Gingival and Periodontal Diseases
➧. Classification System in Periodontal Disease
Gingival Diseases Associated with Medications
Gingival Diseases Associated with Systemic Diseases
Gingival Diseases Associated with Malnutrition
Gingival Diseases Associated with Heredity
Gingival Diseases Associated with Ulcerative Lesions
Gingival Lesions Manifested in Childhood Diseases
Gingival Lesions Associated with Chicken Pox
Gingival Lesions Associated with Mononucleosis
Currently, the most accepted classification is the ‘1999 World Workshop Classification System’.
Classification System in Periodontal Disease
The classification is based on the 1999 International Workshop for Periodontal Disease.
A Dental plaque-induced gingival diseases: These diseases may occur on a periodontium with no attachment loss or on one with attachment loss that is stable and not progressing.
1. Gingivitis associated with dental plaque only
2. Gingival diseases modified by systemic factors
B Non-plaque-induced gingival lesions
1. Gingival diseases of specific bacterial origin
2. Gingival diseases of viral origin
3. Gingival diseases of fungal origin
4. Gingival lesions of genetic origin
5. Gingival manifestations of systemic conditions
A Localized (less than 30% of sites involved)
B Generalized (more than 30% of sites involved)
C Slight (1–2 mm clinical attachment loss)
IV Periodontitis as a manifestation of systemic diseases
A Associated with hematological disorders
V Necrotizing periodontal diseases
VI Abscess of the periodontium
VII Periodontitis associated with endodontic lesions
VIII Developmental or acquired deformities and conditions
A Localized tooth-related factors that modify or predispose to plaque-induced gingival diseases/ periodontitis
B Mucogingival deformities and conditions around teeth
i. Gingival/soft tissue recession, facial or lingual surfaces, interproximal (papillary)
ii. Lack of keratinized gingiva
iii. Decreased vestibular depth
C Mucogingival deformities and conditions on edentulous ridges
Gingival Diseases
Plaque Microbiology
Terminology
Biofilm: Biofilm is the term that describes the relatively indescribable microbial community associated with tooth surface or any other hard non-shedding material.
Host–Microbial Interaction
Defense Mechanism
These protect the body from attack from microorganisms and can be classified as:
Specific mechanisms
 T-cell response (cell-mediated immune system)
T-cell response (cell-mediated immune system)
 B-cell response (antibody-mediated immune system). Plaque bacteria produce a number of factors (virulence factor), which causes disease directly, or individually by stimulating the immune and inflammatory system. It is now known that individuals prone to periodontal disease have an aberrant immune inflammatory response to plaque which is genetically determined.
B-cell response (antibody-mediated immune system). Plaque bacteria produce a number of factors (virulence factor), which causes disease directly, or individually by stimulating the immune and inflammatory system. It is now known that individuals prone to periodontal disease have an aberrant immune inflammatory response to plaque which is genetically determined.
Plaque-induced Gingivitis
In the initial and established stages of gingivitis, dental plaque is predominantly comprised of gram-positive aerobic microorganisms including Streptococcus mitis, S. sanguis, Actinomyces viscosus, A. naeslundii and Eubacterium spp. As age advances, development and severity of gingivitis is mainly dependent on the quality of the plaque rather than the quantity and the other contributing factors like the host immune response, environmental, genetic and the behavioral factors. The common clinical signs and symptoms of gingivitis include redness, edema, bleeding on probing, tenderness and enlargement (Loe et al, 1965; Suzuki, 1988) (Figure 1).
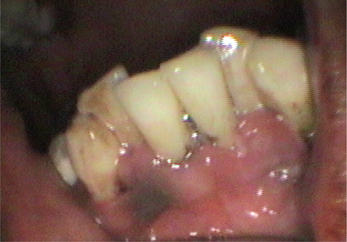
Figure 1 Edematous gingival margin associated with gingivitis. Courtesy: Department of Oral Medicine and Radiology, MCODS, Mangalore
Radiographically no changes will be seen as the inflammation is confined only to the gingival sulcus. Histologic changes include proliferation of basal junctional epithelium leading to apical and lateral cell migration, vascular dilatation and vasculitis of blood vessels adjacent to the junctional epithelium, progressive destruction of collagen fibers, cytopathologic alteration of resident fibroblasts and progressive inflammatory cell infiltrate (Page and Shroeder, 1976).
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses











