS, asthma, arthritis, or lupus) where the patient’s immune system is not functioning properly can be helped by implant therapy which does not rely on the host response to stay healthy.
The mobility of a natural tooth can cause a loss of attachment, periodontal disease, or trauma that can be reversed. The natural tooth can also test positive or negative for mobility due to periodontal disease or occlusion. Implant mobility is caused by occlusion, trauma, or infection, but with a much more negative result, often the loss of osseointegration which means the loss of the implant. Since an implant is held in by the bone with no periodontal ligament, such as a cement post in the ground, if it becomes mobile there is a good chance the implant will fail. The good news is that in most cases it can be replaced with a new implant.
The attachment of the tissue that surrounds the natural tooth and implants is where the bigger differences lie. The attachment of the gingival tissues to the neck of the implant is distinct from the attachment to natural teeth. Both the natural tooth and the implant have junctional epithelium (hemidesmosomes and basal lamina) and sulcular epithelium but implants have no evidence of Sharpey’s fibers between an implant or implant abutment and bone.
The junctional epithelium of a natural tooth attaches to the tooth coronal to the bone up to 2 mm and has a sulcular epithelium of 2–7 mm with a definite connective tissue attachment. The implant has only an adhesion attachment of connective tissue with a junctional epithelium up to 1.5 mm. It runs parallel and circular to the fixture with a sulcular epithelium of 0.5–1.0 mm, but these do not insert into the implant surface, making this attachment much more fragile and susceptible to damage by trauma and/or infection. This tissue–implant interface is known as the perimucosal seal. The perimucosal seal is the tissue barrier that prevents microorganisms and other inflammatory agents from the oral cavity from entering the tissues that surround the implant. It contains the sulcular epithelium, and its presence is important for the longevity and success of the implants (see Table 3.1).
Table 3.1 Comparison between natural dentition/tissue and dental implants.
| Structure | Natural Dentition | Implant |
|---|---|---|
| Attachment | Cementum, periodontal ligament, and bone | Bone (osseointegration) |
| Tissue: junctional epithelium, sulcular epithelium and connective tissue (CT) | Junctional epithelium: Attaches to the tooth coronal to the bone up to 2 mm Sulcular epithelium: 0.2 to 0.7 mm CT: Has attachment |
Junctional epithelium: Run parallel and circular to the fixture up to 1.5 mm, do not attach Sulcular epithelium: 0.5 to 1.0 mm CT: Adhesion, no attachment |
| Vascularity and bleeding on probing (BOP) | Vascularity: Greater BOP: Reliable Supraperiosteal and periodontal ligament |
Vascularity: Less BOP: Less reliable Periosteal only |
| Temperature | Sensitive to heat, pulpitis possible | No heat sensitivity |
| Decay | Decay is possible | Do not decay |
| Infection | Yes, gingivitis and periodontitis | Yes, mucositis and peri-implantitis |
| Mobility | Yes, caused by loss of attachment, periodontal disease, or trauma. Reversible | Yes, caused by peri-implant disease, occlusion, or trauma. Not reversible |
Bone: it’s all about the bone!
In implant dentistry the most important factor is bone: quality, quantity, and density influence successful outcomes in implant dentistry. The volume density of bone matrix in cortical (outer layer) bone is approximately 80–90% and 20–25% in cancellous (inner layer) bone (1, 2). Bone is composed of cortical and cancellous bone, and intertwined between these two parts is a lattice network of trabecular that is the reservoir for active bone metabolism (see Figure 3.2). The bone structure is continuously repairing and remodeling to keep its form and function.
Figure 3.2 Cortical and cancellous bone. Courtesy of Keystone Dental.
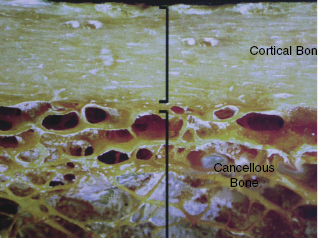
An understanding of osteogenesis, osteoconduction, and osteoinduction is important to grasp the full picture of how bone remodels and the principles behind regeneration and osseointegration of implants (see Table 3.2). Osteogenesis is bone regeneration based on living bone cells that transfer into an area to make new bone. Osteoconduction is the scaffold that guides the reparative growth of this process and provides the space maintenance for the bone to regenerate effectively without the interference of tissue (3). Osteoinduction is a process to signal and stimulate the bone cells to assist and accelerate the body’s own bones to regenerate.
