11
Oral appliance therapy for sleep-related breathing disorders
CONCEPTUAL OVERVIEW
Oral appliance (OA) therapy has been used for the management of sleep-related breathing disorders (SRBD) since the early 1930s.1 Subsequent investigations into the obstruction of the upper airway led to the study of OA therapy as a nonsurgical treatment option. These early research endeavors identified OA devices that were of two different designs: (1) mandibular repositioning appliance (MRA) that advanced the mandible in a protrusive position and (2) tongue-retaining device (TRD) that held the tongue in an advanced position. Both these OA designs demonstrated promise in reducing the upper airway obstruction.2– 4 Following these early discoveries, there has been a myriad of studies assessing the clinical efficacy and long-term use of OA therapy for the management of SRBD.
The first standards of practice guidelines relative to the use and effectiveness of OA therapy for obstructive sleep apnea (OSA) and snoring were published in 1995 by the American Academy of Sleep Medicine (AASM, formerly the American Sleep Disorders Association).5 Case studies comprised the evidence on which those initial clinical guidelines were based. The AASM guidelines document noted that OA therapy can be considered as a first-line treatment option for the management of mild OSA and simple snoring and also as a second-line treatment option for moderate OSA after unsuccessful attempts with other treatment options. Following the publication of these guidelines, significant research-based findings pertaining to OA therapy have been published.6
In 2006, the AASM published two documents that further recognized OA therapy as a medical device option for the management of OSA and snoring: (1) an evidence-based review of literature regarding OA therapy in sleep medicine6 and (2) a practice parameters update.7 Scientific literature published since 1995 comprised the evidence on which the current practice parameters were based. The updated practice parameters indicate that OA therapy is now an option for patients with mild to moderate sleep apnea and who prefer this method of treatment as opposed to using continuous positive airway pressure (CPAP). In addition, OA therapy may be utilized in patients with severe sleep apnea who (1) are unable to tolerate CPAP, (2) have failed surgery, or (3) are primary (benign) snorers (i.e., snoring without apnea).
OAs have also been reviewed by the United States Food and Drug Administration (FDA), and OAs are regarded as class II medical devices.8 The FDA document states that special controls apply to these devices, and they are deemed to be medical devices appropriate for the treatment of OSA. As such, OAs marketed to the public for the treatment/management of OSA and snoring are required to have a 510(k) or premarket notification clearance in order to be commercially available.
HISTORY OF OAs
In the early part of the 1900s, the first OA that repositioned the mandible was used to address mandibular retrognathia.9 Of interest, this particular device also had a positive secondary effect on the airway. In the 1950s, OAs were utilized mainly to stimulate growth of the mandible for correction of a malocclusion, and they were, for the most part, not recognized for their role in airway improvement during sleep.
In the 1960s and 1970s, research related to the impact of OAs on the growth of the mandible was increasing. During this time, the impact on the airway from OAs designed to reposition the mandible was also being investigated.10 The research demonstrated the impact that airway compromise and obstruction had on growth of the mandible as well as the rest of the craniofacial complex. Accordingly, the resolution of airway compromise along with the resultant improved breathing had the potential to beneficially impact human growth and development.
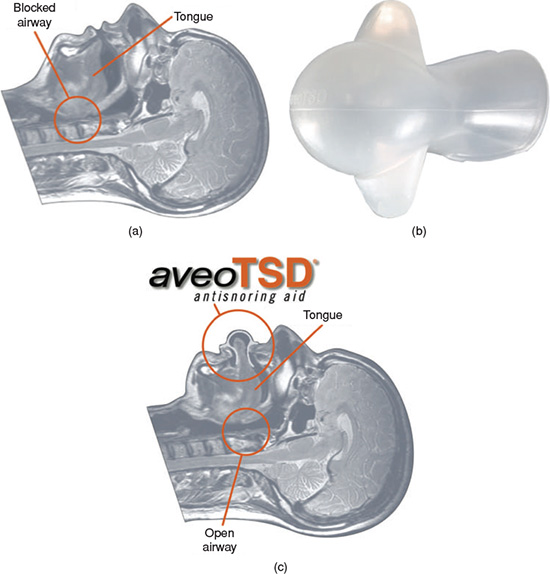
The first available OA that was designed specifically for the management of SRBD was a TRD developed by a physician who was attempting to treat his own sleep breathing issues.11, 12 Fabricated out of a flexible material with a bulb-like receptacle in the anterior portion, the TRD was intended to pull and maintain the tongue in a forward position during sleep so that the tongue would not collapse into the airway against the posterior pharyngeal wall, especially since there can be a tendency for the musculature to exhibit decreased muscle tone during sleep (Figures 11.1a–11.1c).
Other OA designs were introduced in the 1980s and 1990s, all of which were intended to reposition or advance the mandible to improve the upper airway patency. All these OA designs were referred to as anterior repositioning or mandibular repositioning OAs, with the term MRA often used.
Figure 11.1 (a) Sagittal MRI of head: supine position demonstrating blocked airway from base of tongue impinging against posterior pharyngeal airway. (Courtesy of Innovative Health Technologies, 2009. Used with permission.) (b) Sample of a TRD: “aveoTSD”. (Courtesy of Innovative Health Technologies, 2009. Used with permission.) (c) Sagittal MRI of head: supine position demonstrating an opened airway from the use of the “aveoTSD” to maintain an advanced position of the tongue. (Courtesy of Innovative Health Technologies, 2009. Used with permission.)
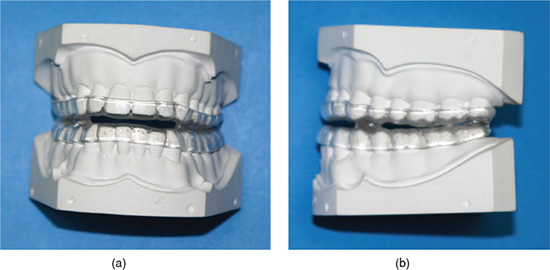
The devices that were introduced in the 1980s were mostly a one-piece design that joined the maxillary and mandibular acrylic components together, and they were referred to as monoblocs (Figures 11.2a and 11.2b). However, this particular design did not allow for any mandibular movement, such as the teeth grinding component of bruxism, and some patients reported discomfort with the masticatory muscles and/or temporomandibular joints (TMJs) after wearing this rigid type of design. In addition, if an alternative maxillomandibular relationship was deemed necessary for therapeutic or patient comfort purposes, it was not possible to modify the MRA without having to either (1) remake the entire OA or (2) separate the two components and reconnect them in a different maxillomandibular relationship with acrylic.
Figure 11.2 (a) Frontal view of monobloc type of MRA design. (b) Sagittal view of monobloc type of MRA design.
From the 1990s to the present time, most devices have been fabricated as separate maxillary and mandibular acrylic-type components that are joined together in a specific fashion that is unique to their respective manufacturer’s mechanism, material, and purpose. Although the means by which the separate components are held together vary by manufacturer, the majority of them (1) account for bruxism by allowing for some degree of mandibular lateral excursions and (2) permit incremental anteroposterior adjustability (titration) for the dentist to easily modify the maxillomandibular relationship for optimum therapeutic effectiveness and patient comfort.
TREATMENT PROTOCOL, OBJECTIVES, AND CONTRAINDICATIONS
Because snoring and OSA are regarded as medical conditions, the dentist and physician need to cooperate with each other regarding the pretreatment assessment and management plan for the patient. The AASM6 and the American Academy of Dental Sleep Medicine13 recommend similar treatment protocols for utilizing OA therapy in the management of OSA and snoring.
Both protocols outline the roles of the dentist and physician:
The AASM documents relative to its practice parameters and evidence-based review of literature lay the scientific foundation for the AASM’s recommendations about the use of OA therapy for the management of OSA or snoring.6, 7 The AASM defined the three levels of recommendations used in those documents as follows:
- Standard: “This is a generally accepted patient-care strategy, which reflects a high degree of clinical certainty.”
- Guideline: “This is a patient-care strategy, which reflects a moderate degree of clinical certainty.”
- Option: “This is a patient-care strategy, which reflects uncertain clinical use.”
When considering the recommendations in the practice parameters, these definitions will serve as references for the dentist when engaging in the management of patients with OSA and/or snoring.
Assessment prior to initiation of treatment
Regarding assessment and diagnosis of SRBD relative to OA therapy, the AASM’s practice parameters made the following recommendation:7
The presence or absence of OSA must be determined before initiating treatment with oral appliances to identify those patients at risk due to complications of sleep apnea and to provide a baseline to establish the effectiveness of subsequent treatment. Detailed diagnostic criteria for OSA are available and include clinical signs, symptoms and the findings identified by polysomnography. The severity of sleep related respiratory problems must be established in order to make an appropriate treatment decision. (Standard)
Treatment objectives
Once the pretreatment assessment and diagnosis have been accomplished and OA therapy has been established as a treatment option for the SRBD patient, then the following treatment objectives for OA therapy in the management of SRBD, specifically OSA and snoring, are recommended per the AASM practice parameters:7
- “For patients with primary snoring without features of OSA or upper–airway resistance syndrome, the treatment objective is to reduce the snoring to a subjectively acceptable level. (Standard)”
- “For patients with OSA, the desired outcome of treatment includes the resolution of the clinical signs and symptoms of OSA and the normalization of the apnea-hypopnea index and oxyhemoglobin saturation. (Standard)”
- “Oral appliances are appropriate for use in patients with primary snoring who do not respond to or are not appropriate candidates for treatment with behavioral measures such as weight loss or sleep-position change. (Guideline)”
- “Although not as efficacious as CPAP, oral appliances are indicated for use in patients with mild to moderate OSA who prefer OAs to CPAP, or who do not respond to CPAP, are not appropriate candidates for CPAP, or who fail treatment attempts with CPAP or treatment with behavioral measures such as weight loss or sleep-position change. (Guideline)”
- “Patients with severe OSA should have an initial trial of nasal CPAP because greater effectiveness has been shown with this intervention than with the use of oral appliances. Upper airway surgery (including tonsillectomy and adenoidectomy, craniofacial operations and tracheostomy) may also supersede use of oral appliances in patients for whom these operations are predicted to be highly effective in treating sleep apnea. (Guideline)”
Dental contraindications
The following dental contraindications for OA therapy in the management of SRBD, specifically OSA and snoring, are identified per the AASM evidence-based review of literature:6
- “Patients need to have an adequate number of healthy teeth (not compromised by periodontal disease) in the upper and lower dental arch to use an MRA.”
- “The patient should have the ability to protrude the mandible forward and open the jaw widely without significant limitation in order to be fitted with an MRA.”
- “Moderate to severe TMJ problems or an inadequate protrusive ability may be contraindications to OA therapy—mild TMJ problems may be lessened by the forward jaw position.”
- “Significant bruxism may be a contraindication to OA therapy.”
- “Patients with full dentures are generally unable to use an MRA but some of these patients may be treated with a TD” (tongue device).
Table 11.1 summarizes the indications and contraindications for OA therapy in the management of SRBD, specifically OSA and snoring.
Even though the presence of severe temporomandibular disorders (TMD) may be overstated, the dentist needs to (1) recognize any preexisting TMD prior to the initiation of OA therapy for SRBD and (2) discern whether the TMD may need to be addressed accordingly. In many instances, the TMD is not an intracapsular disorder of the TMJ itself, but rather more of a disorder involving the soft tissue components of the TMJ and/or the musculature of the head and neck.
Frequently, the masticatory and cervical muscles are tender to palpation, and they may have ability to refer pain to the area of the TMJ (Table 11.2).14 The ability for the musculature to refer pain to a location remote from the trigger points of the involved musculature is known as myofascial pain. Because the pain referral is from soft tissue, it typically can be relieved by exercise, physical medicine, and the use of muscle relaxation medications. In addition, the presence of sleep bruxism needs to be assessed since it can be associated with snoring and OSA, and it may be a precipitating mechanism.15, 16
Table 11.1 Indications and contraindications for oral appliance therapy.
Source: Adapted from Kushida CA, Morgenthaler TI, Littner MR, et al. Practice parameters for the treatment of snoring and obstructive sleep apnea with oral appliances: an update for 2005. Sleep. 2006; 29(2):240–262.
| Indications | Contraindications |
| Treatment of snoring alone (benign snoring) | Poor dental status |
| Patient with mild to moderate obstructive sleep apnea who prefers oral appliance over continuous positive airway pressure (CPAP) or did not respond to CPAP | Inadequate healthy teeth |
| Patient with severe sleep apnea who either failed CPAP or was intolerant to it | Active periodontal disease |
| Inadequate mandibular function or limited range of motion | |
| Severe temporomandibular disorders |
Table 11.2 Myofascial pain and trigger point referral for the most common muscles of the jaw and neck associated with oral appliance therapy.
Source: Adapted from Travell JG and Simons DG. Myofascial Pain and Dysfunction: The Trigger Point Manual. Baltimore: Williams & Wilkins. 1983.
| Muscle | Common site for referral |
| Temporalis | Forehead and over the eyes |
| Maxillary teeth (anterior portion to the anterior teeth, middle portion to the bicuspids, posterior portion to the molars) | |
| Masseter | Ear and around the temporomandibular joint (TMJ) area |
| Supraorbital region | |
| Mandibular posterior teeth | |
| Lateral pterygoid | TMJ area |
| Mid-face and infraorbital area | |
| Medial pterygoid | TMJ and surrounding area |
| Posterior face and mandible | |
| Sternocleidomastoid | Face, supraorbital, back and top of the head, under the mandible |
| Ear, behind the ear, forehead (only muscle that will refer across the midline to the other side of the forehead) | |
| Diagatrics | Anterior teeth |
| Neck, under the mandible at the posterior aspect (may feel like a sore throat) | |
| Posterior cervicals | Face and TMJ area |
| Forehead and the temples | |
| Back of the head |
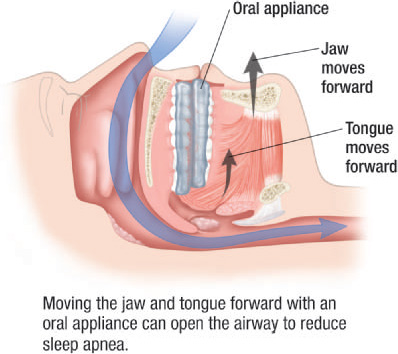
FUNCTION OF MRA THERAPY
For the management of OSA and snoring, the basic function of OA therapy with an MRA design is to reposition the mandible in an increased vertical or open position as well as in an advanced or protrusive position relative to the maxilla. The basic function of a TRD design is to hold the tongue in a more forward position. Although the exact mechanism of the OA on the airway is not fully understood, it is believed that OA therapy improves the patency of the upper airway by increasing the pharyngeal volume or size and/or reducing the collapsibility of the airway through muscle tone improvement.
The upper airway of OSA patients is more narrowed during sleep than those who do not have OSA.17 Studies have shown that anterior repositioning of the mandible and tongue increases the size and volume of the upper airway, typically in the retropalatal and retroglossal areas.18–24 Not all studies, though, demonstrate such airway size increase. One study found that there was no change in the airway size with mandibular advancement.25 Lending to the suggestion that increasing the airway size beneficially affects the severity of OSA are the studies that reflect a reduced apnea–hypopnea index (AHI) with increased mandibular advancement.26–29
Repositioning the mandible also impacts the muscles that support the pharyngeal airway and comprise the tongue. The action of mandibular repositioning may do the following: (1) prevent the tongue from collapsing posteriorly into the oropharyngeal area and (2) stabilize the musculature that supports the airway. A study demonstrated that improvement in the airway is impacted by the palatoglossus muscle on the tongue base when accompanied by mandibular opening.30
The musculature that is related to the tongue and the pharynx may also be beneficially affected by mandibular repositioning. With the mandible advanced, the musculature of the pharynx and the tongue are stabilized, thus preventing these structures from collapsing, narrowing, and obstructing the pharynx during snoring or OSA. Studies on mandibular and tongue advancement with OA therapy found increased muscle tone of the upper airway and decreased genioglossus muscle tone.31, 32
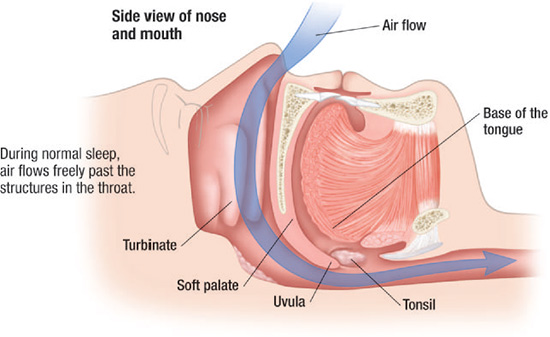
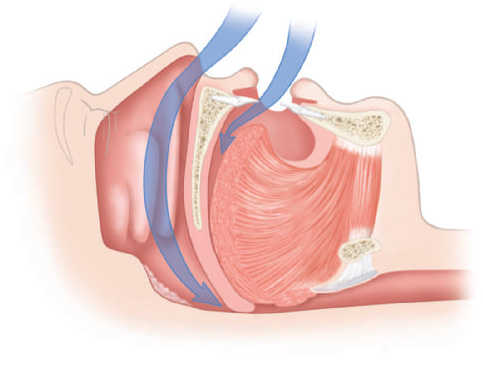
In addition, the repositioning of the mandible also positively impacts the velopharyngeal area and thereby improves the patient’s ability to nose breath.33 It is believed that the MRA’s impact on the palatopharyngeus creates an increased amount of tension on the soft palate, and the repositioning moves the tongue base forward and away from the inferior surface of the soft palate, resulting in increased patency of the nasopharynx for improved breathing (Figures 11.3–11.6).
Figure 11.3 Normal sagittal view of the upper airway. (© Krames Medical Publishing, 2009. Used with permission.)
DESIGN CHARACTERISTICS OF MRAs
For MRAs to be optimally effective and reduce the impact of side effects, there are several design characteristics that should be considered when selecting an MRA design. The selection should be based on (1) specific and related medical and dental conditions of the patient, (2) patient preference, and knowledge, experience, and preferences of the dentist providing the service.
Figure 11.4 Sagittal view of partially blocked upper airway. (© Krames Medical Publishing, 2009. Used with permission.)
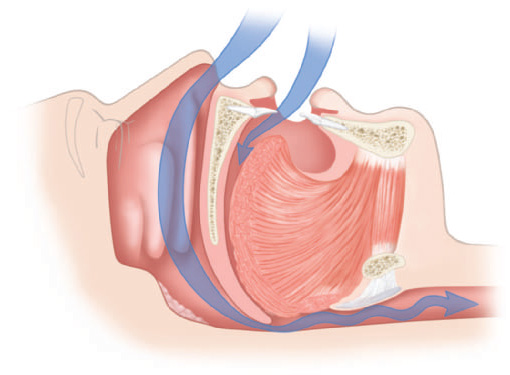
Figure 11.5 Sagittal view of completely blocked upper airway. (© Krames Medical Publishing, 2009. Used with permission.)
The following are suggested characteristics of MRAs for optimum effect in the management of OSA and snoring:
- Adjustable: It (1) is easily adjusted to accommodate changes in the dental status, (2) can be modified to address a new dental restoration, and (3) can be relined to improve retention or the fit.
- Posterior support: It (1) addresses sleep bruxism as well as SRBD and (2) provides adequate support for the TMJs.
- Titration: It incorporates a mechanism that allows incremental positional changes (e.g., anterior, posterior, vertical).
- Full coverage of teeth: It covers all the teeth to prevent tooth movement or unwanted supereruption, and maintains the teeth in the pretreatment position.
- Mandibular mobility: It (1) allows freedom of mandibular movement during sleep bruxism that is also termed as rhythmic masticatory muscle activity, which is commonly found in all patients; (2) improves comfort of the OA during sleep, especially with sleep bruxism; and (3) allows for normal function such as swallowing and licking the lips.
- Lip seal and patent nasal airway: It (1) allows for comfortable lip seal to limit mouth breathing during sleep and (2) improves blood oxygen saturation levels via an open nasal airway, otherwise adjunctive treatment of the nasal airway may be required.
- Maximum tongue space: It affords maximum space for the tongue to prevent further collapse of the tongue into the pharyngeal airway space, especially since encroachment of the OA into the tongue space may cause the tongue to be in a more posterior position in the mouth.
Figure 11.6 Sagittal view of an opened airway from the use of an OA. (© Krames Medical Publishing, 2009. Used with permission.)
EFFECTIVENESS OF OAs
There are a number of studies and literature reviews that have evaluated OAs and their effectiveness, including the AASM evidence-based review of literature which included objective sleep data before and after OA therapy.6 Another literature review document also summarized in 2007 many of the studies published on OAs relative to their effectiveness for the management of SRBD.34 Although the data in these documents support the use of OAs, it continues to be difficult to predict an outcome with OA therapy for any specific SRBD patient. Other than an objective sleep study that includes a polysomnogram, the currently accepted means by which OA effectiveness can be reliably documented through self-reports is with the use of the Epworth Sleepiness Scale (ESS). It has been shown that the ESS score was improved in OA users from 11.2 to 7.8,34 and other self-report assessments also demonstrated significant reductions in ESS scores regarding the efficacy of OA therapy.27,35–37
When using respiratory sleep data to assess the affect of OA therapy on severity of respiratory disturbances for individuals with OSA, the definition of success varied among the studies considered in the AASM review of literature: “the most stringent definition of success was a reduction to less than five respiratory events per hour of sleep while the most liberal definition was a reduction of 50% or more from the baseline AHI.”6
OAs have also been found to be effective in the improvement of cardiovascular disease. Three studies documented improvement in blood pressure measurements with the use of OA therapy.35, 38, 39 One study found that in 161 patients who used an OA for 60 days, the improvement in the blood pressure went from 132.0/82.1 to 127.5/79.2. Another study indicated that the improvement in blo/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


