Case 2
Implant Procedures and Maintenance
Medical History
Patient has hypertension and hypercholesterolemia. Current medications include Lipitor, Losartan, and 81 mg Aspirin. Patient states allergy to clindamycin. On maintenance visit blood pressure was 132/80 mmHg, pulse 74 beats/min.
Social History
Patient drinks socially and does not report the use of recreational drugs or tobacco.
Dental History
The patient’s maxillary arch is restored with seven implants and has a history or periodontal disease.
Extra‐Oral Examination
No significant findings were found. No abnormalities were identified and the TMJ was within normal limits.
Intraoral Examination
Generalized gingival inflammation was found due to plaque accumulation. The gingival tissue around implant #13 was erythematous and edematous. Upon palpation, suppuration was present. The platform margin of implant #12 was visible (Figure 6.2.1A).
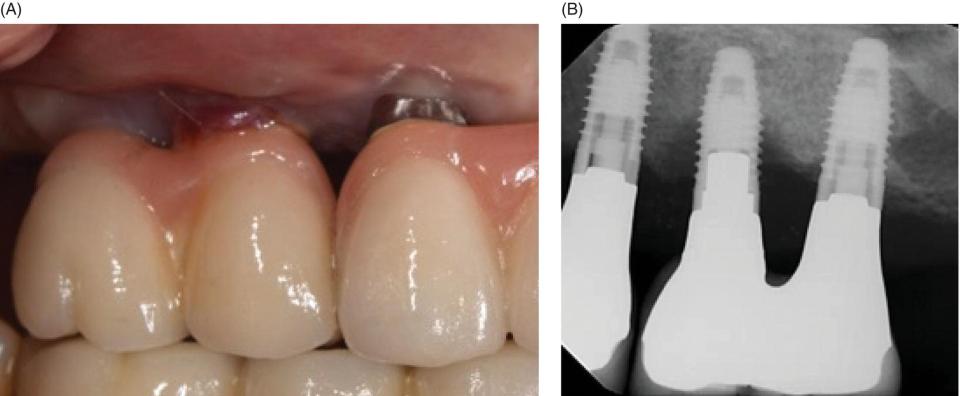
Figure 6.2.1: Clinical (A) and radiographic (B) images of implant #13 with peri‐implantitis. Note the erythema and edema in the peri‐implant mucosa (A) and the crater formed destruction around the implant (B).
Radiographic Examination
A radiograph revealed a saucer shaped defect with bone loss up to and around the fourth thread of implant #13 (Figure 6.2.1B).
Diagnosis
After reviewing the history and both the clinical and radiographic examinations, a clinical diagnosis of peri‐implantitis was rendered (Figures 6.2.2 and 6.2.3).

Figure 6.2.2: After crown removal and reflection of the gingival flap, the extent of the bone loss is observed.

Figure 6.2.3: Bone graft placed to restore defect.
Peri‐implant diseases are inflammatory reactions in the tissues surrounding an implant. Peri‐implant mucositis is confined to the mucosa without signs of loss of supporting bone. Peri‐implantitis includes inflammation of the mucosa and loss of supporting bone (Zitzmann and Berglundh 2008). If left untreated, peri‐implantitis may progress and lead to implant loss. It has been reported that peri‐implantitis occurs between 28–56% of patients treated with implants, and peri‐implant mucositis in 80% of patients (Lindhe and Meyle 2008).
Treatment Plan
The patient first completed an initial debridement of the area using hand instrumentation with titanium curettes and irrigation with chlorhexidine. This initial therapy was completed to reduce the amount of bacteria in the area and to improve the tissue consistency prior to surgical therapy. Six weeks after this initial treatment, the inflammation of the gingival tissue had reduced, but erythema was still present. Surgical therapy was initiated to restore the bony architecture that had been lost. The implant crowns were removed to gain access to the area of the defect. Once all the granulation tissue was removed, a detoxification of the implant was completed by chemical treatment of 35% phosphoric acid etching gel (Strooker et al. 1998) and implantoplasty using a fine diamond bur (Schwarz et al. 2011). A bone graft was placed to resolve the defect with an acellular dermal matrix to increase the soft tissue.
Discussion
There are many factors that can lead to peri‐implant disease including (El Chaar 2009):
- history of periodontitis
- tobacco smoking
- bone quality
- excess cement
- lack of compliance and limited oral hygiene
- systemic diseases
- soft tissue defects or lack of keratinized gingiva
- implant malpositioning
- overcontouring the restoration.
The development of peri‐implant disease appears to be accompanied by an increase in the bacterial species that have similarly been found to increase in periodontitis such as Porphyromonas gingivalis, Tannerella forsythia, and Aggregatibacter actinomycetemcomitans (Leonhardt et al. 1999).
The diagnostic parameters for peri‐implant disease are probing, bleeding on probing, suppuration, implant mobility, and radiographic bone loss (Salvi and Lang 2004). Probing around an implant is used for bone sounding, as the epithelial and connective tissue around an implant differs from that of a natural tooth. Due the fibrotic nature of the gingival tissue around an implant, the connective tissue attachment is loosely arranged and not as tightly bound when compared to a natural tooth. Therefore, probing depth measurements at implants and teeth yield different information (Shou et al. 2002).
Treatment for peri‐implant mucositis would be a nonsurgical intervention.
Nonsurgical treatment includes:
- Hand instrumentation using titanium or plastic curettes
- Prophy jet
- Laser
- Local antiseptic medication
- Oral hygiene instructions.
Treatment for peri‐implantitis would be a surgical intervention.
Surgical therapy includes:
- Open flap debridement
- Regenerative procedures – such as bone grafting and guided tissue regeneration
- Resective procedures – implantoplasty
- Implant removal.
Assessment
At each maintenance visit, record:
- any sign of inflammation or changes in color, contour, or consistency of the gingiva
- if the gingiva around the implant is keratinized or nonkeratinized
- bleeding on probing
- probing depth
- mobility
- if suppuration is present
- review radiographs if any changes in bone level or crater like defect present (Wingrove 2013)
- if patient reports any discomfort around the implant.
For radiographs, if one to four implants: take vertical bitewing or periapical (PA) of each implant. Five or more implants: panoramic x‐ray or individual PAs of all implants. The panoramic radiograph will not be able to assess peri‐implant bone loss as well as individual PAs, but can be used to examine multiple implants at once. Radiographs should be present for time of implant placement, restoration, and one year for comparison. Radiographs should be taken yearly for implant sites (Wingrove 2013). Report all findings to the dentist.
Instrumentation
The key to proper instrumentation is the removal of plaque and any other deposits without scratching the surface of the implant or prosthesis. Instrument tip designs should provide the clinician with adequate options to address the patient’s individual implant abutment and prosthetic components’ needs. The instrument should offer some of the same basic features as a metal scaler or curette, while being made for an implant‐friendly/compatible material. Some qualities to assess are: familiar design, ergonomic handles, rigidity to withstand scaling pressure without the risk of fracture or breakage, the ability of the instrument not to leave any residue behind, blade dimensions that allow easy and atraumatic access to implant abutment angulations and tissue tightness, and lightweight to avoid hand and wrist fatigue (http://www.rdhmag.com/articles/print/volume‐32/volume‐12/features/more‐than‐maintenance.html).
Implant Scalers Selection
Safe instrumentation on titanium implant surfaces should be performed with an implant scaler that is biocompatible, “titanium on titanium,” to prevent instrument debris from becoming lodged on the surface, as it can harbor bacteria and increase the risk for peri‐implantitis. They are thinner than plastic or graphite implant scalers, yet provide more strength to dislodge calculus and residue cement, which makes them more effective (Karring et al. 2005). Some implant instruments have been shown to achieve optimal implant maintenance results while preserving abutment and prosthesis surfaces without altering or scratching (Rentsch‐Kollar et al. 2010). Polishing with a rubber cup and nonabrasive toothpaste, fine prophy paste, and tin oxide have all been shown not to alter titanium implant surfaces (http://www.rdhmag.com/articles/print/volume‐32/volume‐12/features/more‐than‐maintenance.html).
Take‐Home Hints
- The formation of plaque biofilm is considered the primary etiological factor in peri‐implant disease initiation, and it is essential for the development of typical peri‐implant infections (Mombelli and Lang 1994; Salvi et al. 2012).
- Peri‐implant mucositis is confined to the soft tissue with no signs of bone loss and can be treated nonsurgically. Peri‐implantitis affects the soft tissue and displays bone loss. Peri‐implantitis can be treated by nonsurgical and/or surgical therapy.
- Early diagnosis of peri‐implant diseases is important to ensure that the pathological processes can be arrested before the stability of the implant is compromised. After completion of dental implant treatment, the patient must be put on a tightly monitored maintenance schedule. Dental hygienists who perform these maintenance treatments play important roles in detecting the initial signs of peri‐implant diseases.
- Implant maintenance is a corner stone in the prevention of peri‐implant diseases. Maintaining an implant patient is very similar to maintaining a patient with treated periodontitis. The maintenance recall interval is based on the patient’s past history, susceptibility to periodontitis, and ability to control local factors at home. A maintenance visit should consist of a thorough visual examination of peri‐implant soft tissues, tissue response upon external pressure by a periodontal probe, presence of suppuration, and periodical radiographs (El Chaar 2016). At home recommendations for patients with fixed implant‐borne restorations include patient education regarding brushing and use of oral hygiene aids (Kumarswamy 2015).
- Medical grade titanium scalers are recommended because they are biocompatible and thinner than plastic implant scalers. They also provide the needed strength to dislodge calculus and even residue cement.
- Professional preventive measures need to be personalized to the individual patient, based upon clinical findings and lifestyle factors. This requires diagnosis to stratify subjects into:
- i. periodontally healthy, (ii) gingivitis (peri‐implant mucositis), and (iii) periodontitis (peri‐implantitis) as well as risk assessment for future status. Validated periodontal screening methods are now a fundamental requirement for all patients, given the high prevalence of periodontal and peri‐implant conditions and should be applied universally (Tonetti et al. 2015).

VIDEdental - Online dental courses


