Introduction to Amalgam Restorations
Lee W. Boushell, Terence E. Donovan and Theodore M. Roberson
Amalgam
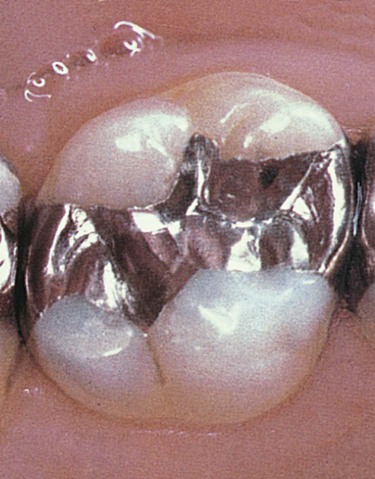
Dental amalgam is a metallic restorative material composed of a mixture of silver–tin–copper alloy and mercury. The unset mixture is pressed (condensed) into a specifically prepared undercut tooth form and contoured to restore the tooth’s form and function. When the material hardens, the tooth is functional again, restored with a silver-colored restoration (Fig. 13-1). Dental amalgam is described in detail in Online Chapter 18. In this book, dental amalgam is referred to as amalgam. Amalgam has been the primary direct restorative material for more than 150 years in the United States. It has been the subject of intense research and has been found to be safe and beneficial as a direct restorative material.1–3 Many people have benefited from amalgam restorations, which restore a tooth in the most cost-effective manner. The U.S. Public Health Service stated, “In fact, hundreds of millions of teeth have been retained that otherwise would have been sacrificed because restorative alternatives would have been too expensive for many people.”4
History
Amalgam was introduced to the United States in the 1830s. Initially, amalgam restorations were made by dentists filing silver coins and mixing the filings with mercury, creating a putty-like mass that was placed into the defective tooth. As knowledge increased and research intensified, major advancements in the formulation and use of amalgam occurred. Concerns about mercury toxicity in the use of amalgam were, however, expressed in many countries; concerns reached major proportions in the early 1990s. The American Dental Association (ADA) and the U.S. Public Health Service have issued many statements expressing their support for the use and safety of amalgam as a restorative material.1,4
Current Status
Today, the popularity of amalgam as a direct restorative material has decreased.5,6 This decline is attributed, in part, to the reduction in caries rates and to esthetic concerns. However, the primary cause of the reduction in the use of amalgam is the recognition of the benefits and improved esthetics of composite as a restorative material. Thus, concerns about the use of amalgam restorations relate primarily to poor esthetics and the greater potential for the weakening of the tooth structure: Dental amalgams inherently require greater removal of tooth structure to accommodate its strength requirements.
Even with the concern about the disposal of mercury, this textbook advocates the continued use of amalgam as a direct restorative material, especially in light of the finding that bonded composite resin restorations have an increased risk of development of secondary caries.7,8 Research repeatedly has shown the safety of amalgam and the success of restorations made from amalgam. Although the scope of the clinical uses of amalgam presented in this book are narrower than in the past, amalgam still is recognized as an excellent material for restoring many defects in teeth.
Types of Amalgam Restorative Materials
Low-Copper Amalgam
High-Copper Amalgam
High-copper amalgams are the materials predominantly used today in the United States. In this book, unless otherwise specified, the term amalgam refers to high-copper dental amalgam. The increase in copper content to 12% or greater designates an amalgam as a high-copper type. The advantage of the added copper is that it preferentially reacts with the tin and reduces the formation of the more corrosive phase (gamma-two) within the amalgam mass. This change in composition reduces possible deleterious corrosion effects on the restoration. However, enough corrosion occurs at the amalgam–tooth interface to result in the successful sealing of the restoration.9,10 These materials can provide satisfactory performance for more than 12 years.11,12 High-copper materials can be either spherical or admixed in the composition.
Spherical Amalgam
A spherical amalgam contains small, round alloy particles that are mixed with mercury to form the mass that is placed into the tooth preparation. Because of the shape of the particles, the material is condensed into the tooth preparation with little condensation pressure. This advantage is combined with its high early strength to provide a material that is well suited for very large amalgam restorations such as complex amalgams or foundations.13
New Amalgam Alloys
Because of the concern about mercury toxicity, many new compositions of amalgam are being promoted as mercury-free or low-mercury amalgam restorative materials. Alloys with gallium or indium or alloys using cold-welding techniques are presented as alternatives to mercury-containing amalgams. None of these new alloys shows sufficient promise to become a universal replacement for current amalgam materials.14–16
Important Properties
The linear coefficient of the thermal expansion of amalgam is 2.5 times greater than that of tooth structure, but it is closer than the linear coefficient of thermal expansion of composite.17–19 Although the compressive strength of high-copper amalgam is similar to tooth structure, the tensile strength is lower, making amalgam restorations prone to fracture.8,20,21 Usually, high-copper amalgam fracture is a bulk fracture, not a marginal fracture. All amalgams are brittle and have low edge strength. The amalgam material must have sufficient bulk (usually 1 to 2 mm, depending on the position within the tooth) and a 90-degree or greater marginal configuration.
Creep and flow relate to the deformation of a material under load over time. High-copper amalgams exhibit no clinically relevant creep or flow.22,23 Because amalgam is metallic in structure, it also is a good thermal conductor. An amalgam restoration should not be placed close to the pulpal tissues of the tooth without the use of a liner or base (or both) between the pulp and the amalgam.
Amalgam Restorations
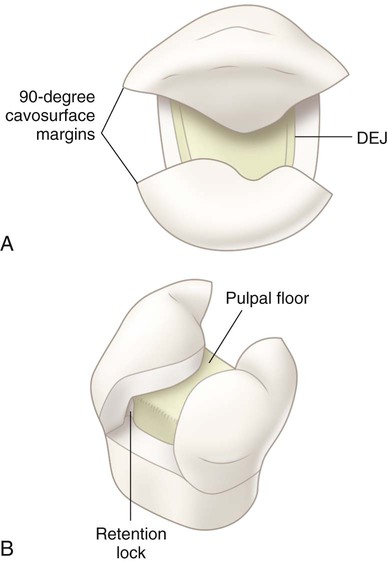
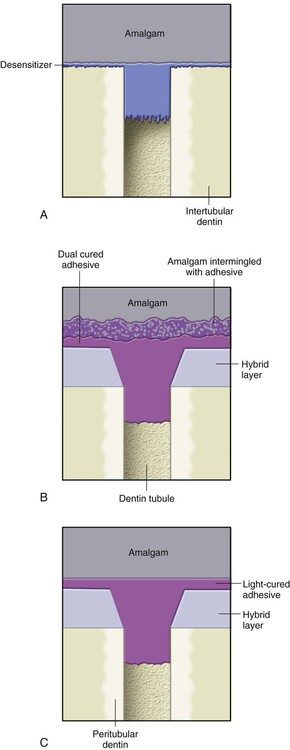
Amalgam functions as a direct restorative material because of its easy insertion into a tooth preparation and, when hardened, its ability to restore the tooth to proper form and function. The tooth preparation form not only must remove the fault in the tooth and remove weakened tooth structure, but it must also be formed to allow the amalgam material to function properly. The required tooth preparation form must allow the amalgam to (1) possess a uniform specified minimum thickness for strength, (2) produce a 90-degree amalgam angle (butt-joint form) at the margin, and (3) be mechanically retained in the tooth (Fig. 13-2). Without this preparation form, the amalgam possibly could be dislodged or could fracture. After desensitizing the prepared tooth structure, mixing, inserting, carving, and finishing the amalgam are relatively fast and easy (Fig. 13-3, A). For these reasons, it is a user-friendly material that is less technique sensitive or operator sensitive compared with composite.
Some practitioners have continued to use bonded amalgam restorations in their practice (see Fig. 13-3, B). As noted previously, this book no longer promotes the use of bonded amalgams.24–27 The mechanism of bonding an amalgam restoration is similar to that for bonding a composite restoration in some aspects, but it is different in others. A bonded amalgam restoration, done properly, may seal the prepared tooth structure and may strengthen the remaining unprepared tooth structure. The retention gained by bonding, however, is minimal; consequently, bonded amalgam restorations still require the same tooth preparation retention form as do nonbonded amalgam restorations.28,29 Isolation requirements for a bonded amalgam restoration are the same as for a composite restoration.
Another amalgam technique uses light-cured adhesive to seal the dentin under the amalgam material (see Fig. 13-3, C). This procedure, as is true of all procedures that use adhesive technology, requires proper isolation. The prepared tooth structure is etched, primed, and sealed with adhesive. The adhesive is polymerized before insertion of the amalgam. (Usually, a one-bottle sealer material that combines the primer and the adhesive is used.) This technique seals the dentinal tubules effectively.30,31
Uses
Because of its strength and ease of use, amalgam provides an excellent means for restoring large defects in non-esthetic areas.32 A review of almost 3500 4-surface and 5-surface amalgams revealed successful outcomes at 5 years for 72% of the four-surface and 65% of the five-surface amalgams. This result compared favorably with the 5-year success rates for gold and porcelain crowns, which were 84% and 85%, respectively.33 Generally, amalgams can be used for the following clinical procedures:
Handling
Because of the concern about mercury, amalgam restorations require meticulous handling to avoid unnecessary mercury exposure to the environment, the office, the personnel, or the patient. Proper mercury hygiene procedures are described in Online Chapter 1 and in the subsequent chapters on amalgam restorations.
General Considerations for Amalgam Restorations
The following sections summarize general considerations with regard to all amalgam restorations. Information for specific applications is presented in Chapters 14, 15, and 16. Because the typical decision about direct restorative materials is usually a choice between amalgam and composite, some of the following information involves a comparative analysis of these two materials.
Indications
Occlusal Factors
Amalgam has greater wear resistance than does composite.2,34 It may be indicated in clinical situations that have heavy occlusal functioning. It also may be more appropriate when a restoration restores all of the occlusal contacts of a tooth.
Clinical Indications for Direct Amalgam Restorations
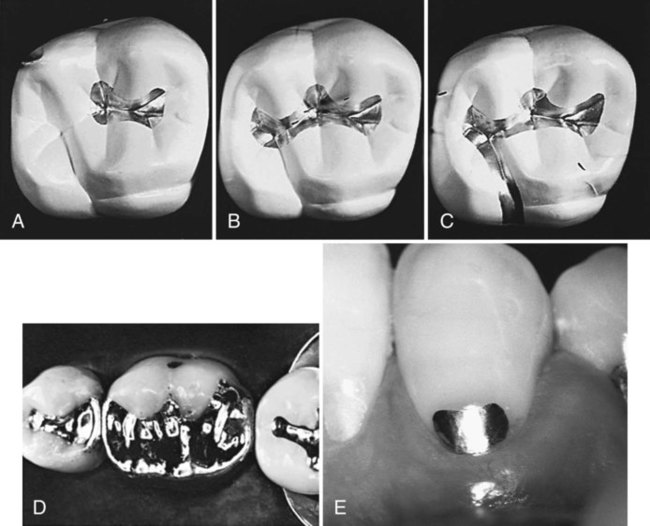
1. Moderate to large Class I and II restorations (especially restorations that involve heavy occlusion, cannot be isolated well, or extend onto the root surface) (see Fig. 13-4, A and B).
2. Class V restorations (including restorations that are not esthetically critical, cannot be well isolated, or are located entirely on the root surface) (see Fig. 13-4, C).
3. Temporary caries-control restorations (including teeth that are badly broken and require a subsequent assessment of pulpal health before a definitive restoration) (see Chapter 2).
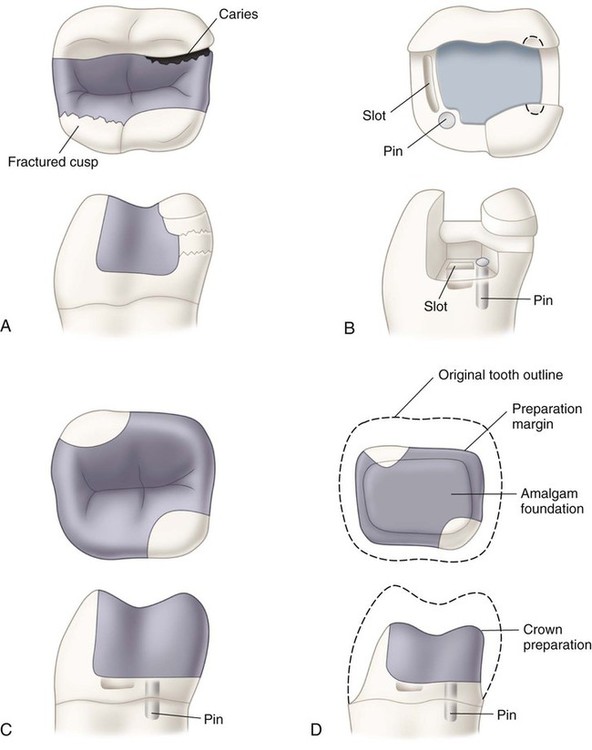
4. Foundations (including for badly broken teeth that require increased retention and resistance forms in anticipation of the subsequent placement of a crown or metallic onlay) (see Fig. 13-5).
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses