Impression materials
Introduction
Impression materials are used to produce a detailed replica of the teeth and the tissues of the oral cavity. From this replica, or impression, a model can be made that is used in the construction of full dentures, partial dentures, crowns, bridges and inlays.
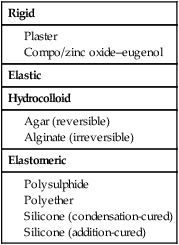
Over the years, a wide variety of impression materials and associated techniques have been developed, all striving to achieve the optimum in desirable characteristics. The impression materials can be classified in terms of rigid and elastic impression materials (Table 2.7.1).
The rigid impression materials cannot engage undercuts that may be present on the teeth or the bone. Consequently, their use is restricted to edentulous patients without bony undercuts.
The elastic impression materials are subdivided into hydrocolloid and elastomeric impression materials. Both are able to engage undercuts and may be used in edentulous, partially dentate and fully dentate patients. The choice will depend on the particular requirements of each individual case.
The choice of impression material may also be affected by the technique to be adopted, with a major consideration being the selection of a stock tray or special tray. These trays are needed to support the impression material (especially when it is still fluid), so that it can be carried to the patient, inserted in the mouth, and removed once it is set. The trays also provide support when the model is poured from the impression.
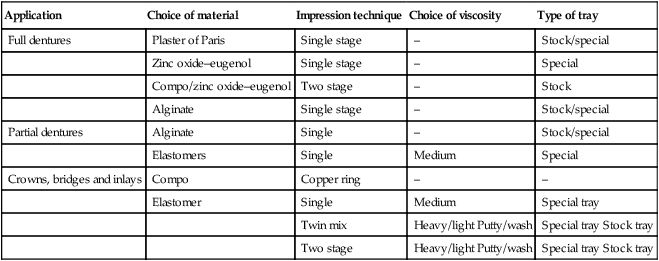
The variety of applications of and techniques used with the impression materials are presented in Table 2.7.2. The choice of impression tray is determined, to some extent, by the viscosity of the impression material.
Table 2.7.2
Indirect impression techniques
| Application | Choice of material | Impression technique | Choice of viscosity | Type of tray |
| Full dentures | Plaster of Paris | Single stage | – | Stock/special |
| Zinc oxide–eugenol | Single stage | – | Special | |
| Compo/zinc oxide–eugenol | Two stage | – | Stock | |
| Alginate | Single stage | – | Stock/special | |
| Partial dentures | Alginate | Single | – | Stock/special |
| Elastomers | Single | Medium | Special | |
| Crowns, bridges and inlays | Compo | Copper ring | – | – |
| Elastomer | Single | Medium | Special tray | |
| Twin mix | Heavy/light Putty/wash | Special tray Stock tray | ||
| Two stage | Heavy/light Putty/wash | Special tray Stock tray |
An impression material that is very fluid when it is first mixed cannot be used with a stock tray, and a close-fitting special tray needs to be produced. This can be done either by constructing an acrylic special tray from a preliminary model, or by using a high-viscosity material, which is placed in a stock tray; once this has set, a special tray is produced. Some impression materials are not available in a sufficiently high-viscosity version for use in a stock tray, and these include zinc oxide–eugenol, polyether and polysulphide elastomers. Others, such as impression compound (compo), plaster of Paris, alginate and the silicones are available in formulations that can be used with a stock tray. Although compo can be used in a stock tray, the impression obtained does not reproduce surface details adequately unless a zinc oxide–eugenol wash is used with it. Similarly, alginates, when used in a stock tray, do not always give the required degree of accuracy and are then better used in a special tray.
Requirements of an impression material
Some of the requirements of an impression material have already been touched on in the above discussion and now need to be defined more explicitly. The important characteristics of impression materials can be identified from the point of view of the patient or the dentist (Table 2.7.3).
Table 2.7.3
Requirements of an impression material
| The patient | The dentist |
| Neutral taste and odour | Easily mixed |
| Short setting time | Short working time |
| Small tray | Easily removed |
| Easily removed | Good-quality impression |
| Non-toxic | Low-cost materials |
| Easily disinfected |
Accurate reproduction of surface detail
The accuracy of reproduction of the surface detail depends on the viscosity of the mix and the ability of the impression material to adapt closely to both the soft and the hard tissues. A low viscosity is therefore desirable, but it should not be so low that the material is not easily contained within the impression tray.
Some materials are hydrophobic (water-repellent) and will be repelled by moisture on the surface. If this should happen in a critical area, then important surface detail may be lost as a blow hole is formed on the impression surface due to trapped air. A dry field is essential for such materials. If this is not feasible, an alternative material must be used that is compatible with moisture and saliva.
There are instances in which the patient will have mobile soft tissues. This occurs particularly with edentulous patients, who can present with a flabby ridge. If the impression material is very stiff, it may displace such tissues and produce a distorted impression. When this is reproduced in the prosthesis, the soft tissues will need to adapt to the prosthesis rather than the other way round; this will cause discomfort to the patient. Such impression materials are classed as being muco-compressive.
Ideally, the impression material to be used should be sufficiently fluid on placement to prevent displacement of the soft tissues, with such materials being considered to be mucostatic.
Dimensional accuracy and stability
These are dependent on the factors outlined below.
Type of tray
If the tray is prone to distortion, then the resultant model poured from such a tray will also be distorted. Hence, highly flexible trays should be avoided. A good bond between the tray and the impression material is very important. If the impression material comes away from the tray, this will again distort the impression. Manufacturers of impression materials will supply a suitable adhesive for their material to ensure a good bond. It is important that the manufacturer’s instructions are followed to the letter; otherwise failure of the adhesive bond may result. Additional retention may be achieved by the use of perforated trays.
Shrinkage of the impression material
Whether the impression material sets by a chemical reaction or some change in physical state, both usually result in some shrinkage of the impression material. Provided the impression material is firmly adhered to the tray, this increases the space previously occupied by the hard or soft tissues. In the case of a simple crown preparation, the result is a die which is slightly larger than the original tooth preparation (Figure 2.7.1). If the contraction of the impression material is excessive, this will result in a loosely fitting crown. Should the impression material expand on setting, then the opposite problem of a tightly fitting crown would result, with too little space for the cement that is needed to hold it in place.
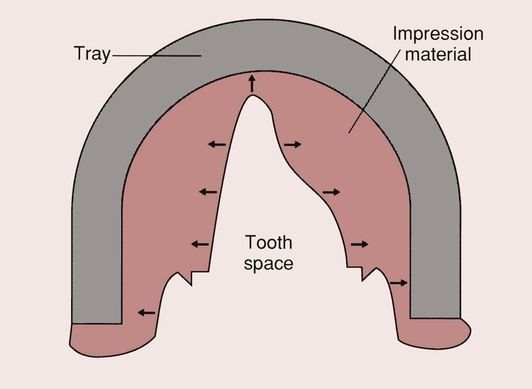
In addition to the changes in dimensions on setting, there is also a slight thermal contraction of the impression material as it cools from mouth to room temperature. The coefficient of thermal expansion of both the tray and the impression material needs to be small. Ideally, an impression material should show a very small contraction (<0.5%), as this will result in the production of a crown that is slightly larger than the situation that it is designed for. This will provide the necessary space for the cement that is to be used.
Permanent set
When an impression is taken of a dentate patient, there will be undercuts due to the bulbous shapes of the tooth crowns. In this case, the impression material must be sufficiently flexible to allow removal from the undercut regions without causing distortion; rigid impression materials would therefore be unsuitable. The elastic impression materials must then be used, but, as most are actually viscoelastic materials (see Chapter 1.6), there is a possibility of some permanent deformation.
Impression technique
In the case of silicone impression materials in particular, there are a number of impression techniques that can be employed. It is important that the appropriate technique is used for each material. This will be dealt with later, in the discussion of the silicone impression materials.
Rigid impression materials
Impression compound (compo)
Impression compound is a thermoplastic material with a glass transition temperature of about 55–60°C. Above its glass transition temperature it becomes soft and will take up a new form. On cooling to mouth temperature, it hardens and can be removed, retaining an impression of the oral cavity. Thus, no chemical reaction is involved in the use of this material.
Composition
The composition of impression compounds tends to vary from product to product and is usually a trade secret. They consist of a combination of resins and waxes, plasticizers and fillers, each having a specific function:
Properties
Impression compound is muco-compressive, as it is the most viscous of the impression materials used. This can present particular problems in those patients who have a flabby mandibular ridge.
Compo is rigid once cooled and therefore cannot be used to record undercuts. It has a high viscosity, so reproduction of surface detail is not very good. However, the reproduction can be improved by reheating the surface of the impression material after taking the first impression and then reseating it in the patient’s mouth. Even then, the surface detail is not as good as can be achieved with virtually all of the other impression materials. It is therefore better to use compo as a simple and quick means of producing a special tray, and then use a wash of zinc oxide–eugenol to provide the surface detail.
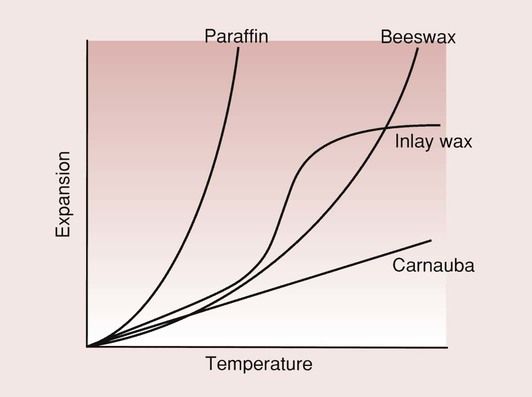
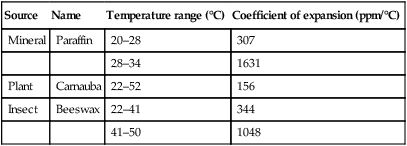
The coefficient of thermal expansion of resins and waxes is very high, as indicated in Table 2.7.4, and are highly non-linear within the temperature range of dental interest (Figure 2.7.2). Shrinkage is in the order of 1.5%, and is due to the thermal contraction from mouth to room temperature.
Table 2.7.4
Coefficient of expansion of waxes
| Source | Name | Temperature range (°C) | Coefficient of expansion (ppm/°C) |
| Mineral | Paraffin | 20–28 | 307 |
| 28–34 | 1631 | ||
| Plant | Carnauba | 22–52 | 156 |
| Insect | Beeswax | 22–41 | 344 |
| 41–50 | 1048 |
The material has poor dimensional stability and the model must be poured as soon as possible after the impression is taken; this should take place within 1 hour.
The thermal conductivity of impression compound is very low, meaning that, on softening, the outside will always soften first. This can give the impression that the material is ready for use when the inside might still be quite hard. Differential expansion gives rise to internal strains, which relieve themselves in due course by distortion of the impression. Thus, the material must be placed in the water bath to allow sufficient time for it to achieve a uniform temperature. Even then, internal strains will inevitably build up during cooling, and will eventually give rise to distortion, which is why the model must be poured as soon as possible.
Application
Its main application is for recording preliminary impressions of edentulous arches. This gives a model on which a special tray can be constructed, and which can subsequently be used with a low-viscosity impression material (such as zinc oxide–eugenol) for recording the fine surface detail (see below). The material is used relatively little these days, as other impression materials are preferred.
Zinc oxide–eugenol paste
Whereas there are many zinc oxide–eugenol products that are presented as powder–liquid systems, the impression material is in the form of two pastes. There is typically a base paste consisting of zinc oxide, olive oil, linseed oil, zinc acetate and a trace of water, and a reactor paste, consisting of eugenol and fillers, such as kaolin and talc.
Zinc oxide and eugenol are the reactive components that take part in the setting reaction (see Chapter 2.4). The water initiates the setting reaction, and the zinc acetate is present to speed up the setting process. The oils and fillers are inert substances, which allow the material to be used in a paste–paste formulation, and help to provide the appropriate handling characteristics.
Properties
The liquid is very fluid, i.e. mucostatic, and, being a water-based system, readily adapts to the soft tissues. It therefore provides a detailed reproduction of the soft tissues without causing displacement of the soft tissues, but is rigid once set and is thus unable to record undercuts. This limits its application to the edentulous mouth, where it is used with a special tray.
It has the advantage of being dimensionally stable and shows little shrinkage on setting. However, as it is used with a special tray, the tray may impose limitations on the dimensional stability of the whole impression.
Although the material is non-toxic, eugenol can cause a burning sensation in the patient’s mouth and leave a persistent taste that the patient may find unpleasant. The paste tends to adhere to skin, so the skin around the lips should be protected with petroleum jelly.
Impression plaster
Presentation and composition
Impression plaster consists of a powder to which water is added to produce a smooth paste. The composition of the powder is similar to that of the model materials discussed in more detail in Chapter 3.1, as is the setting process. The impression material consists typically of calcium sulphate β-hemihydrate (CaSO4)2·H2O, potassium sulphate to reduce the expansion, borax to reduce the rate of setting, and starch to help disintegration of the impression on separation from the plaster/stone model.
Properties
The impression plaster is easy to mix, but great care must be taken to avoid trapping air bubbles, as these will give rise to surface inaccuracies. The material has well-controlled working and setting characteristics, which are governed by the relative amounts of borax and potassium sulphate.
The amount of potassium sulphate is generally more than would be found in model plaster, as for impressions the expansion must be kept to a minimum. Since the potassium sulphate also acts as an accelerator of the set, borax is needed to counteract it. The working time is of the order of 2–3 minutes, as is the setting time.
The mixed material has a very low viscosity, and so is mucostatic. It is hydrophilic and thus adapts readily to the soft tissues, recording their surface detail with great accuracy. The material is best used in a special tray, made of acrylic or shellac, to a thickness of 1.0–1.5 mm. Alternatively, it can be used as a wash with a compo special tray.
The dimensional stability of impression plaster is very good, so a time delay in pouring the model is of no consequence, although extremes of temperature should be avoided. A separating medium (usually a solution of sodium alginate) must be used between the model plaster and the impression plaster.
The material is rigid once set and thus unable to record undercuts. This limits its application to the edentulous patient.
From the patient’s point of view it is not an unpleasant material, although it tends to leave a sensation of dryness in the mouth for some time after the impression has been taken.
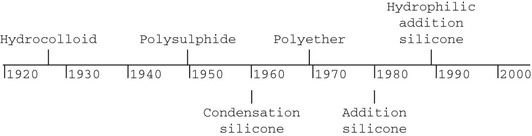
Elastic impression materials
The first elastic dental impression material was the hydrocolloid, which was introduced to dentistry in 1925. Since then, many elastomeric impression materials have become available, as indicated on the following time line:
Colloids
The word colloid is derived from the word kola, meaning glue, and oid, meaning like. Thus a colloid has a glue-like physical character.
The colloidal state represents a highly dispersed phase of fine particles within another phase, somewhere between a solution and a suspension:
The difference between a colloid and a suspension is that in a colloid the dispersed phase is not readily detectable microscopically. Examples of colloids in dental use are:
The size of the finely dispersed phase cannot easily be defined accurately, but it is slightly greater than the simple molecular size, usually in the range of 1–500 nm.
The colloid can exist in the form of a viscous liquid, known as a sol, or a solid, described as a gel. If the particles are suspended in water, then the suspension is called a hydrocolloid, with the liquid being a hydrosol and the solid a hydrogel. In the case of the hydrogel, there is an entanglement network of solid particles with the liquid trapped in the interstices. The solid particles are in the form of fibrils or chains of molecules.
Agar
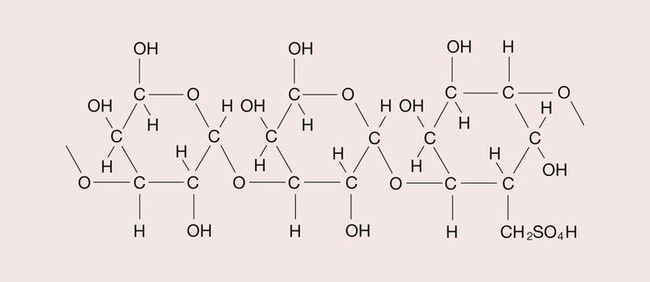
Agar is a galactose sulphate, which forms a colloid with water. It liquefies between 71°C and 100°C and sets to gel again between 30°C and 50°C. It is a long-chain molecule, with a molecular weight of approximately 150 000. The structure of this polysaccharide is shown in Figure 2.7.3. The hydroxyl (OH) groups undergo hydrogen bonding, leading to the formation of a helical structure.
When heated, this hydrogen bond is broken, the helix is uncoiled and the gel is turned into a viscous fluid. This process is therefore reversible in the manner shown below:
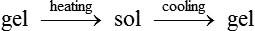
These materials are analogous to thermoplastics and have the advantage that they can be used repeatedly.
The agar is heated in a water bath until it becomes fluid. It is placed in a special metal tray through which water can be passed when it is placed in the patient’s mouth. The water cools the agar, whereupon it resolidifies as a gel, having taken up the shape of the oral tissues.
Composition
The composition and purpose of the various ingredients of a typical agar impression material are as shown in Table 2.7.5. As can be seen from the composition, only a small amount of agar is needed to form a gel.
Presentation and application
The material is provided in tube-like sachets for loading the tray, or in a syringe for easy adaptation to the teeth. The agar content of the syringe-applied material is lower than that of the tray material, so it is more fluid and easy to eject from the syringe and inject around the teeth.
When immersed in a temperature-controlled water bath, the material turns into a viscous liquid after approximately 8–12 minutes, and can be left for several hours. It is important that the material is not overheated, as this will cause breakdown of the polymer.
The water bath usually consists of three compartments, each held at a different temperature. One compartment contains water near its boiling point and is used for liquefying the agar. A second compartment is kept at 63–66°C for storing the agar. The third compartment is kept at 46°C and is used for tempering the agar after it is placed in the special water-cooled tray. This compartment is necessary to ensure that the agar is cooled to a temperature that the patient finds acceptable, and that will not burn the tissues. Due to the relatively small amount used, the syringe-applied material does not require tempering and can be kept in the storage compartment.
The contents of the tube are squeezed into an impression tray and placed in the tempering bath. Once the agar has cooled sufficiently, which takes about 2 minutes, the tray is placed in the patient’s mouth. The water supply is connected only at this stage.
The temperature of the water for the water-cooled tray should be around 13°C, so as to be comfortable for the patient; if it is too cold, the resultant thermal shock can cause considerable pain and discomfort. The water is allowed to circulate through the tray and after about 5 minutes the agar will have solidified. The tray can now be removed from the patient’s mouth and an accurate reproduction of the tissues obtained.
Should the material not be used, for whatever reason, it can be used again at a later date. The time required to reliquefy the material may be somewhat longer and can add up to 4 minutes to the process. Each time the material is heated, it will cause some breakdown of the polymer structure and the agar will become noticeably stiffer. Thus, />
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses