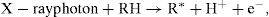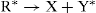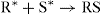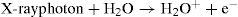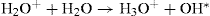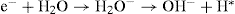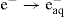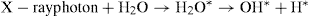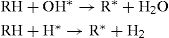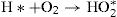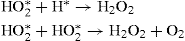Radiation Biology
Effects on Living Systems
Direct effect
These reactions may be represented as follows:
The altered molecules differ structurally and functionally from the original molecules resulting in a biological change in the irradiated tissues.
Indirect effect
This positively charged water molecule reacts with another water molecule, thus:
The free electron produced in the first reaction combines with a water molecule:
The electron may also be dissolved in the solution to form aqueous electron, e−aq.
X-ray photons may act directly on water molecules to produce electronically excited water molecules, which in turn breakdown to hydroxyl and hydrogen radicals:
The hydrogen free radical combines with dissolved oxygen to form hydroperoxyl free radicals:
Molecular and Cellular Radiobiology
Radiation Effects in DNA
Radiation causes a wide range of lesions in DNA such as:
Cell cycle effects
 First Gap (G1) phase or phase characterized by division of synthesis of organelles
First Gap (G1) phase or phase characterized by division of synthesis of organelles
 Synthetic (S) phase characterized by division of DNA
Synthetic (S) phase characterized by division of DNA
 Second Gap (G2) phase characterized by preparation for mitosis
Second Gap (G2) phase characterized by preparation for mitosis
Radiosensitivity differs throughout the cell cycle with late S phase being most radioresistant, G2/M being most radiosensitive and G1 phase taking an intermediate position. In G2/M phase, chromatin is tightly compacted and a poor repair capacity. This explains the high radiosensitivity of this period. DNA synthesis is almost complete by late S phase. Damage by X-rays at this phase can be repaired, and thus the cell is radioresistant at this phase. DNA has an open structure in G1 phase having a better repair capacity; this makes G1 phase radioresistant.
Deterministic and Stochastic Effects
Deterministic Effects on Tissues and Organs
Cellular and tissue response
Effects of Total Body Radiation
Acute radiation syndrome
Hematopoietic syndrome
It occurs after exposure to 2–7 Gy of radiation. It occurs due to the exposure to active hematopoietic areas such as sternum and pelvis. This results in destruction of the blood cell precursors, which in turn leads to lower levels of peripheral blood cells. Circulating blood cells themselves are not affected by radiation. Granulocytes which have a short lifespan, are not replaced by maturation of precursor cells. This predisposes to infections. As thrombocytes disappear in peripheral blood, bleeding ensues. As erythrocytes, which have a long lifespan, disappear, anemia occurs.
Effects of Radiation on Oral Tissues
This results in several effects which are described below.
Mucositis
Based on severity, mucositis has been classified into four grades:
Xerostomia
Salivary glands are frequently in the path of radiation and are exposed during radiotherapy. The parenchyma of these glands is radiosensitive, which finally results in a fibrosis of the gland. This results in a progressive decrease in salivary secretion, termed xerostomia. This decrease is dose dependent and secretion reaches zero at about 60 Gy. Patients complain of dry mouth and difficulty in swallowing. As lubricating properties of saliva are lost and its pH decreases, demineralization of enamel begins.
Radiation caries
Three types of radiation caries have been described:
Type 1. The most common type. It is characterized by caries occurring around the teeth in the cervical region. As caries deepens, there is amputation of the crown.
Type 2. It is characterized by the initiation of caries on all crown surfaces, eroding away the entire coronal structure.
Type 3. The least common type. It is characterized by color changes in dentin resulting in a diffuse blackening or dark brown discoloration of the crown.
Radiation Safety and Protection
Sources of Radiation (Table 1, Figure 1)
Radiation is the transmission of energy through space and matter. It is natural and part of our lives. The radioactive materials present naturally in the earth’s crust can be encountered in the building construction materials, food substances and the air we breathe. Muscles, bones, and tissues of our own bodies contain naturally occurring radioactive elements. It is estimated that about four-fifths of the average annual radiation dose worldwide is the contribution from natural radiation.
Table 1
Average effective dose of ionizing radiation from different sources
| Source | Dose (μSv) |
| Natural | |
| Cosmic | 0.4 |
| Terrestrial | |
| External | 0.5 |
| Radon | 1.2 |
| Other | 0.3 |
| Total | 2.4 |
| Man-made | |
| Medical (estimated) | |
| Diagnostic X-ray | 2 |
| Nuclear medicine | 0.5 |
| Other consumer products | 0.08 |
| Other | |
| Professional | 0.01 |
| Fallout | 0.01 |
| Nuclear fuel cycle | 0.01 |
| Dental radiology | 0.01 |
| Total | 2.5 |
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses