Dental Implants
Objectives
After reading this chapter, the reader should be able to:
1. Describe the clinical treatment sequence used to place endosseous implants.
< ?mpslid E1?>< ?mpslid S2?>
2. Compare and contrast biointegration and osseointegration.
< ?mpslid E2?>< ?mpslid S3?>
< ?mpslid E3?>< ?mpslid S4?>
< ?mpslid E4?>< ?mpslid S5?>
< ?mpslid E5?>< ?mpslid S6?>
< ?mpslid E6?>< ?mpslid S7?>
7. Explain why ceramic coatings are applied to endosseous implants.
< ?mpslid E7?>< ?mpslid S8?>
< ?mpslid E8?>
Key Terms
Biointegration
Ceramic coating
Commercially pure titanium
Endosseous implants
Implant abutment
Implant-retained complete denture
Implant-supported denture
Locator abutment
Mini-implants
Osseointegration
Peri-implantitis
Titanium alloy
Zirconia
Attempts to implant a material into bone to replace a tooth dates back thousands of years, yet modern dental implantology is less than 40 years old. Implant placement into the maxilla or mandible creates complex interfaces between the implant material and the oral tissues. The implant material must not harm the hard or soft tissues and must withstand the physical and chemical environment of the oral cavity over a service life that may span decades. The tissues must remain viable and functional to retain the implant when it is placed under loads during chewing, brushing, speaking, and other oral functions.
This chapter focuses on the endosseous implant, which is the most common type of dental implant used today (Figure 15-1). Endosseous implants are placed into bone, as opposed to other types of implants that might be placed external to the bone. Today, the endosseous implant is used to solve increasingly complex clinical problems in an increasing number of individuals. More than 2 million endosseous implants were placed in the