Cysts of the Jaws and Neck
Odontogenic Cysts
Periapical (Radicular) Cyst
Etiology and Pathogenesis
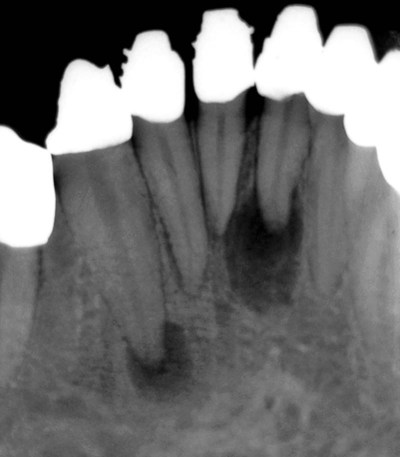
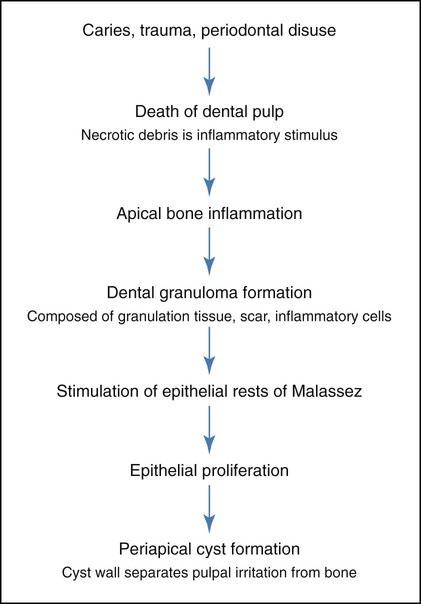
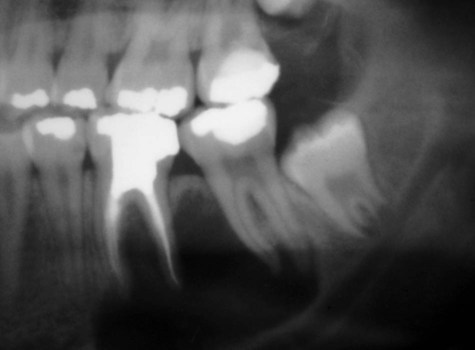
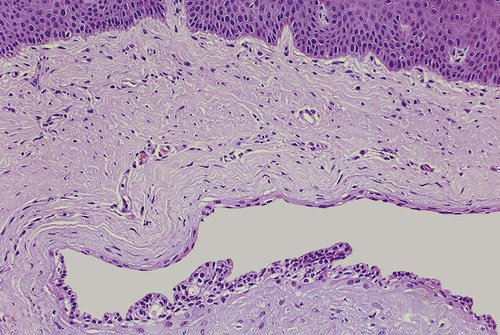
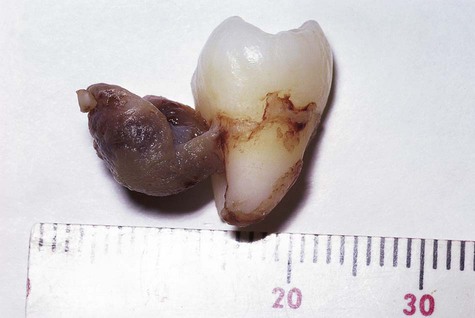
A periapical cyst develops from a preexisting periapical granuloma, which is a focus of chronically inflamed granulation tissue located at the apex of a nonvital tooth (Figures 10-1 and 10-2). Periapical granulomas are initiated and maintained by the degradation products of necrotic pulp tissue. Stimulation of the resident epithelial rests of Malassez occurs in response to the products of inflammation (Table 10-1). Cyst formation occurs as a result of epithelial proliferation, which helps to separate the inflammatory stimulus (necrotic pulp) from the surrounding bone (Figure 10-3).
TABLE 10-1
CYSTS OF THE JAWS: EPITHELIAL ORIGIN
| Type | Source | Origin of Rests | Cyst Examples |
| Odontogenic rests | Rests of Malassez | Epithelial root sheath | Periapical (radicular) cyst |
| Reduced enamel epithelium | Enamel organ | Dentigerous cyst | |
| Rests of dental lamina (rests of Serres) | Epithelial connection between mucosa and enamel organ | Odontogenic keratocyst | |
| Lateral periodontal cyst | |||
| Gingival cyst of adult | |||
| Gingival cyst of newborn | |||
| Glandular odontogenic cyst | |||
| Nonodontogenic rests | Remnants of nasopalatine duct | Paired nasopalatine ducts (vestigial) | Nasopalatine canal cyst |
Clinical Features
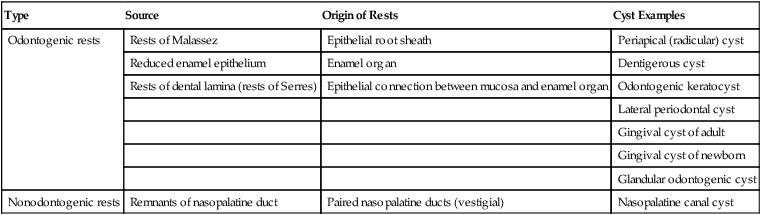
Periapical cysts constitute approximately one half to three fourths of all cysts in the jaws (Box 10-1). The age distribution peaks in the third through sixth decades. Of note is the relative rarity of periapical cysts in the first decade, even though caries and nonvital teeth are rather common in this age group. Most cysts are located in the maxilla, especially the anterior region, followed by the maxillary posterior region, the mandibular posterior region, and finally the mandibular anterior region.
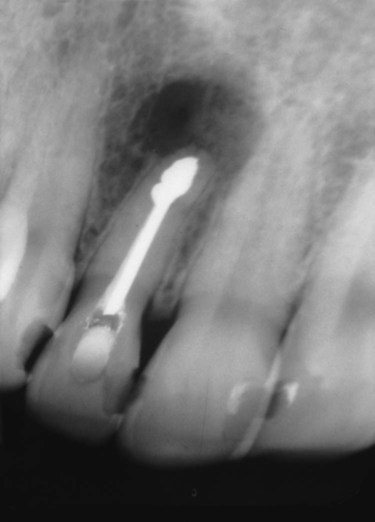
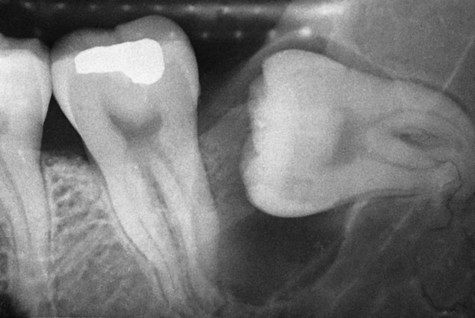
Periapical cysts usually are asymptomatic and often are discovered incidentally during routine dental radiographic examination (Figures 10-4 and 10-5). They cause bone resorption but generally do not produce bone expansion. By definition, a nonvital tooth is necessary for the diagnosis of a periapical cyst.
Histopathology
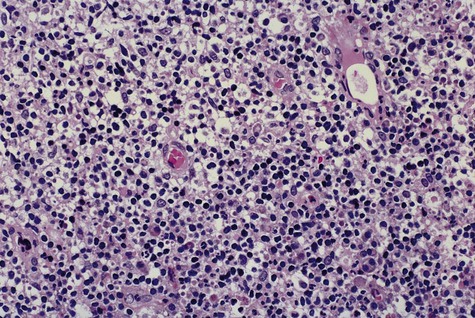
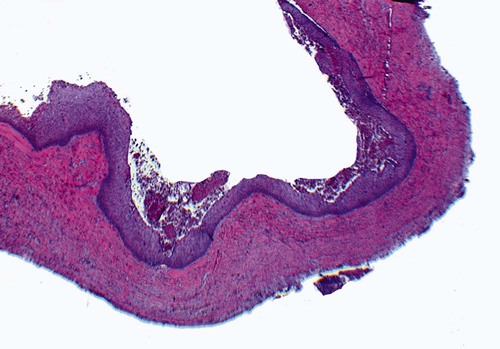
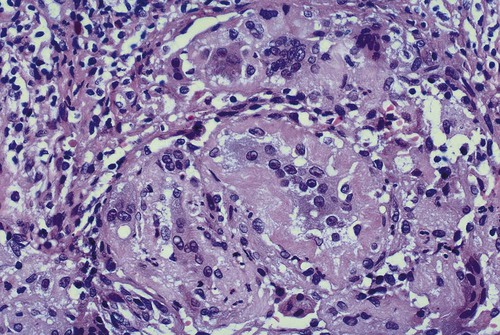
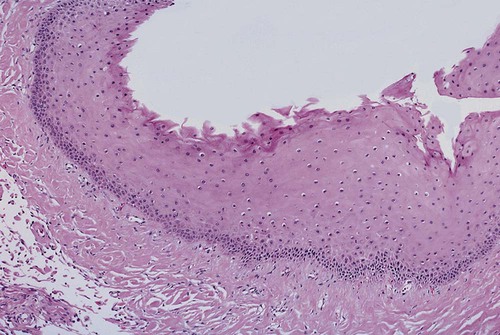
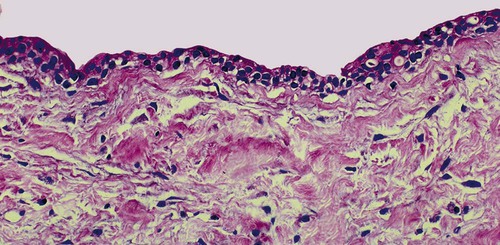
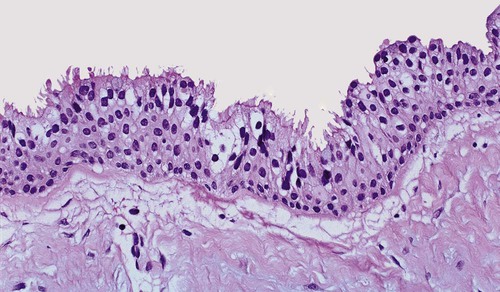
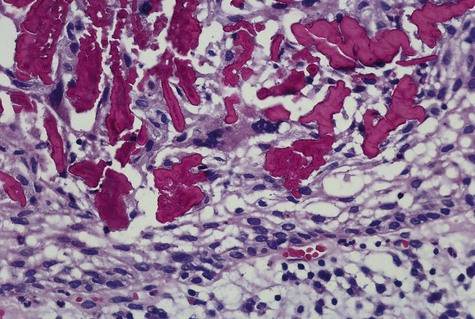
The periapical cyst is lined by nonkeratinized stratified squamous epithelium of variable thickness (Figure 10-6). Transmigration of inflammatory cells through the epithelium is common, with large numbers of neutrophils (polymorphonuclear leukocytes [PMNs]) and fewer numbers of lymphocytes involved. The underlying supportive connective tissue may be focally or diffusely infiltrated with a mixed inflammatory cell population. The molecular signature of a periapical granuloma differs from that of the radicular cyst, with a high level of matrix metalloproteinase (MMP) activity compared with that within a radicular cyst. Plasma cell infiltrates and associated refractile and spherical intracellular Russell bodies, representing accumulated gamma globulin, are often found and sometimes dominate the microscopic picture. Foci of dystrophic calcification, cholesterol clefts, and multinucleated foreign body–type giant cells may be seen subsequent to hemorrhage in the cyst wall. A foreign body reaction to vegetable matter (Figure 10-7) (pulse or seed granuloma) is occasionally found in periapical cyst walls, indicating apical communication with the oral cavity through the root canal and carious lesion.
