10
What can Orthodontists Learn from Orthopaedists Engaged in Basic Research?
Summary
The first part of this chapter focuses on the need for an integration of the terminology used within bone biology into the orthodontic world. According to the appropriate biological terminology, bone modelling is defined as uncoupled resorption and formation activities that occur at different sites, while bone remodelling consists of coupled resorption and formation activities occurring on the same site in a specific sequence, i.e. resorption always preceding formation. While bone modelling occurs mainly during growth, bone remodelling occurs throughout life, and plays a role both in the maintenance of the phosphate–calcium homeostasis and in the repair of bone microdamages. The movement of the teeth ‘with’ the alveolar bone obtained with orthodontic appliances is a result of bone modelling rather than bone remodelling, although orthodontists generally refer to this phenomenon as remodelling. Bone modelling can be used by the orthodontist for implant site development, by moving a tooth away from the area where new bone has to be created. The bone remodelling rate in fact influences tooth movement rate. The orthodontists may face situations where the bone metabolic rate has changed (due to disease or pharmacological treatment), or may want to accelerate tooth movement rate by producing local bone damage. The latter case will stimulate bone healing, therefore increasing tooth movement rate. Clinical situations with metabolic impairment are exemplified, and related clinical suggestions are given. In the light of the latest morphological and finite element analysis, the pressure–tension theory is no longer valid in the explanations about bone modelling reactions to orthodontic force. This chapter suggests a new paradigm, founded on the assumption that bone cells are able to react to deformation. Finally, the alveolar bone reaction to skeletal temporary anchorage devices is described, and a potential role for them in bone width maintenance is suggested.
A Common Language
As correctly indicated by the editors of this book, the face is part of a whole body. Therefore, there is no reason to believe that the stomatognathic system would not follow general physiopathological rules applying to the entire body. Basic research carried out by orthopaedists can, therefore, be utilized by orthodontists. However, a precondition for a learning process is a common language.
Modeling
Regarding the biological reaction occurring as a result of orthodontic force systems, there is no lexical consistency between orthodontists and orthopaedists, the former describing it as remodelling, the latter as modelling. In bone-biological terms, bone remodelling and modelling are two distinctly separate processes, with different physiological roles.
Modelling is the term applied to the changes in shape and size of bones (macro-modelling) resulting from resorption and formation activities, and is the dominant biological activity during growth. Modelling also occurs at the trabecular level, the so-called mini-modelling (Frost, 1990). Resorption and formation drifts do not necessarily follow a given sequence, i.e. they are biologically uncoupled. In adult life the physiological mesial drift of the alveolar socket is a result of modelling. Bone modelling reflects the structural adaptation of bone mass and shape to varying demands. It is thus largely controlled by functional and applied loads, and less by systemic metabolic requests. Formation drifts may occur in both lamellar and woven bone. Woven bone is a result of larger strain values than lamellar bone, and usually appears after fracture healing, in response to neoplasms, infections, and in reaction to large mechanical loads produced by orthodontic appliances (Frost, 1994) (Figure 10.1).
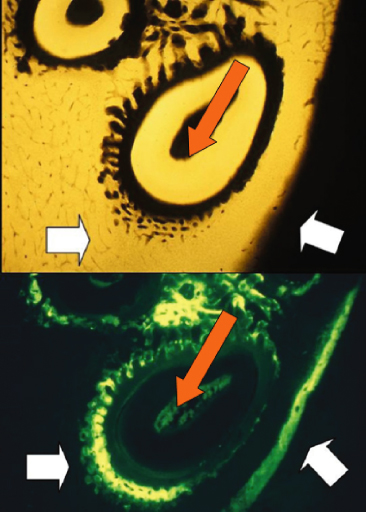
Figure 10.1 Alveolar bone modelling following the application of 25 g of force. After 14 days of treatment, the microradiographic analysis (top) shows new bone formation ahead of the tooth in the direction of the force (red arrow). On the buccal side, new periosteal bone formation is observed, as shown by the wide strips of bone markers (white arrow).
(From Verna et al. (1999b) with permission from Elsevier.)
Clinical Implications of Modelling
When teeth are moved ‘with bone’, modelling of the alveolar bone occurs, and thus the teeth can be moved beyond the immediate alveolar envelope while maintaining the marginal bone level. This principle underpins the treatment of partially dentate adult patients with atrophied alveolar bone areas (Mantzikos and Shamus, 1999; Chandler and Rongey, 2005). An example of the so-called tooth movement with bone is illustrated in Figure 10.2, where the tooth has been moved into an atrophic edentulous area, with ‘rebuilding’ of the alveolar process, giving the impression that the tooth has carried the alveolus along to the new position.
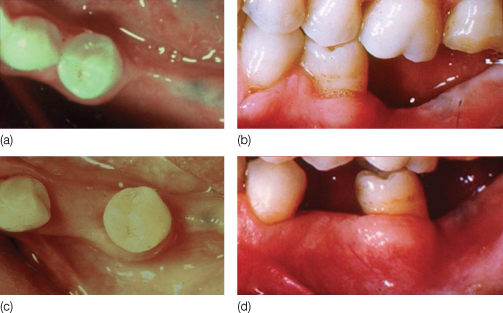
Figure 10.2 (a,b) Tooth movement with bone in an edentulous alveolar crest, where no bone is present. The alveolar drift allows for the movement of the whole alveolus (c,d), and the osteogenic potential of the periodontal ligament is fully expressed in the formation of bone.
(From Melsen B, Garbo D [2004] with permission from Quintessence.)
When determining the force system necessary for a planned tooth movement, the quantity and the quality of the bone surrounding the tooth should be taken into consideration. High-resolution micro-computed tomography (CT) scans of alveolar bone samples show that the bone surrounding the roots is highly variable in density and that the so-called lamina dura is a non-uniform structure (Dalstra et al., 2006) (Figure 10.3). Therefore, even very light forces may result in overcompression over small surfaces of the alveolar bone. This is in line with the finding that hyalinization is rarely avoidable (von Bohl et al., 2004), although the amount may vary.
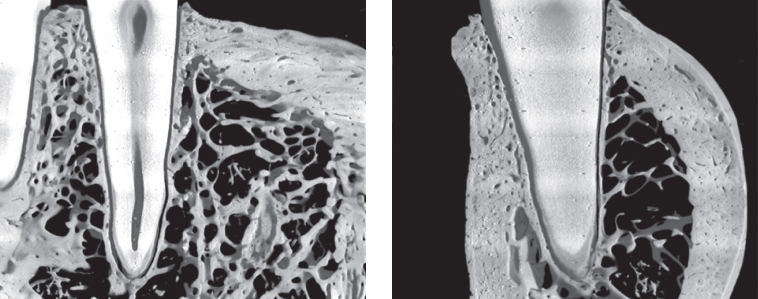
Figure 10.3 Mesiodistal (left) and buccolingual (right) views of the alveolus of the canine. Note the uneven thickness of the alveolar bone proper and the sparse trabecular support.
(From Dalstra et al. [2006] with permission from Blackwell Munskgaard.)
Particular attention should be paid to the origin of the cells that will activate the modelling process. In the case of indirect resorption in areas dominated by dense cortical bone, for example in labial movement of lower incisors, tooth movement requires modelling of the cortical bone, and the osteoclasts should originate from the cortical bone, or from the periosteum. Labial tipping of incisors in the presence of thin cortical bone increases the probability of bone fenestration (Baron, 1975). To reduce the risk of iatrogenic dehiscence, the initial force system should be directed at the displacement of the apex of the root, which is usually surrounded by trabecular bone. Thereafter, the bending of the alveolar process will generate formation on the buccal side of the alveolar process. The use of controlled, uniformly distributed, low-magnitude forces is recommended, and, since the modelling rate of cortical bone is slower than that of trabecular bone, adequate time should be allowed for initiation of cellular activation and subsequent bone formation at the periosteal side. An example is shown in Figure 10.4, where the system was checked monthly to adjust the line of action of the force without activation.