Table 3.2 The principles behind regeneration.
| Product Type | Definition | Example |
|---|---|---|
| Osteogenesis “cells” | Bone regeneration is based on living bone cells in the graft material to contribute to bone remodeling | Autografts, allografts, xenografts, and alloplasts |
| Osteoconduction “scaffold” | A scaffold that guides the reparative growth of the natural bone regeneration. An artificial membrane used near the bone edge to ensure the bone regenerates in the defect without interference of tissue | Autografts, allografts, xenografts, and alloplasts |
| Osteoinduction “signals” | A signaling growth factor (biologic) that stimulates the division and differentiation of particular types of cells for true regeneration | Platelet-rich plasma (PRP) Platelet-rich fibrin (PRF) Platelet-rich growth factor (PRGF) |
These three factors are a guideline to understanding how the regeneration principles and products for regeneration were established. Bone regeneration procedures that are used today are socket preservation, implant defects (dehiscence or fenestration), and sinus and ridge bone augmentation.
Dental hygienists need to have a clear understanding of bone quality and density to be able to explain this to patient in terms of how much time will it take for the patient to complete his or her implant treatment and to help the patient understand the expense associated with possible added procedures to have the necessary bone for successful treatment results. Research clearly states that the strength of the bone is directly related to the density of the bone (4, 5). Also, the quality and density is directly related to the type of implant the dental professional will chose to place, the healing time needed for the patient, and success rate for the implant. Actual healing times may vary based on the patient’s ability to remodel bone and his or her overall health.
For learning purposes and for a visual image to present bone types of the oral cavity to a patient (see Table 3.3), refer to Reference 6, which identifies four distinct tissue types: woven, lamellar, bundle, and composite. Woven bone is rapidly replaced by mature, stress-bearing bone. Lamellar bone is the main component of mature cortical and trabecular bone. Bundle bone generally is found adjacent to the periodontal ligament with characteristics of ligaments and tendon attachments. Composite bone is a variation of fine cancellous compaction (osteons) or coarse cancellous compaction (whorling bone) (6). The literature points out that there are different surgical protocols for different bone types that affect healing and treatment planning (5–10). There are exceptions to the rule in location and type of bone for patients, but for an initial conversation with the patient, this classification is ideal.
Table 3.3 Bone classification (11, 12).
Source: Figures courtesy of Keystone Dental.
| Bone Type | Example |
|---|---|
| Type One Very compact, dense cortical bone 3–4 months of healing time Compares to oak/hard maple |
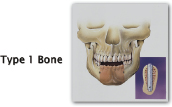 Anterior mandible |
| Type Two Porous, compact cortical bone 4–6 months of healing Compares to spruce/white pine |
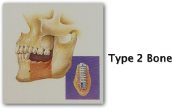 Posterior mandible |
| Type Three Coarse, trabecular-less cortical bone Usually a 6-month healing time Compares to balsa wood |
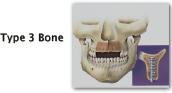 Anterior maxilla |
| Type Four Fine, trabecular-minimal cortical bone 6–8-months of healing time Compares to Styrofoam |
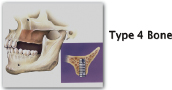 Posterior maxilla |
Four classifications of bone
The hygienist needs a fundamental understanding of each type of bone and to be able to relate to the patient a tactile sense of the density of the bone in relation to where the implant will be placed (11, 12). Each bone type can be compared to a type of wood to help the patient understand, visualize, and be able to relate to the surgeon’s recommendation for healing time.
The bone is classified according to structure, composition, density, and volume with four types of bone referred to as types 1–4 or D1–D4 (the reference is the same; only the terminology is different). To further define this, the types are as follows:
Frequently asked questions
Why is bone density or type so important?
The type of bone is critical in implant therapy. It is directly related to implant placement, implant selection, and the length of healing time for osseointegration. A hygienist must know this for treatment planning and to be able to explain to the patient the different “types” or “densities” of bone in relation to healing time and restoration selection options.
What is the tooth relationship to type of bone?
Basel bone forms with or without teeth or implants. Alveolar bone forms because of teeth and residual bone is alveolar bone that has been resorbed.
What happens if teeth are lost and not replaced?
If teeth are lost and not replaced, atrophy and bone loss becomes apparent to facial esthetics. This is commonly recognized as premature aging, increased wrinkles, jowl development, and loss of function.
Bone loss
Emphasize these points with patients with patients when having the conversation on why bone is important. The width of bone decreases 25% in the first year after a tooth is lost or extracted and the bone height decreases 4 mm in the first year (13,14). In the first 2–3 years after an extraction, 40–60% of the ridge width can be lost (15) and the overall bone can continue to be resorbed 0.5–1% yearly for the patient’s life (16).
When there are missing teeth involved, bone and tissue degrade over time, losing their function. If a tooth is extracted and left to heal on its own, it will repair, but with tissue regenerating faster than bone causing a sunken-in effect (see Figure 3.3). Resorption and remodeling of the alveolar bone occurs after teeth are lost due to periodontal disease, trauma, or tooth extraction (17,18). If regenerative procedures such as socket preservation are done at the time of extraction, the bone will regenerate and keep its structure.
Figure 3.3 Ridge width lost with traditional extraction, no socket preservation. Courtesy of Dr. Kevin/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