Treatment and Prognosis
When the necrotic tooth is extracted but the cyst lining is incompletely removed, a residual cyst may develop months to years after the initial extirpation (Figure 10-8). If a residual cyst or the original periapical cyst remains untreated, continued growth can cause significant bone resorption and weakening of the mandible or maxilla. Complete bone repair is usually seen in adequately treated periapical and residual cysts.
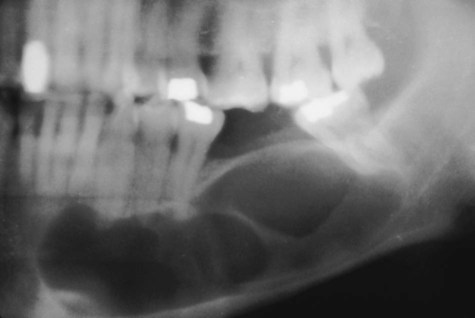
Lateral Periodontal Cyst
Clinical Features
Most lateral periodontal cysts and gingival cysts of the adult occur in the mandibular premolar and cuspid regions and occasionally in the incisor area (Figure 10-9; Box 10-2). In the maxilla, lesions are noted primarily in the lateral incisor region. A distinct male predilection has been noted for lateral periodontal cysts, with a greater than 2 : 1 distribution. Gingival cysts show a nearly equal gender predilection. The median age for both types of cysts is between the fifth and sixth decades of life, with a range of 20 to 85 years for lateral periodontal cysts, and 40 to 75 years for gingival cysts of the adult.
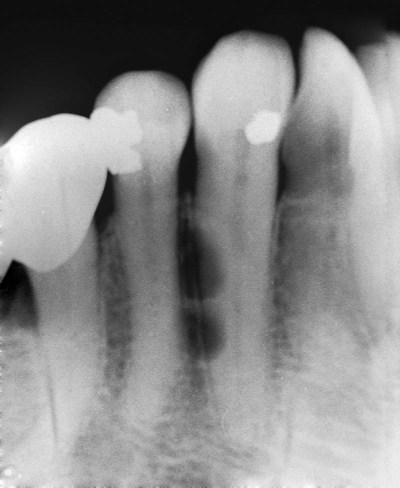
Clinically, a gingival cyst appears as a small soft tissue swelling within or slightly inferior to the interdental papilla (Figure 10-10). It may assume a slightly bluish discoloration when it is relatively large. Most cysts are less than 1 cm in diameter. Radiography reveals no findings.
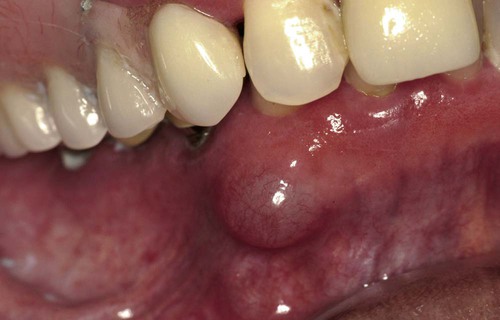
Dentigerous Cyst
Clinical Features
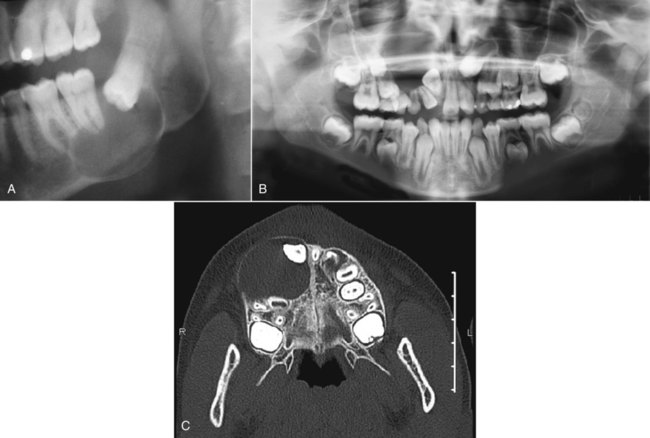
Dentigerous cysts are most commonly seen in association with third molars and maxillary canines, which are the most commonly impacted teeth (Box 10-3; Figure 10-14). The highest incidence of dentigerous cysts occurs during the second and third decades. A greater incidence in males has been noted, with a ratio of 1.6 : 1 reported.
Radiographically, a dentigerous cyst presents as a well-defined, unilocular radiolucency with corticated margins in association with the crown of an unerupted tooth. The unerupted tooth is often displaced (Figure 10-15). These cysts range in size from several millimeters to several centimeters, where they may compromise jawbone integrity and produce facial asymmetry. In the mandible, associated radiolucency may extend superiorly from the third molar site into the ramus or anteriorly and inferiorly along the body of the mandible. In maxillary dentigerous cysts involving the canine region, extension into the maxillary sinus or to the orbital floor may be noted. Resorption of roots of adjacent erupted teeth may occasionally be seen.
A variant of the dentigerous cyst arising at the bifurcation of molar teeth is the paradental cyst or buccal bifurcation cyst (Figure 10-16). Originally, this cyst was described along the buccal root surface of partially erupted mandibular third molar teeth, but later, involvement of other mandibular molar teeth was recognized. Often in these latter circumstances, the molar teeth are fully erupted. Radiographically, paradental cysts are characterized as well-circumscribed radiolucencies in the buccal bifurcation region. Often buccal tipping of the crown can be demonstrated by occlusal radiography.
Histopathology
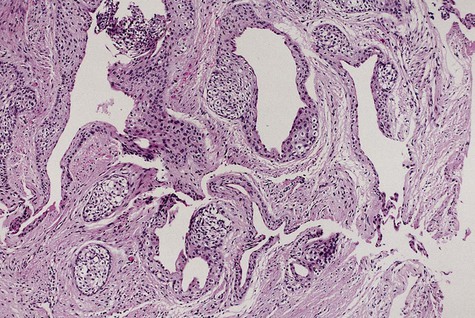
Microscopically, the dentigerous cyst is formed by a fibrous connective tissue wall and is lined by stratified squamous epithelium (Figures 10-17 to 10-19). In an uninflamed dentigerous cyst, the epithelial lining is nonkeratinized and tends to be approximately four to six cell layers thick. On occasion, numerous mucous cells, ciliated cells, and, rarely, sebaceous cells may be found in the lining of the epithelium. The epithelium–connective tissue junction is generally flat, although in cases of secondary inflammation, epithelial hyperplasia may be noted.
Eruption Cyst
An eruption cyst results from fluid accumulation within the follicular space of an erupting tooth (Figure 10-20). The epithelium lining this space is simply reduced enamel epithelium. With trauma, blood may appear within the tissue space, forming an eruption hematoma. No treatment is needed because the tooth erupts through the lesion. Subsequent to eruption, the cyst disappears spontaneously without complication.
Glandular Odontogenic Cyst
Clinical Features
A strong predilection is seen for the mandible (80%), especially the anterior mandible (Box 10-4; Figure 10-21). Maxillary lesions tend to be localized to the anterior segment. A slow growth rate is characteristic and symptoms are absent. Jaw expansion is not uncommon, particularly in association with mandibular lesions. The gender ratio is approximately 1 : 1. The mean age is 50 years, with a wide age range from the second through ninth decades.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses