3
Pharmacology of local anaesthetics
Local anaesthetics are the most commonly used pharmaceuticals in dentistry. They interfere reversibly with the generation of the action potential and with cellular impulse conduction by blockading the sodium channels in the nerve cell. This results in a local insensibility to pain stimuli.
3.1 Classification
Local anaesthetics share several common characteristics in their molecular structure (Figure 3.1). A lipophilic group can be identified which determines lipid solubility. Another part contains a hydrophilic group that determines the degree of water solubility. Usually the lipophilic part of the molecule is an aromatic structure that contains a benzene ring. The hydrophilic part contains a secondary or a tertiary amine. Both parts, present at the opposite ends of the molecule, are connected by an intermediate section. This intermediate section consists of an ester or an amide group and a relatively short chain of four to five carbon atoms (Box 3.1).
Figure 3.1 Molecular structure of an ester-linked (procaine) and amide-linked local anaesthetic (lidocaine). (Modified from Borchard (1985) Aktuelle Aspekte der Zahnärzlichen Lokalanasthesie. Hoechst AG.)
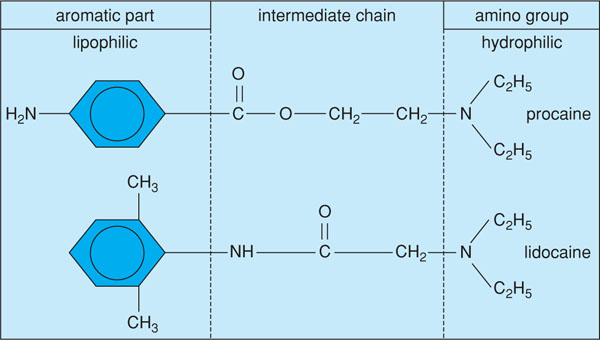
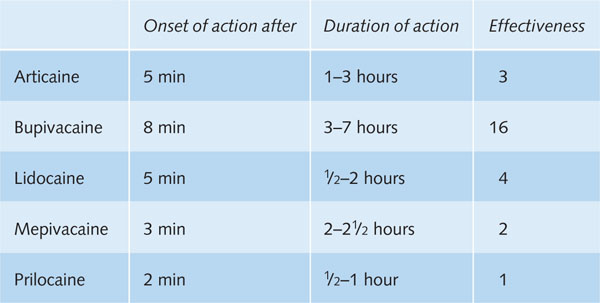
Table B3.1 An overview of regularly used local anaesthetics with average values of onset time, duration of action and effectiveness relative to prilocaine.
In other words, local anaesthetics can be classified according to their molecular structure into two classes: the (amino-) esters and the (amino-) amides. Both groups differ in the way in which they are metabolised.
In ester anaesthetics, the aromatic part containing the benzene ring is derived from para amino benzoic acid (PABA). The intermediate chain, characteristic for this group, contains an ester-binding (see Figure 3.1). A well-known example of an ester-anaesthetic is procaine.
Amide-anaesthetics have been developed more recently and are characterised by the presence of an amide-binding in the intermediate chain, as in lidocaine (see Figure 3.1). The amides can further be divided into three subgroups: xylidines, toluidines and thiophenes. Xylidines are tertiary amines with an aromatic part that contains two methyl groups. Representatives from this group are lidocaine (Xylocaine®), mepivacaine (Scandicaine®) and bupivacaine (Marcaine®) (Box 3.2).
In toluidines, the benzene ring contains a single methyl group and the amine part contains a secondary amine. A frequently used local anaesthetic from this group is prilocaine (Citanest®).
Thiophenes, such as articaine (Ultracain®; Septanest®), have a slightly different molecular structure. They contain a sulphur ring in the aromatic part of the molecule. This is probably the reason why this local anaesthetic has a better penetration into the mucosa and the jaw bone as well.
3.2 Pharmacodynamics
Local anaesthetics differ considerably in onset time, duration of action and strength of the analgesic effect. The strength of the local anaesthetic effect is directly related to the lipid solubility of the local anaesthetic.
Of the series bupivacaine–articaine–lidocaine–prilocaine–mepivacaine, the first anaesthetic is the most lipid soluble and the last one the least. The same order applies to the strength of the analgesic effect of these compounds (Box 3.3). The differences in analgesic strength of the compounds, however, seem smaller than theoretically expected (see table in Box 3.1). Probably, pharmacokinetic aspects are also important for the strength of the analgesic effect. An overview of factors that can affect the intrinsic activity of local anaesthetics is presented in Table 3.1.
Table 3.1 Some factors that affect the intrinsic properties of local anaesthetics.
| Factor | Mechanism |
| Pregnancy | Progesterone can potentiate the nerve blocking effect of the local anaesthetic. |
| pH alteration | Inflammation and uraemia lower the tissue pH. This reduces the percentage of the neutral base form. A pH alteration can also affect the binding to plasma and tissue proteins, and seems relevant for the rapid appearance of tolerance during a repeat injection. |
| Vasodilatation | Intrinsic vasodilatation causes rapid elimination from the area of injection. For example, bupivacaine is a vasodilator. |
| Vasoconstriction | A vasoconstrictor masks the inherent vasodilatory properties of the local anaesthetic and causes an increased effect that also lasts longer. |
3.3 Pharmacokinetics
3.3.1 Physical–chemical characteristics
Local anaesthetics are weak bases, unstable and poorly water soluble. With a single exception, they are tertiary amines. Therefore hydrochloric acid (HCl) is added to the local anaesthetic, which converts the tertiary amine, expressed below as R3N, into a chloride salt:
R3N + HCl → R3NH+ + Cl−
This increases the stability and water solubility of a local anaesthetic:
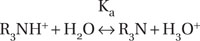
In which R3NH+ represents the water-soluble, ionised, quaternary cation form of the local anaesthetic, which is responsible for the analgesic effect. This ‘active’ form is in equilibrium with the ‘inactive’ form, the uncharged tertiary base R3N (Boxes 3.4 and 3.5). The equilibrium constant Ka of most local anaesthetics is between 7 and 8.
3.3.2 Diffusion
From the site of administration, a local anaesthetic must cross several barriers to reach the actual site of action. For this, the uncharged base form of the local anaesthetic is necessary (R3N). This form is lipophilic (fat soluble), enabling it to pass through the cell membrane of neurons. The fat solubility of a local anaesthetic is primarily determined by the charge of the molecule and – to a lesser extent – by the length of the molecule skeleton: the longer the chain, the greater the fat solubility.
Once the uncharged lipophilic form is inside the cell, a new equilibrium with the water-soluble charged form will be established. The intracellular pH is lower than outside, so the equilibrium will shift towards R3NH+, the active cationic form of the anaesthetic. Consequently, the proportion between both forms (R3N and R3NH+) plays an />
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