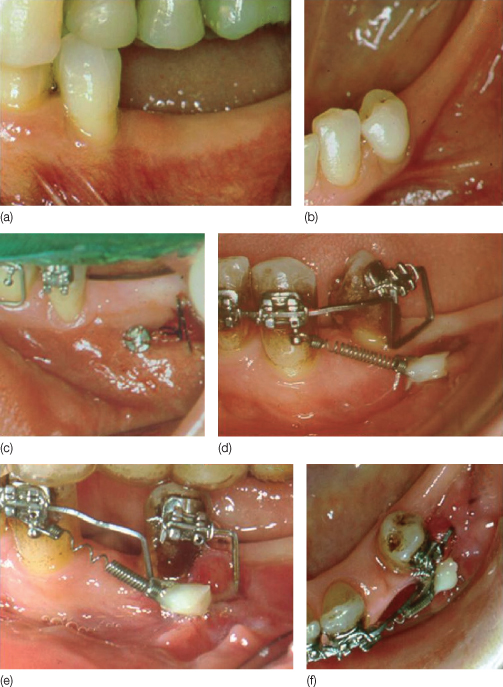
Figure 10.4 (a,b) Modelling of the alveolar bone achieved by bodily movement of a premolar in an atrophic area. (c,d) Following the insertion of a mini-implant (Aarhus System) the biomechanical system consisted of a box loop in a titanium molybdenum in a .017 × .025 inch archwire activated to apply a distal force and uprighting moment. This enabled a bodily tooth movement, with uniform distribution of the force in the periodontal ligament. Anchorage was ensured by the use of a 50 g superelastic coil spring attached to the skeletal anchorage device. (e,f) The same biomechanical system was used to achieve the final displacement, and at each appointment the appliance was adjusted to maintain the force-to-moment ratio for continuation of the translation movement.
(From Melsen B, Garbo D [2004] with permission from Quintessence.)
In sum, the drift of the alveolar bone occurring following the application of an orthodontic force can be described as modelling, in response to a modified functional demand. Since the anatomy of the teeth differs both within an individual dental arch and between patients, modelling of the alveolar bone without bone loss can only be achieved by use of customized biomechanical systems. However, based on this premise, it is difficult, if not impossible, to agree on absolute standardization of treatments.
Remodeling
As mentioned above, orthodontists do not distinguish between modelling and remodelling. Remodelling is a phenomenon that occurs throughout life in discrete locations in the whole skeleton. It involves cycles of resorption and subsequent formation, spatially and temporally coupled. Remodelling allows bone adaptation to mechanical stresses by minimizing fatigue damages, and is influenced by the action of hormones and cytokines (Burr, 1993; Mori and Burr, 1993; Frost, 1998).
The bone remodelling cycle is carried out by a team of cells called the bone multicellular unit (BMU) (Frost, 1986). The cycle in humans lasts 120–160 days, and is composed of four phases: activation, resorption, reversal, and formation (Eriksen et al., 1984). Although the stimulus necessary for the initiation of a new BMU is still not fully identified, it has been shown that microcracks can initiate remodelling via the sensory input to the osteocytes. In the activation phase, the lining cells, the preosteoblasts, change to a cuboidal shape, and secrete a receptor ligand, RANKL (receptor activator of nuclear factor kappa B ligand), on the cell surface. The preosteoclasts within the bone marrow present receptors for RANKL on their cell membrane, called RANK. The interaction between RANK and RANKL determines the activation of the preosteoclasts, which then fuse and differentiate into mature multinucleated osteoclasts and resorb bone. The resorption of bone also occurs as a result of inhibitory activities directed towards osteoblasts, as in the case of the release of sclerostin (SOST), which inhibits bone formation and enhances osteoblast apoptosis (Winkler et al., 2003). The RANKL present on the cell surface of the preosteoblast is neutralized by a free-floating decoy receptor belonging to the tumour necrosis factor (TNF) family, osteoprotegerin (OPG), thus controlling further activation of the pre-osteoclasts via RANK. Bone resorption lasts for about 2 weeks, after which the osteoclasts undergo programmed cell death or apoptosis.
Following resorption by the osteoclasts, the lacuna is deepened by mononuclear phagocyte cells (Eriksen et al., 1984). At the end of this process the reversal phase is initiated, and further preosteoblasts migrate into the resorbed cavity and differentiate into osteoblasts. The interface between the resorbed old bone and the new-formed bone is called the cement line, or reversal line, and these osteoblasts now start to form bone. The precursor preosteoblasts are probably attracted by bone-derived growth factors (such as transforming growth factor-β, TGF-β) and are derived from marrow stromal cells. Through a positive feedback mechanism the preosteoblasts also secrete growth factors, osteopontin, osteocalcin, and other proteins, such as insulin growth factor-1 (IGF-1), interleukin-6 (IL-6), bone morphogenetic proteins (BMPs), and fibroblast growth factors (FGF), all of which enhance the osteoblast’s activities. Fibroblasts also migrate towards the cement line and secrete the thin collagen fibrils around which the osteoblasts start synthesizing osteoid. The osteoid is non-mineralized bone matrix that cannot be resorbed by osteoclasts. The mineralization process starts when the osteoid thickness reaches about 6 µm. After about 4 months the resorption cavity is filled with new bone, although not completely, with closely packed mineral crystals and subsequent increase in bone density.
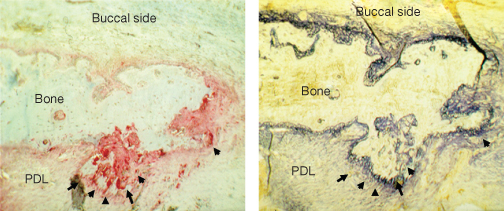
It is essential for the orthodontist to understand the role of remodelling following the application of an orthodontic load. Bone formation has been observed not only in the periodontal ligament (PDL) tension side, but also in the direction of the force (Mohri et al., 1991; King et al., 1992); bone resorption has also been observed at this site (Mohri et al., 1991; Zaffe and Verna, 1995; Melsen, 1999) (Figure 10.5). The presence of markers of both resorption and formation adjacent to the alveolar wall reflects the coupled activity of osteoclasts and osteoblasts that characterizes what orthopaedists describe as bone remodelling. Synchronized initiation of numerous BMUs results in bone turnover on both the pressure and tension sides in the PDL (Tanne et al., 1990). The RANKL-RANK mechanism has been shown to be active in the PDL and is strengthened by mechanical load (Ogasawara et al., 2004). Tooth movement can be accelerated by local delivery of RANKL (Kanzaki et al., 2006), whereas the delivery of OPG decreases the rate of tooth movement (Oshiro et al., 2002; Kanzaki et al., 2004). A lack of OPG has been found to be associated with a significantly higher level of bone resorption in the compression area, whereas the upregulation of RANKL induced by orthodontic forces depends on the presence of prostaglandin (PG) E2, which is released by osteoblasts and osteocytes (Kanzaki et al., 2002; Yamaguchi, 2009).
Figure 10.5 Alveolar bone on the buccal side of a rat’s tooth after 14 days of orthodontic treatment. On the side facing the periodontal ligament (PDL; arrows), there is staining for alkaline phosphatase (dark blue, right panel) and for acid phosphatase (red, left panel), confirming bone remodelling activities.
(From Verna and Melsen [2002] with permission from Libra Ortodonzia.)
It is clear, therefore, that the application of a mechanical load initiates remodelling as well as bone modelling. The variation in the quality of remodelling between patients may explain the individual variations in tissue response to orthodontic treatment.
Clinical Implications of Bone Remodelling
Cortical anchorage was defined by Ricketts as movement of the roots towards the cortex of the alveolar bone to resist the active force of the appliance (Ricketts, 1998). The biological principle behind cortical anchorage is the utilization of the differential turnover rates of cortical and trabecular bone, the former being 5–10 times slower than the latter (Steineche and Hauge, 2003). Teeth closer to the cortical bone need more time to be moved, but at a later stage anchorage loss will occur. Absolute anchorage can, however, only be achieved with ankylosed teeth or skeletal anchorage devices (Melsen and Costa, 2000; Melsen and Verna, 2000).
The biological reaction to an orthodontic force system interacts with the already occurring physiological bone turnover. Our knowledge of turnover rate of the bone in the alveolar process is, however, scarce. The sole human histomorphometric study comparing static cortical remodelling in the mandible with that of the iliac crest, which is the most frequent site for bone biopsies, showed that the levels of remodelling parameters were lower in the basal part of the mandible than in bone samples harvested from load-bearing areas, confirming that remodelling is maintained by function (Verna et al., 1999a). Recently, the site-specificity of bone remodelling has been confirmed by Deguchi et al. (2008), who demonstrated faster bone remodelling and faster tooth movement in a dog’s maxilla than in the mandible. This is due to not only a different ratio between cortical and trabecular bone in the two anatomical sites, but also differences in remodelling rates. At the clinical level, it is advisable to start the orthodontic treatment in the lower jaw to avoid unnecessary prolongation of treatment time.
The rate of tooth movement is influenced by the rate of bone remodelling. Any substance able to interfere with bone remodelling may, in principle, influence the rate of tooth movement rate. Different substances have been tried experimentally and clinically to increase/decrease the rate of tooth movement, but their use is not recommended in the routine clinical setting. On the other hand, as clinicians, we need to know whether substances that may interfere with orthodontic treatment are being taken by our patients. Non-steroidal anti-inflammatory drugs, vitamin D, estrogen, and doxycycline are known to interfere with bone remodelling and to decrease the tooth movement rate (Krishnan and Davidovitch, 2006; Bartzela et al., 2009). However, acetaminophen (paracetamol) is an analgesic that does not interfere with tooth movement (Arias and Marquez-Orozco, 2006). Patients should therefore be instructed preferably to take acetaminophen for relief of pain, so that overall treatment time remains unaffected.
Corticosteroid treatment has an inhibitory effect on bone turnover that subsequently leads to secondary hyperparathyroidism. The effect on tooth movement will therefore depend on the duration of the drug treatment, and is enhanced in patients undergoing long-term corticosteroid treatment (Kalia et al., 2004). In the case where treatment is of short duration, the rate of tooth movement may not to be affected, but, at a tissue level, the remodelling process seems to be delayed. In clinical terms, if a patient is under active orthodontic treatment and has to undergo short-term steroid treatment, such as hay fever therapy, minimal adjustments should be made to the appliance during this period and the interval between appointments may be longer. In treatments of longer duration (about 12 months), the tooth movement rate is increased and the appliance should be checked as usual or more frequently. The effect of steroid treatment on root resorption has also been investigated, with higher rates of root resorption reported in rats administered steroids for a short amount of time (Verna et al., 2006). This is not surprising, since the slower turnover rate will lead to relatively larger amounts of bone covered by osteoid. Since osteoid cannot be resorbed by osteoclasts, the clastic activities are diverted towards the mineralized root surfaces. Once again, in such situations, activation of the appliance will not help. On the contrary, less frequent activation will protect against further root damage. In patients with a history of allergy, who are undergoing steroid treatment for a short time, it is advisable to postpone treatment until the patient has completed their pharmacological treatment and to follow the above-mentioned suggestions in case of a sudden need for steroids during the orthodontic treatment. The effect of corticosteroid treatment on tooth movement is also dose dependent, since high dosages induce osteoporosis, which means there is less bone available for resorption, and therefore the rate of tooth movement increases but with a higher degree of relapse (Ashcraft et al., 1992).
Tooth movement can also be accelerated by local mechanisms that increase the remodelling rate without use of drugs, such as the regional acceleratory phenomenon (RAP) that occurs after fracture healing, as described by Frost (Frost, 1994; Roblee et al., 2009). RAP is characterized by local increased resorption and formation activities with less mineralized bone formation, which is a sign of quick repair after an injury. RAP normally occurs following orthodontic tooth movement (Verna et al., 1999b) and its intensity is related to the intensity of the damage. Cortical perforations and corticotomies (with or without surgical flap) performed as part of the orthodontic treatment increase the tooth movement rate approximately twofold (Sanjideh et al., 2010) by enhancing the repair process locally, for example in tooth movement immediately following extraction.
In animal experiments the tissue reaction occurring during corticotomy-assisted tooth movement is characterized by transient bone resorption, followed by the deposition of fibrous tissue after 21 days and by bone after 60 days (Wang et al., 2009). After corticision (a transmucosal corticotomy technique) less hyalinization and more rapid removal of hyalinized tissue has been observed, together with extensive areas of resorption followed by a 3.5-fold higher accumulated mean apposition area of new bone than the control side (Kim et al., 2009). When cortical perforation is combined with alveolar augmentation, the original alveolar volume and any existing osseous dehiscences may no longer be a limitation for orthodontic tooth movement (Wilcko et al., 2009). Since bone density is decreased, the resulting dissipation of the physiological as well as orthodontic forces over the bone surface and the more effective blood supply may reduce root resorption risk. However, to date, no histological studies have analyzed the root status in areas with more relative osteoid formation, such as in the enhanced bone formation phase of RAP. Futur/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