United States alone in 2009. It is likely that the role of endosseous implants will expand as more practitioners master placement techniques and new materials and strategies are developed to predictably treat complex clinical situations. Dental auxiliaries must understand the nature of implant materials to provide appropriate care for an increasing number of dental patients.
Important Principles
Implant Surgery
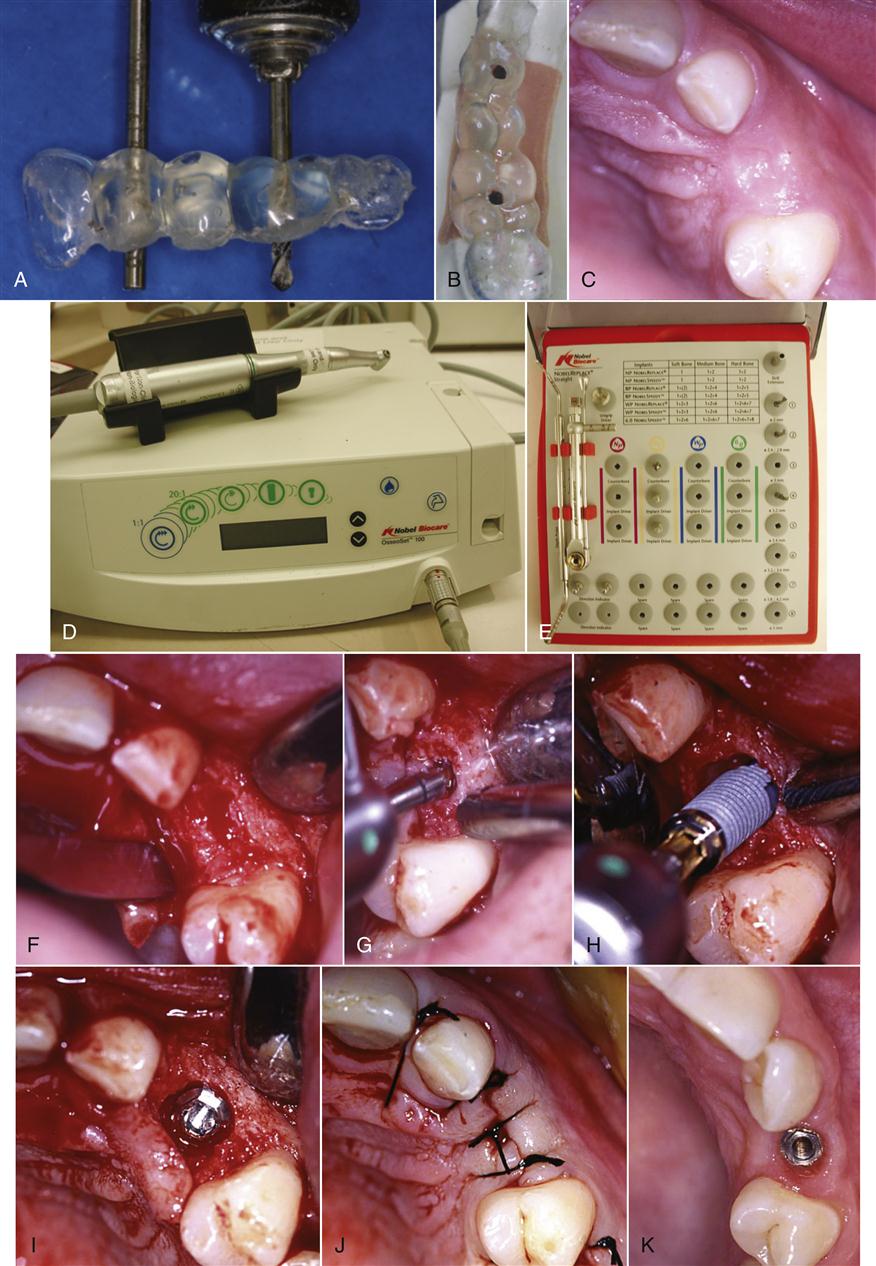
The successful placement of an endosseous implant relies on a careful and methodic series of surgical steps that preserve bony support for the implant and impart minimal trauma and inflammation that might impede healing (Figures 15-2 and 15-3). Traditionally, the extraction socket of a tooth is allowed to fill in with bone for 3 to 6 months before the implant is placed. Once healing is complete, the bone is prepared with special metallic burs driven at slow speeds (<2000 rpm) to specific depths with special handpieces and copious water cooling. Many times, the hole preparation (osteotomy) is guided by an acrylic stent prepared from a model, often with guidance from traditional radiographs or computed tomography (CT) scans. During the osteotomy, the objective is to avoid temperatures in the bone above 37° C that will damage tissues, cause protein coagulation, and trigger inflammation. The burs are composed of special alloys of titanium to avoid deposition of trace metal contaminants that may themselves cause adverse biologic reactions. The water coolant is sterile to avoid contamination of the placement site with bacteria or other microorganisms. The amount of bone removed is controlled carefully in a series of steps to coincide with the diameter and length of implant to ensure the bone and implant heal in close proximity to each other.
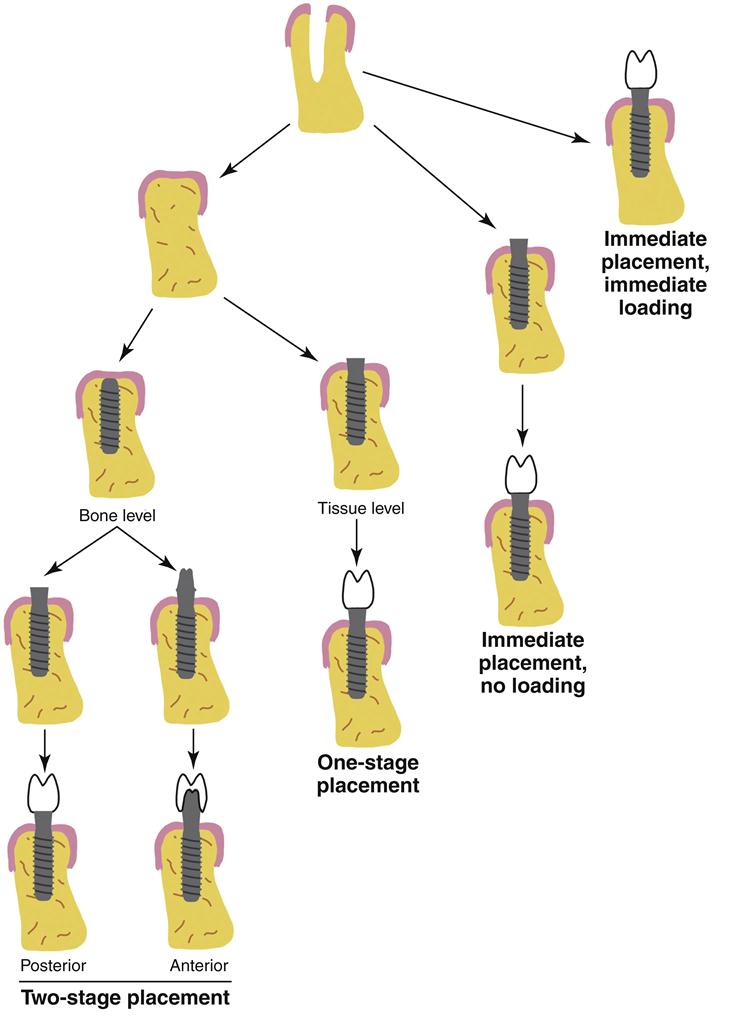
Once the implant site is prepared in the bone, implants may be placed to the bone level or tissue level. The screw-type implant is the most common style used today (see Figure 15-1) because it allows a snug fit of the implant into the socket and controlled application of force to final position of placement. As with the surgical preparation, special instruments are used in an atraumatic manner to ensure preservation of the bone and minimal contamination of the wound site. The implant itself is meticulously handled to avoid chemical or microbial contamination.
Depending on the type of implant placed, several strategies are possible (see Figure 15-2). For tissue-level implants, the soft tissue may be sutured around the implant, which will be left protruding through the tissue, and a small cap will be placed to prevent contamination of the internal parts of the implant. This strategy is called a one-stage surgery; it is important to understand that the implant is generally not in occlusion during the healing. For bone-level implants, two strategies may be used. In a one-stage surgery, a healing abutment with a cap is placed immediately after the implant, and the tissue is managed around the abutment as with the tissue-level implant. In a two-stage surgery, a healing cap is placed on the implant and the tissue is positioned over the cap to allow healing with the implant submerged. In this case, a second surgery is needed to place the abutment. Two-stage surgeries may be used when there are questions about the likelihood of osseointegration or when other procedures such as bone grafting are done at the time of the initial implant. In all cases, the implant is allowed to heal for at least 6 weeks (generally 2 to 3 months) without occlusal loading. The avoidance of any movement (<100 µm) is imperative to successful integration of the implant with the bone.
The ability to satisfactorily restore the teeth is determined, to a large extent, on the position of the implants. Thus, advanced planning by the dentist and good communication with the surgeon are essential to ensure that the restoration will be able to be appropriately constructed and placed. The dental auxiliary is a critical link in this chain of communication. Several types of final abutments are possible (see Figure 15-2). In posterior teeth, a platform abutment is common; this type of abutment is a relatively flat junction onto which the posterior implant crown is fabricated (see Figure 1-9). In anterior teeth, a tapered abutment, either with a standard shape or custom made, is most often used. Once the final abutments are in place, the prosthesis is constructed in much the same manner as for natural teeth. The prosthesis may be cemented permanently or screwed into place with a small screw that penetrates the top of the crowns and threads into the abutments. The use of screws allows the prosthesis to be removed if necessary to permit reevaluation or cleaning of the implants. The hole for the screw is often filled with a small composite to obscure the opening.
The placement strategies described in the previous paragraphs are currently the most common and reliable methods of ensuring a successful result. However, some clinicians have advocated the placement of an implant into the extraction socket of a tooth immediately after extraction (see Figure 15-2). A few clinicians promote the immediate loading of an implant after its placement at an extraction site. However, all of these ideas are controversial and far less common than the two-stage, no-loading procedure previously described. Success rates with immediate loading or immediate placement are generally lower.
Osseointegration and Biointegration
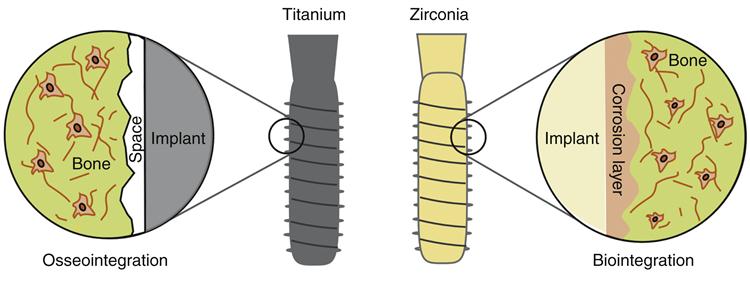
For dental implants made of titanium alloy, the interface between the alloy and bone must be nearly direct with no intervening fibrous tissue and as little space as possible. This special interfacial condition is called osseointegration and occurs when the bone grows to within 100 Å (1 angstrom = 10−10 meters) of the titanium surface (Figure 15-4). Within the 100 Å, there is only extracellular matrix and no collagen or fibrous tissue. This degree of proximity is an extremely rigorous requirement achieved only if the surgical technique is atraumatic (see the previous discussion on surgical techniques) and the material does not have adverse biologic effects. To date, only a few materials, including commercially pure titanium, titanium-aluminum-vanadium alloy, tantalum, and several ceramic materials such as zirconia, are known to permit osseointegration (see the later discussion on implant materials). When the implant is osseointegrated, virtually no mobility of the implant in the bone is clinically detectable, and to the clinician it feels similar to the pathologic condition of natural tooth ankylosis. For an implant to be deemed successful, osseointegration must be maintained throughout the lifetime of the implant.
For ceramic implant materials made of zirconia, the integration of the ceramic with the bone has no intervening space (see Figure 15-4). Rather, a continuity exists from the implant to the bone. This interface condition is called biointegration. Biointegration is thought to require a chemical degradation of the ceramic implant that favors bone formation and is able to integrate with the surrounding bone, which itself has a significant ceramic component. The nature of the biointegrative interface and material characteristics that promote and maintain it are not fully understood. Furthermore, the advantages of biointegration over osseointegration are not known, particularly over the long term. Both interfaces are similar to the clinical ankylosis of natural teeth. Metallic implants coated with a ceramic initially promote a biointegrated interface, but long term, the stability of the interface is less clear because coatings tend to degrade with time.
Implants and Force
Although endosseous implants provide an esthetic substitute for a lost or missing tooth, they also are placed to restore lost masticatory function. Thus, the interface between the bone and the implant must withstand the forces generated during chewing and other oral habits for up 40 years. Research has shown definitively that excessive force will compromise osseointegration or biointegration, but the amount of force and the clinical conditions that lead to deterioration of bony support (versus forces that bone can tolerate) are not fully understood. Presumably, a prime consideration is the fundamental difference between the implant–bone interface and the tooth–bone interface. These two interfaces transmit occlusal forces quite differently (Figure 15-5). In the natural state, occlusal forces are transferred from tooth to bone via the periodontal ligament as tensile forces. Therefore, a downward (apical) force on the tooth results in a tensile force on the bone. These tensile forces stimulate bone deposition, limit bone resorption, and favor the maintenance of the bony support. Thus, unless the forces are excessive or grossly misdirected, occlusal loads on natural teeth favor bone maintenance. However, in the dental implant, no periodontal ligament exists, and occlusal loads are transmitted to the bone mostly by compressive forces. These compressive forces tend to cause resorption of bone. Thus, the design of implant-supported dental prostheses must limit the magnitude of compressive force transmitted to bone to levels below that which causes bone resorption. Limiting compressive stress in bone is one area of active investigation in implantology research. Of particular interest is how much compressive force may be tolerated without resorpti/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses