10 Oral Complications Associated with Cancer Therapy
10.0 INTRODUCTION
Treatment for cancer may result in short- and long-term changes that can adversely affect oral health, general health, and quality of life. Malignancies with the greatest potential to impact the oral cavity include head and neck cancers, hematologic cancers, and cancers that have metastasized to bone; however, almost any cancer, depending on the location and treatment, can potentially cause adverse oral effects. Oral complications may develop from direct damage to oral tissues as a result of surgery and/or radiation therapy (RT), as well as indirect damage due to regional or systemic toxicity associated with cytoreductive chemotherapy (CT), immunotherapy, and/or targeted molecular therapies. Even supportive care in cancer therapy can cause oral complications, such as hyposalivation due to use of opioid analgesics and medication-associated osteonecrosis of the jaw. Oral infections are common when salivary gland and/or immune function are compromised by cancer therapy and can exacerbate therapy-related toxicity to oral structures. With recent advances in the understanding of the basic biology of cancer and the increasing use of targeted molecular therapies, new oral complications are emerging that were not previously encountered in oncology; for example, aphthous-like stomatitis secondary to mammalian target of rapamycin (mTOR) inhibitors.
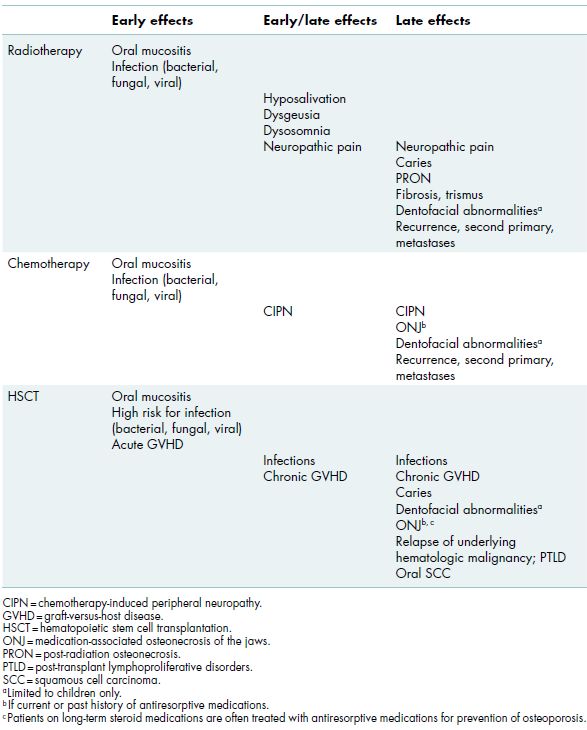
Oral complications can be characterized as acute or chronic (Table 10.1). Acute or early effects occur during the course of therapy and are primarily due to direct tissue toxicity. Acute reactions are generally self-limiting and typically resolve over weeks to months following the completion of therapy. Oral mucositis is a serious acute complication in which lesions of the oropharynx often precede the oral cavity. Mucositis may cluster with full-spectrum gastrointestinal toxicities to include nausea, vomiting, and diarrhea. Chronic or late effects occur months to years after completion of therapy, often when patients are in long-term remission from their primary cancer, and result in permanent tissue damage and lifelong morbidity that have a profound impact on overall health and quality of life. Oral health care providers play a key role in the prevention, identification, and management of such complications in cancer patients.
Table 10.1 Early and late effects of cancer treatment.
10.1 Cancer pre-treatment risk assessment
Prior to initiation of cancer therapy, the oral cavity should be carefully assessed and appropriate interventions should be provided to reduce the severity of oral complications during and after cancer treatment (Table 10.2). First, it is important to establish a rapport with the patient’s oncology team and to have an understanding of the planned cancer treatment and potential complications. The most common cancers in the United States are solid tumors, such as breast, lung, prostate, and/or colon cancers; these patients are generally considered to be at low risk for developing significant oral complications and are not typically evaluated prior to initiation of cancer treatment. Regardless, if CT is planned, individuals are at risk for the systemic toxic effects of various regimens. For these patients, potential causes of acute infections (Table 10.3) should be eliminated prior to treatment and patients should be evaluated during treatment if oral complications arise.
Table 10.2 Risk assessment prior to cancer treatment.
| Complication | Procedures | Clinical Implications |
| Infection/bleeding following dental procedures | Evaluate laboratory values Consult with oncologist prior to performing dental treatment |
Identify need for platelet transfusion or localized hemostatic measures Determine need for antibiotics Modification of treatment plan or timing of care |
| Infection during cancer treatment | Extraction of hopeless/non-restorable teeth Endodontic therapy/caries control Periodontal treatment |
Eliminate/stabilize oral disease Anticipated periods of neutropenia |
| Mucosal injury | Smooth sharp edges of teeth and restorations Adjust ill-fitting prostheses |
Impaired healing of soft tissues Increased risk of oral mucositis |
| Increased caries risk | Fabrication of fluoride trays and prescription-strength fluoride Oral hygiene instruction |
Potential for salivary hypofunction Increased caries risk |
Table 10.3 Potential causes of acute infection during cancer treatment.
| –Dental caries extending to or approximating the pulp –Active periodontal disease –Symptomatic tooth –Retained root –Periapical pathology –Recurrent episodes of pericoronitis |
Individuals at a higher risk of developing early and/or late oral complications of cancer treatment include the following: (a) patients with head and neck cancers, including squamous cell carcinoma, salivary gland tumors, and extranodal (intraoral) lymphoma, (b) patients with hematologic malignancies, including leukemia, lymphoma, and multiple myeloma, and (c) patients treated with high-potency antiresorptive medications due to metastatic disease to the bone. In all of these high-risk patients, a comprehensive oral evaluation focused on minimizing the risk of oral complications should be completed. Necessary dental treatment should be delivered expeditiously, while providing adequate healing time for any surgical procedure, so that initiation of cancer treatment is not delayed. During the pre-treatment phase, the goal is to prevent acute infections or exacerbation of existing chronic oral infections during cancer treatment and to minimize the risk of complications arising during and post-therapy when patients may have complicated courses or remain immunosuppressed for extended periods.
10.1.1 Head and neck cancers
Patients undergoing treatment for head and neck cancer are typically treated with single modality therapy (e.g., surgery) or a combination of surgery, RT, and/or CT, all of which often contribute to the development of significant short- and long-term oral complications. Standard RT provides between 50 and 70 Gy (Gray unit—absorbed dose of radiation) cumulative dose, delivered in daily doses (fractionations) over 4–8 weeks. Patients who undergo RT to the head and neck have risks of long-term reduced healing capacity, tissue fibrosis, and salivary hypofunction due to permanent tissue damage. To improve tumor control and the risk of long-term treatment effects, advanced radiotherapy modalities and treatment regimens are being used for certain head and neck cancers. Intensity-modulated radiation therapy (IMRT) utilizes a three-dimensional dose distribution that spares normal tissues and reduces the volume of tissue exposed to radiation, thus reducing the risk for tissue toxicity. Other advanced treatment regimens for specific head and neck cancers (such as salivary gland) offer smaller and/or more frequent doses over a shorter period of time and have the capacity to improve tumor control while increasing the risk and severity of early and late RT effects.Hyperfractionation, the use of multiple smaller dose fractions, makes it possible to increase the total dose and increase the probability of tumor control. Accelerated fractionation typically involves twice daily dosing, which shortens the overall treatment time and minimizes the risk of tumor repopulation during treatment.
CT regimens are often administered concurrently with RT (combined modality chemotherapy) and have systemic toxic effects that may result in periods of immunosuppression. In some circumstances, neoadjuvant CT may be utilized initially to shrink the primary tumor prior to delivering other treatment, such as surgery or RT.
The pre-treatment phase should take into consideration the risk of acute complications during treatment and the long-term risk of cancer treatment upon orofacial structures. Acute oral infections during cancer treatment can be difficult to manage, may disrupt therapy, and in patients who are at risk for developing neutropenia, more serious infection and sequelae may ensue. Due to permanent diminished bony healing capacity caused by RT, teeth with poor prognosis resulting from caries and/or periodontal disease in the RT field should be extracted, ideally 2–3 weeks prior to the start of RT to allow for appropriate bony healing (Alert Box 10.1). Untreated caries and periodontal disease should also be managed during this time to eliminate any potential sources of acute infection and caries progression during treatment and prevent the need for future surgery in radiated sites. Further, potential causes of oral trauma such as sharp tooth edges or ill-fitting dental prostheses should be adjusted to prevent mucosal injury during treatment, and long-term daily utilization of fluoride delivered in custom trays is an important preventive strategy to minimize radiation-associated dental caries.
Dentoalveolar surgery and/or extraction of teeth with poor prognosis 2–3 weeks prior to initiation of therapy
Consider alternative treatment to prevent medication-associated ONJ
Dentoalveolar surgery and/or extraction of teeth with poor prognosis in planned RT field2–3 weeks prior to initiation of therapy
Consider alternative treatment to prevent PRON if planned treatment is in RT field
Consider HBO treatment prior to and after surgical procedure if planned treatment is in RT field
10.1.2 Hematologic malignancies
Patients with hematologic malignancies often have profoundly compromised immune systems prior to initiation of therapy, are typically treated with multiple courses of high-dose chemotherapy resulting in extended periods of myelosuppression/immunosuppression, and in some cases may be considered for hematopoietic stem cell transplantation (HSCT).
Chemotherapy regimens may involve induction therapy, in which combined agent chemotherapy is utilized to rapidly obliterate most of the tumor cells to cause the cancer to go into remission, followed by consolidation chemotherapy, when high doses of chemotherapeutics serve to further reduce tumor burden. In autologous HSCT, the patient’s stem cells are collected prior to intensive myeloablative conditioning (with or without total body irradiation) and then reinfused as a “stem cell rescue” after chemotherapy. Allogeneic HSCT involves either myeloablative or reduced-intensity conditioning followed by infusion of stem cells from a human leukocyte antigen (HLA)-matched donor, which results in restoration of hematopoiesis and immune function, and long-term anticancer therapy through the graft-versus-tumor effect. Intensive chemotherapy regimens place patients at high risk for infection and bleeding (see chapters 3 and 4), and patients undergoing allogeneic HSCT are at significant risk for developing oral chronic graft-versus-host disease and its many sequalae.
Patients scheduled for HSCT should have potential sources of acute infection removed at least 1 week prior to the start of induction CT to allow for soft tissue healing and to assess for resolution of symptoms. This includes extractions of hopeless teeth, endodontic therapy, caries control, periodontal treatment, and dental prophylaxis. Patients may be at risk for infection and/or bleeding following dental procedures secondary to neutropenia and thrombocytopenia, especially surgical procedures or dental scaling. A thorough evaluation of laboratory values, including platelet and absolute neutrophil counts, must be obtained prior to performing dental treatment, and precautions should be taken if necessary (see chapters 3 and 4). Finally, patients should be instructed on meticulous oral hygiene practices and educated about the potential early and late oral effects of cancer treatment.
10.1.3 Antiresorptive therapy
Patients with multiple myeloma and solid cancers that have metastasized to bone may be treated with intravenous (IV) aminobisphosphonate or high-dose denosumab (a receptor activator of nuclear factor kappa-B ligand [RANKL, a potent mediator of bone resorption] inhibitor) therapy to prevent cancer growth and reduce the risk of developing skeletal-related events, such as bone fractures and spinal cord compression. These antiresorptive medications are powerful inhibitors of osteoclastic bone resorption. An untoward side effect of these otherwise very effective treatments is an increased risk of medication-associated osteonecrosis of the jaw (ONJ; section 10.4.4).
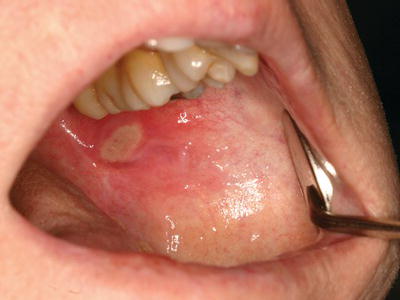
Figure 10.1 Aphthous-like lesion on the left buccal mucosa of a patient on mTOR inhibitor therapy (targeted molecular cancer therapy) for sarcoma management.
Prior to initiation of IV bisphosphonate or high-dose denosumab therapy, non-restorable and hopeless teeth should be extracted and necessary elective dentoalveolar surgery should be completed at least 2–3 weeks prior to treatment to allow for adequate soft tissue and osseous healing (Alert Box 10.1). Further, potential causes of oral trauma such as sharp tooth edges or ill-fitting dental prostheses should be adjusted to prevent mucosal injury. Dental prophylaxes, caries control, and conservative restorative dentistry can be and should be performed while on therapy. For dental management of patients on antiresorptive medications, see section 10.4.4.
10.2 Early effects of cancer treatment
10.2.1 Surgical procedures
Acute pain is expected secondary to surgical procedures for head and neck cancer. Surgery-related pain usually involves acute inflammatory responses related to the extent of the surgery and may be associated with a variable degree of concomitant nerve injury. Post-surgical pain is usually self-limiting and is managed with analgesics.
10.2.2 Oral mucositis
Oral mucositis is a common acute complication of RT to the head and neck and/or CT. It is a painful and debilitating condition that often results in limited oral intake and weight loss and has a profound effect on quality of life. The increasing use of more aggressive therapy to improve cancer cure rates has increased the frequency, severity, and duration of oral mucositis. In addition, newer anticancer therapies, such as the mTOR inhibitors, are associated with new forms of mucositis that present differently from classic CT- or RT-induced lesions (Figure 10.1).
Oral mucositis generally manifests as erythema followed by ulceration of mucosal tissue (Figure 10.2). Diagnosis is made through clinical examination, and routine assessment of the oral cavity during cancer treatment permits early identification of lesions. In an effort to standardize measurements of mucosal integrity, oral assessment scales have been developed to grade the severity and extent of mucositis. The most frequently utilized scales are the World Health Organization Oral Mucositis Scale, which combines subjective and objective measures of oral mucositis (Figure 10.3), and the National Cancer Institute (NCI) Common Terminology Criteria for Adverse Events (CTCAE) version 4.0, a subjective scale used by oncologists in clinical practice to grade treatment-related toxicity (Figure 10.4).
Figure 10.2 Severe mucositis of the left lateral tongue in a patient undergoing chemotherapy and radiotherapy for squamous cell carcinoma of the left lateral tongue.
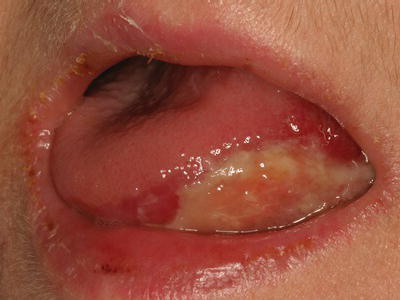
Figure 10.3 World Health Organization Oral Toxicity Scale.
Reference: Sonis ST, Filers JP, Epstein JB, et al. Validation of a new scoring system for the assessment of clinical trial research of oral mucositis induced by radiation or chemotherapy. Mucositis Study Group. Cancer 1999;85(10):/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


