Allergic and Immunologic Diseases of the Oral Cavity
Recurrent Aphthous Stomatitis: (Aphthous ulcers, aphthae, canker sores)
Bacterial Infection
The work of Barile, of Graykowski and of Stanley very strongly implicated a pleomorphic, transitional L-form of an α-hemolytic Streptococcus, Streptococcus sanguis, as the causative agent of the disease. This organism has been consistently isolated from the lesions of patients with typical aphthous ulcers, and microorganisms morphologically consistent with the L-form Streptococcus have been found histologically in the vast majority of aphthous lesions. Once again, it should be emphasized that the herpes simplex virus cannot be isolated from these aphthous ulcers.
Allergic Factors
Many patients with recurrent aphthous ulcers have a history of asthma, hay fever, or food or drug allergies. This may be a purely fortuitous finding because of the high incidence of allergies in the general population. However, the outbreak of aphthae following the use of certain foods or drugs in the same patients has been reported so frequently that allergy must be considered a precipitating factor.
Classification
• Recurrent aphthous minor, which is the most common form of the disease and the one referred to by the lay public as the ‘canker’ sore.
• Recurrent aphthous major, which is now believed to be simply a more severe form of recurrent aphthous minor but which was thought at one time to represent a separate disease entity known as periadenitis mucosa necrotica recurrens (Mikulicz’s scarring aphthae or Sutton’s disease).
• Recurrent herpetiform ulcerations, which consist of clusters of ulcers resembling herpetic lesions but lacking evidence of the presence of viruses in patients with a low incidence of antibody to oral mucosa.
• Recurrent ulcers associated with Behçet’s syndrome, will be considered separately.
Clinical Features
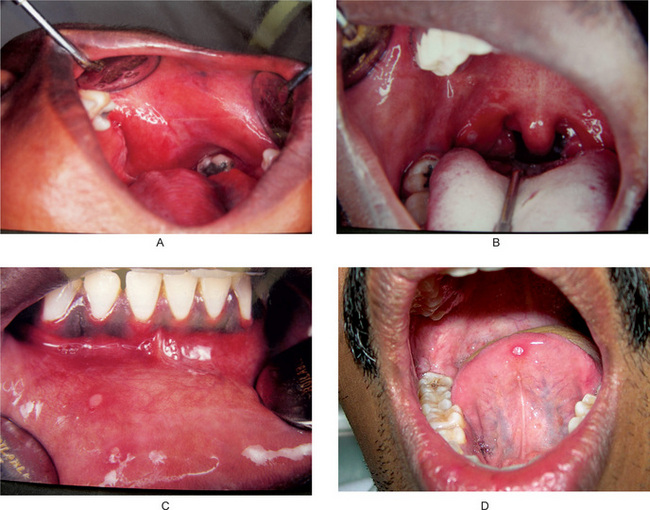
The aphthous ulcer begins as a single or multiple superficial erosions covered by a gray membrane (Fig. 16-1). It generally has a necrotic center with clearly defined raised margins surrounded by an erythematous halo. The lesion is typically very painful so that it commonly interferes with eating and speech for several days. At one time it was thought that the aphthous ulcer begins with the formation of a vesicle, as does the lesion of herpes simplex infection. The majority of the evidence now indicates that this is not the case—vesicle formation does not appear to be a stage in the development of the usual aphthous ulcer.
Recurrent aphthous major is characterized by the occurrence of large painful ulcers, usually 1 to 10 in number, on the lips, cheeks, tongue, soft palate, and fauces and cause severe pain and dysphagia (Fig. 16-2). Their incidence is more in patients with HIV infection. These ulcers occur at frequent intervals, and many patients with this disease are seldom free from the presence of at least one ulcer. Usually these lesions occur after puberty and persist up to 20 years or more. Unlike the typical ulcers of recurrent aphthous minor, these lesions may exceed one cm in diameter and persist for up to six weeks and leave a scar upon healing. Not uncommonly, the ulcers recur in waves over a long period of time, so that eventually the oral mucosa may show a great deal of scarring. Patients with these severe major aphthae also occasionally show similar lesions on vagina or penis, rectum, and larynx, with associated rheumatoid arthritis or conjunctivitis.
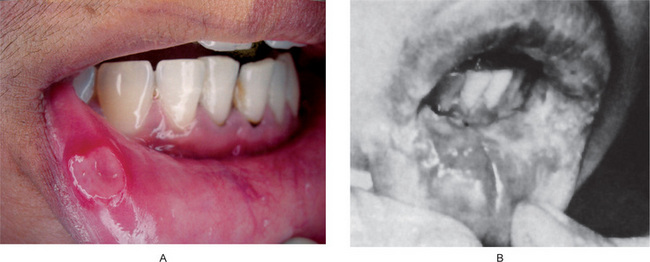
Figure 16-2 Recurrent aphthous ulcers, major. Deep crateriform ulcer (A) and scars (B). (B, Courtesy of Dr TH Century).
Recurrent herpetiform ulcers are characterized by crops of multiple small, shallow ulcers, often up to 100 in number, which may occur at any site in the oral cavity. They were first described by Cooke in 1960, while Lehner as well as Brooke and Sapp have expanded our knowledge of this condition. Cooke pointed out the clinical similarities of this disease to the lesions of herpes simplex and that the corresponding histologic changes were not similar, since these lesions resemble the recurrent aphthous ulcer rather than a viral lesion. These lesions have female predisposition with later age of onset and are not associated with herpes virus.
• Numerous small lesions may be found on any intraoral mucosal surface.
• Lesions begin as small pinhead-sized erosions that gradually enlarge and coalesce.
• Lesions are more painful than would be suspected by their size.
• Lesions are present almost continuously for one to three years, with relatively short remissions.
• Patients receive immediate but temporary relief from symptoms with a 2% tetracycline mouthwash.
• The herpes simplex virus cannot be cultured from the lesions or demonstrated by electron microscopy, although Sapp and Brooke have demonstrated non-viral intranuclear bodies in adjacent epithelial cells.
• Cytologic smears fail to reveal the typical multinucleated epithelial giant cells found in herpetic lesions.
• The microscopic findings are nearly identical with those described for the recurrent aphthous ulcer.
• Immunofluorescent and serologic techniques are negative for antibodies to herpes virus as well as to oral epithelium.
Histologic Features
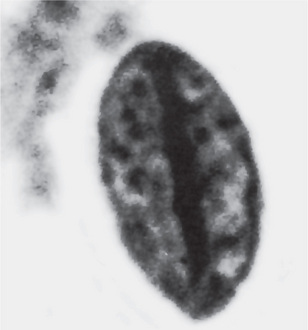
Wood and his associates have described characteristic changes in the nuclei of epithelial cells taken by cytologic smears from around recurrent aphthous ulcers. These have been referred to as Anitschkow cells and consist of cells with elongated nuclei containing a linear bar of chromatin with radiating processes of chromatin extending towards the nuclear membrane (Fig. 16-3). They are quite abundant in patients with recurrent aphthous stomatitis but are not pathognomonic of the disease, since they are also found in patients with sickle cell disease, megaloblastic anemias, and iron-deficiency anemias, in children receiving chemotherapy for cancer, and even in normal people. Their ultrastructure has been described by Haley and his associates, who found that the nuclear chromatin was made up of pleomorphic masses forming an irregular band along the long axis of the nucleus rather than being randomly dispersed.
Treatment
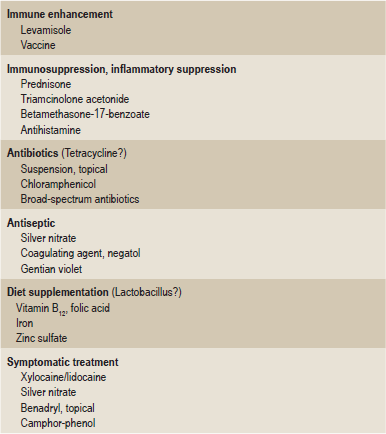
There is no specific treatment for recurrent aphthous ulcers although, over the years, many drugs have been advocated. Graykowski and his coworkers found that a tetracycline mouthwash (250 mg per 5 ml), used four times daily for 5 to 7 days, produced a good response in nearly 70% of the patients tested, by relieving the pain, reducing the size of the lesions, and reducing the healing time. A steroid ointment, 1.5% cortisone acetate, applied locally, and hydrocortisone acetate-antibiotic lozenges also showed some effectiveness but not as great as the tetracycline. Chemical cautery reduced pain but had no other beneficial effects. No significant improvement was found with the use of antihistaminics, gamma globulin, multiple smallpox vaccinations or a Lactobacillus acidophilus-L. bulgaricus preparation, all of which have variously been reported to be effective.
An excellent summary of the many drugs and chemicals which have been used to treat recurrent aphthous stomatitis over the years has been prepared by Antoon and Miller and is shown in Table 16.1. Unfortunately, despite many forms of therapy, there is no known cure for the disease.
Behçet’s Syndrome
Clinical Features
It is characterized chiefly by oral and genital ulcerations, ocular lesions, and skin lesions. The first manifestation of the disease is usually the appearance of oral and/or genital lesions. The oral lesions are painful and very similar clinically and histologically to that of recurrent aphthous ulcers. They occur in crops at any intraoral site and consist of ulcers ranging in size from several millimeters to a centimeter or more in diameter. These ulcers have an erythematous border and are covered by a gray or yellow exudate. The genital ulcers are small and located on the scrotum, root of the penis or labia majora. The most common sites of oral ulcers are lips, buccal mucosa, tongue, gingiva, palate, tonsils, uvula, and pharynx. It must be emphasized that oral ulcers of Behçet’s Syndrome are usually indistinguishable from ordinary recurrent oral ulcers except in multiplicity and involvement of unusual locations (soft palate and oropharynx). Cutaneous lesions include papulopustular lesions (acneiform) or pseudofolliculitis, erythema nodosum, superficial thrombophlebitis, cutaneous ulcers and nodules, cellulitis type lesions, Sweet’s syndrome, gangrenous pyoderma, other rarer lesions, which occur in addition.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


 . Recurrent Aphthous Stomatitis
. Recurrent Aphthous Stomatitis . Behçet’s Syndrome
. Behçet’s Syndrome . Reiter’s Syndrome
. Reiter’s Syndrome . Sarcoidosis
. Sarcoidosis . Uveoparotid Fever
. Uveoparotid Fever . Midline Lethal Granuloma
. Midline Lethal Granuloma . Wegener’s Granulomatosis
. Wegener’s Granulomatosis . Chronic Granulomatous Disease
. Chronic Granulomatous Disease . Angioedema
. Angioedema . Drug Allergy
. Drug Allergy . Contact Stomatitis and Dermatitis
. Contact Stomatitis and Dermatitis . Contact Stomatitis from Cinnamon Flavoring
. Contact Stomatitis from Cinnamon Flavoring . Contact Stomatitis from Chronic Oral Mucosal Contact with Dental Amalgam
. Contact Stomatitis from Chronic Oral Mucosal Contact with Dental Amalgam . Perioral Dermatitis
. Perioral Dermatitis . Latex Allergy
. Latex Allergy