Oral Cancer
Incidence of Cancer of Head and Neck
It has been estimated that 43% of cancer deaths worldwide are due to tobacco, unhealthy diet, physical inactivity and infections. Tobacco use and excessive alcohol consumption have been estimated to account for about 90% of cancers in the oral cavity; the oral cancer risk increases when tobacco is used in combination with alcohol or areca nut.
Etiology and Risk Factors for Oral and Maxillofacial Cancer
Tobacco
Consumption of Tobacco in India
It is estimated that 80–85% of tobacco is consumed for smoking either as beedis or cigarettes (Figure 1). Almost 13% chew tobacco in the form of paan (Figure 2, betel leaf, areca nut, tobacco, slaked lime and flavoring agents) or gutkha. Almost 15% are addicted to both habit of chewing and smoking. Only about 1–3% use tobacco in the form of snuff.
Smokeless Form of Tobacco
People in the Indian subcontinent exhibit various forms of tobacco chewing habits such as khaini, mishri, zarda, gutkha (Figure 3), mawa and nass. Paan is chewed (betel quid) and the quid is usually placed in the buccal vestibule.

Figure 3 One of the popularly consumed packets of gutkha. Courtesy: Department of Oral Medicine and Radiology, Manipal College of Dental Sciences, Mangalore
Tobacco is also used in the form of a powder for inhalation (Figure 4, snuff). Tobacco (Figure 5) contains nicotine (nitrosamine), nitrosodiethanolamine, nitrosoproline, polonium and polycyclic aromatic hydrocarbon (tars). Areca nut (Figure 6 contains cholinergic muscarinic alkaloids such as arecholine and guavacoline) chewing is also widely practiced in India.

Figure 4 Snuff used for inhalation. One of the risk factors for causing nasopharyngeal and maxillary sinus cancers. Courtesy: Department of Oral Medicine and Radiology, Manipal College of Dental Sciences, Mangalore
Smoke Form of Tobacco
In India, beedi (Figure 7), cigarette and chutta smoking is commonly seen. However, other popular forms of smoking include use of pipe, cigar and hookah. It is believed that cigar and pipe smoking is more hazardous than cigarette smoking. Palatal cancers are more common in individuals who place the lit end of the beedis and cigarettes inside the mouth (reverse smoking). It has been proposed that smoking causes pooling of carcinogens in saliva thereby increasing the incidence of cancers of the floor of the mouth, ventral and lateral surface of tongue.

Figure 7 Beedis made of tobacco wrapped in a tendu (Diospyros melanoxylon) leaf, and secured with thread at one end. The tobacco content in beedis is approximately 10–20%. Courtesy: Department of Oral Medicine and Radiology, Manipal College of Dental Sciences, Mangalore
It is estimated that in the combustion mainstream of one cigarette (Figure 8), there are approximately 500 mg (92%) of gaseous content (mainly oxygen, nitrogen and carbon dioxide and to a little extent carbon monoxide) and 8% of particulate matter. Aromatic hydrocarbons in the form of ‘tars’ may constitute less than 1 g to 35 mg. Nicotine content varies from 1 to 3 mg. Other constituents of tobacco smoke include carbon monoxide, hydrogen cyanide and thiocyanate. Benzopyrene is considered the most potent carcinogen. It preferentially binds to nucleoproteins. The enzyme aryl hydrocarbon hydroxylase that is principally produced in human leukocytes, is said to increase the carcinogenic potential of benzopyrene.
Alcohol
Alcohol consumption is the most important risk factor for development of oral cancer in non-smokers. It is also considered as the second independent major risk factor for the development of oral cancer. It is estimated that an average consumption of over 30 ml of alcohol per day increases the risk of oral cancer linearly with the quantity of alcohol consumed. Though any form of alcohol when consumed in large quantities is dangerous, it is believed that dark colored drinks are more hazardous (as they may contain higher beverage congeners such as nitrosamines, hydrocarbons and other impurities which are known carcinogens). Carcinogens present in the tar are insoluble in saliva but are highly soluble in alcohol and easily absorbed in the oropharynx.
 Ethyl alcohol increases the permeability of oral mucosa. It also dehydrates the mucosa. It has a solvent action on the keratinocyte membrane thereby allowing the passage of carcinogens into proliferating cells where they may exert a mutagenic action.
Ethyl alcohol increases the permeability of oral mucosa. It also dehydrates the mucosa. It has a solvent action on the keratinocyte membrane thereby allowing the passage of carcinogens into proliferating cells where they may exert a mutagenic action.
 The immediate metabolite of ethanol is acetaldehyde, which readily damages cells. It is believed that those who are rapid acetylators are at increased risk of developing cancers.
The immediate metabolite of ethanol is acetaldehyde, which readily damages cells. It is believed that those who are rapid acetylators are at increased risk of developing cancers.
 Alcoholic liver disease is common in heavy drinkers and this minimizes the detoxification of carcinogens.
Alcoholic liver disease is common in heavy drinkers and this minimizes the detoxification of carcinogens.
 Alcohol, owing to its high calorific value, suppresses appetite in heavy drinkers. This predisposes to nutritional deficiency which in turn is a risk factor for cancers. Compared to non-users, alcohol users are 3.6 times more likely to have oropharyngeal cancer, 5.8 times for tobacco users, and 19 times for users of both alcohol and tobacco.
Alcohol, owing to its high calorific value, suppresses appetite in heavy drinkers. This predisposes to nutritional deficiency which in turn is a risk factor for cancers. Compared to non-users, alcohol users are 3.6 times more likely to have oropharyngeal cancer, 5.8 times for tobacco users, and 19 times for users of both alcohol and tobacco.
Systemic Health
Syphilis
Michalek et al (1994) reported a study of 16,420 people with syphilis resident in the United States that showed a significantly raised frequency of cancer of the tongue (and Kaposi’s sarcoma) in males. Dickenson et al (1995) screened 63 patients of United Kingdom suffering from squamous cell carcinoma of the tongue. Five of these patients had serological evidence of past syphilis when detected by both specific and non-specific tests.
Role of Viruses
The role of viruses remains unclear. Varying viral genomes have been frequently found within cancer cells. Viruses have been known to modify the DNA and the chromosomal structures and induce proliferative changes in the cells they infect.
Evidence of a viral carcinogenesis is perhaps strongest for infection with human papilloma viruses (HPV). D’souza et al (2007) in a multicenter case-control study reported that infection with HPV-16 increased the risk of cancer of the oral cavity and particularly oropharynx.
Role of Nutrition
It has been shown that patients who ingest high levels of vitamin C and fiber have half the risk of developing oral cancer as those with minimal level of consumption. Block (1991) and Mirvish (1986) showed that a low intake of vitamin C is associated with an increased risk of cancers of the stomach, esophagus, oral cavity, larynx, and cervix.
Molecular Basis of Cancer
Tumor Growth and Metastasis
As a tumor grows, nutrients are provided by direct diffusion from the circulation. Local growth is facilitated by enzymes (e.g. collagenases) and cytokines that alter or destroy adjacent tissues. As the ratio of surface area to volume becomes smaller with increased tumor growth, tumor angiogenesis factors are produced, forming the independent vascular supply required for further tumor growth. Almost from inception, a tumor may shed cells into the circulation. From animal models, it is estimated that a 1-cm tumor sheds more than 1 million cells/24 h into the venous circulation. Although most circulating tumor cells die as a result of intravascular trauma, a tiny number (much less than 1 in 1 million) adhere to the vascular endothelium and penetrate into surrounding tissues, generating independent tumors (metastases) at distant sites. Metastatic tumors grow in much the same manner as primary tumors and may subsequently give rise to other metastases. Experiments suggest that metastasis is not a random event and that the primary tumor may regulate the growth of metastatic tumors, for example, removal of the primary tumor sometimes results in rapid growth of the metastases.
Clinical Signs of Cancer
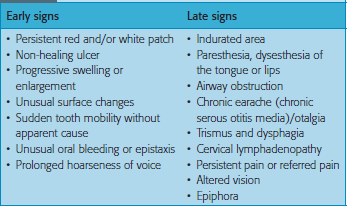
The many signs and symptoms of oral cancer (Table 1) are usually divided into early and late presentations. They can be so diverse that the differential diagnosis may not lead to oral malignancy.
The clinical presentation varies in most cases and there are no two cases that are similar in presentation and treatment. However, there could be a few similarities that are peculiar to the geographic area that could be grouped together and explained as an entity, for example, carcinoma of the buccal mucosa and the gingivobuccal sulcus that are peculiar to the Indian subcontinent and have been nicknamed as ‘Indian Cancers’. Cancers of the floor of the mouth are more common in the western countries because of their habits. The following section is just a brief explanation of the various clinical signs and symptoms based on their area of presentation.
Carcinoma of buccal mucosa
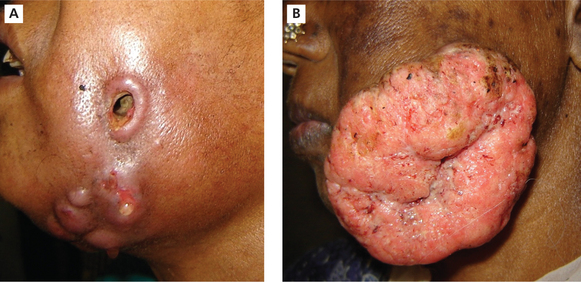
The lesions develop most frequently along or inferior to a line opposite the plane of occlusion. It usually occurs at the regions of the third molar area (Figure 9) as there is a habit of keeping the quid (tobacco) in that area for a long period of time. The tumor may appear as small nodules and enlarges to from a wart-like growth which ultimately ulcerates or may even begin as an ulceration that does not heal. The lesion is often not painful and is noticed by the patient only when there is a secondary infection of the ulcer. There is induration and infiltration into the deeper tissues. Extension into the muscle of mastication, buccinators, alveolar mucosa and ultimately into the bone may occur and if left unchecked which may cause perforation of the overlying skin (Figure 10). The induration of the skin is a bad clinical sign and necessitates wider excision along with the overlying skin.
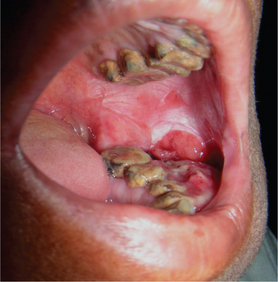
Figure 9 Carcinoma of the left buccal mucosa extending into the buccal vestibule. Courtesy: Dr Abhinandan
Some cases appear to be growing outward from the surface rather than invading the tissues is called exophytic or verrucous growth. The most common site of metastasis is the submandibular lymph nodes as they are the primary echelon nodes for these regions.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses