Casting Metals, Solders, and Wrought Metal Alloys
Chapter Objectives
On completion of this chapter, the student will be able to:
1. Describe the differences among the types of gold alloy used for dental restorations.
3. Differentiate among high-noble, noble, and base-metal alloys.
4. Describe the characteristics needed for porcelain bonding alloys.
5. Describe the characteristics of metals used for casting partial denture frameworks.
6. Explain the biocompatibility problems associated with some alloys.
7. Explain how solders are used.
8. List metals used for solders.
9. Describe how wrought metal alloys differ from casting alloys.
10. Describe the uses of wrought wire.
11. Describe the metals used for orthodontic brackets and how they bond to teeth.
12. Explain the use of the different types of metal wire for orthodontic arch wire.
13. Explain the purpose of a post.
14. List the various classifications of posts.
15. Describe the types of materials used for pre-formed posts.
Key Terms defined within the chapter
Alloy a mixture of two or more metals
High-Noble Alloy alloy containing at least 60% noble metals, 40% of which must be gold
Base-Metal Alloy alloy composed of non-noble metals. Corrodes more readily
Noble Alloy alloy composed of metals that do not corrode readily
Precious Metal classification of metal based on its high cost
Porcelain Bonding Alloys special casting alloys manufactured for their compatibility with porcelain that has been bonded to them at high temperature
Coping a thin covering like a thimble that serves as a substructure for a porcelain-bonded-to-metal crown
Firing a process of heating porcelain at high temperature until it fuses
Yield Strength amount of stress at which a substance deforms
Glazing firing porcelain at high temperature to achieve a smooth, shiny surface
Solder an alloy used to join two metals together or to repair cast metal restorations
Wrought Metal Alloy an alloy that has been mechanically changed into another form to improve its properties
Anneal to modify physical properties of a metal by heating it
Nitinol an alloy of nickel and titanium often used for orthodontic wires
Post a metal or nonmetal dowel placed within the root canal to retain a core buildup
Active Post post that engages the root canal surface like a screw
Passive Post post that sits in the prepared canal space but does not actively engage the root surface
Custom Post a post cast to fit precisely in the root canal space; usually has the core attached
Pre-formed Post factory-made post supplied in several sizes
Historically, the most widely used material in restorative and corrective dentistry has been metal. As esthetic nonmetal materials improve in their physical properties, they are slowly replacing metals in some applications. Metals generally have high strength and consequently make durable dental restorations. They melt at high temperatures, conduct temperature and electricity, can be polished to a high shine (luster), and have varying degrees of ductility (ability to be pulled or drawn into a wire). Gold is ductile and the margins of gold restorations can be burnished for a better adaptation to the tooth preparation margins. Although a pure metal may not possess the physical and mechanical properties desired for a restoration, it may be combined with one or more other metals to form an alloy with the properties desired.
The dental health care worker is in contact with and is involved in the manipulation of metal dental materials in various ways every day. It is essential that the dental assistant or hygienist have an understanding of the characteristics of various metal materials in order to correctly manipulate and care for them and to be able to answer questions by patients relative to a particular material that will be used for their treatment.
Casting Alloys
All-Metal Castings
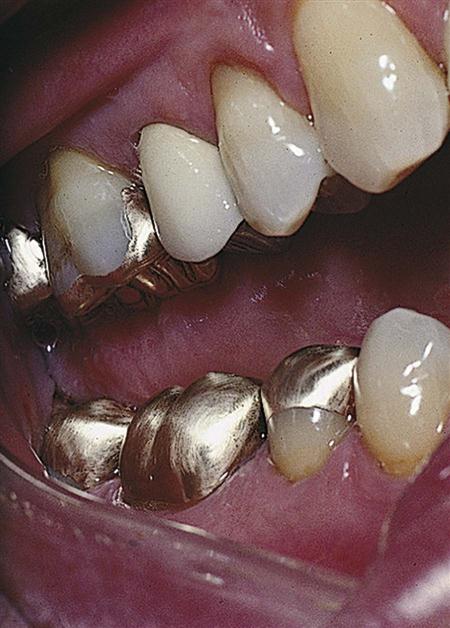
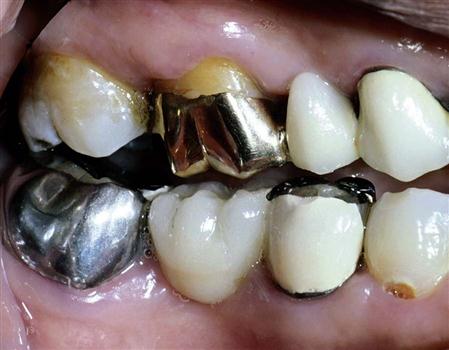
In the early 1900s, Taggart developed a technique for making dental restorations from metal that was melted and cast into a mold using the lost wax technique (see Chapter 18). Pure metals are seldom used in dentistry, because they lack properties that make them useful in the oral cavity. Therefore, they are usually combined with other metals in portions that achieve desirable physical and mechanical properties. This combination of two or more metals is called an alloy. Alloys used with the lost wax technique are called dental casting alloys. Unlike amalgam, restorations made from these alloys are not placed directly into the preparation but are made outside the mouth using an indirect technique, then are cemented in place (see Chapters 6 and 13). Cast-metal restorations can be classified as intracoronal (preparation is made within the crown of the tooth) or extracoronal (preparation is made primarily on the outside of the crown of the tooth). An inlay is an intracoronal restoration, whereas an onlay has both intracoronal and extracoronal components in that it has an inlay preparation and also covers the outer surface of one or more cusps. Other extracoronal cast restorations include partial coverage ( and
and  crowns) and full coverage crowns (Figure 10-1). Cast metal alloys can also be used to make fixed partial dentures (bridges) and removable partial dentures for replacement of missing teeth.
crowns) and full coverage crowns (Figure 10-1). Cast metal alloys can also be used to make fixed partial dentures (bridges) and removable partial dentures for replacement of missing teeth.
The American Dental Association classifies dental casting alloys according to their noble metal content and divides them into three categories. First are the high-noble alloys that must contain at least 60% by weight noble alloys, of which gold must account for at least 40% by weight. Second are the noble alloys that do not require any gold but must contain at least 25% noble metals by weight. Third are the base-metal alloys that do not require gold and require less than 25% by weight of noble metals. The International Standards Organization (ISO) also sets specifications for casting alloys that can be found at its website (www.iso.org).
High-Noble Dental Casting Alloys
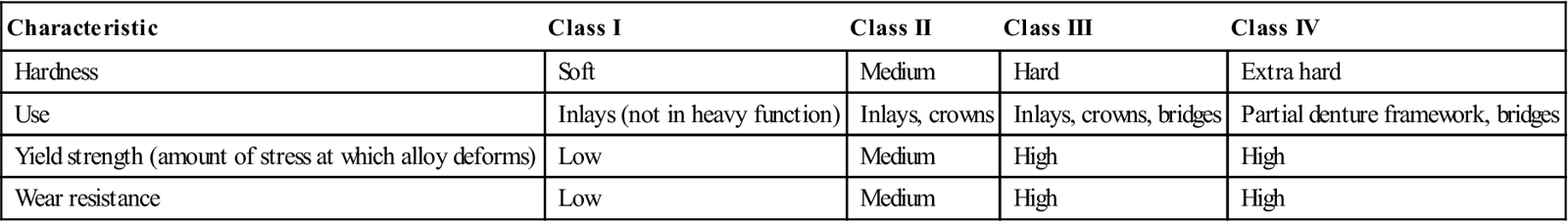
A noble alloy is one that does not corrode very readily in the oral environment. Gold (chemical symbol Au) is the most corrosion resistant of the noble metals and has been used in dentistry for centuries. Gold alloy is classified by its gold content as karats (also spelled carats), percentage, or fineness (obtained by multiplying percentage of gold by 10). Pure gold is 24 karat, 100%, or 1000 fine, and half gold is 12 karat, 50%, or 500 fine. Pure gold has limited use in dentistry today but is used in the form of gold foil by a small number of dentists for direct-placement restorations. Pure gold is too soft to use for dental castings, but gold alloys have excellent properties and handling characteristics. Dental gold casting alloys can be classified by their hardness (resistance to penetration), malleability (ability to be shaped as by tapping or pounding), and ductility (ability to be elongated as by stretching or pulling) (Table 10-1). The more ductile the alloy, the more the margins can be burnished (pushing or pulling the metal at the margins to close small gaps between the restoration and the tooth).
TABLE 10-1
< ?comst?>
| Characteristic | Class I | Class II | Class III | Class IV |
| Hardness | Soft | Medium | Hard | Extra hard |
| Use | Inlays (not in heavy function) | Inlays, crowns | Inlays, crowns, bridges | Partial denture framework, bridges |
| Yield strength (amount of stress at which alloy deforms) | Low | Medium | High | High |
| Wear resistance | Low | Medium | High | High |
< ?comen?>< ?comst1?>
< ?comst1?>
< ?comen1?>
Other Noble Metals for Casting Alloys
Other noble metals include platinum (Pt) and palladium (Pd). Platinum is not used much because of its expense, high melting point, and difficulty mixing with gold. Palladium is used widely because it has good corrosion resistance, increases hardness of the alloy, and is less expensive than gold. (Palladium alloys have greatly increased in price because of shortages in the supply of palladium.) These noble metals are sometimes referred to as precious metals because of their high monetary value. Although silver (Ag) is considered to be a precious metal, it is not considered noble because of its tarnish and corrosion in the oral cavity.
Noble metals are divided into high-noble and noble depending on the percentage of noble elements present. To be in the high-noble class, they must contain at least 60% noble elements (Au, Pd, and Pt), of which 40% must be gold. Base metals (usually copper, silver, or gallium) make up the remaining 40%. Noble class alloys contain at least 25% noble elements with no requirement for gold, and the remaining 75% consist of base metals. Because of the lower cost of the metals used in the noble class, they also have been referred to as semiprecious alloys.
Base-Metal Dental Casting Alloys
The base-metal class of dental casting alloys consists of less than 25% noble metals. The primary base metals used in casting alloys are copper (Cu), nickel (Ni), silver (Ag), zinc (Zn), tin (Sn), and titanium (Ti). Copper and silver are often added to gold alloys to increase their hardness. Zinc is added to reduce oxidation when the alloy is cast. Because of their low cost, base metals have also been called nonprecious metals. Although they are not considered to be as good as the noble metals, the base metals are essential for many applications in dentistry. The stiffness (modulus of elasticity) of base alloys is twice as great as that of gold-based alloys. This property is especially important for use in partial denture frameworks where flexing of the framework would put undue stress on abutment teeth. Drawbacks of base metals include their higher casting temperatures that require different equipment and investing materials than the gold-based alloys and their potential biocompatibility problems. They are much harder than noble metals, making them difficult to cut and to finish. Other base metals used for casting removable prostheses are discussed later in this chapter (see Removable Prosthetic Casting Alloys).
Crystal Formation (Grains)
After casting alloys have been melted and cast into the mold, they cool and form crystals (also called grains). Small crystals produce more desirable properties in the metal alloy than large crystals do. Some elements such as iridium or ruthenium are added to gold-based alloys to keep the crystals from growing too large. Reheating gold-based alloys (called annealing) can improve some of the properties. However, with base-metal alloys, reheating will degrade the physical properties.
Porcelain Bonding Alloys
Porcelain bonding alloys are similar to the other casting alloys with similar physical properties. They are also classified as high-noble, noble, and base-metal alloys (Figure 10-2). However, they have minor changes in their composition that make them compatible with dental ceramic materials. The most common ceramic materials used with these metals are conventional feldspar-based porcelains. The metals in porcelain bonding alloys are selected and blended so that they have the ability to withstand the high temperatures at which porcelain is fired without distorting or melting. They also have a lower thermal expansion than gold alloys used for all-metal crowns, so they do not expand too greatly and crack the brittle porcelain that lies over top. Additionally, small amounts of metals such as indium, iron, tin, or gallium are added to form the oxides on the metal surface to which the porcelain will bond (or fuse) at high temperature. Alloys that contain silver and copper may cause green staining (called greening) of the porcelain when fired at high temperatures, so they are usually avoided for use with ceramic materials. Most of the alloys are silver (white) in color, but alloys that are gold colored (yellow) contain mostly gold with platinum and palladium added to increase the melting temperature. Titanium and titanium alloys have such high melting temperatures and difficulty in handling that they have limited use for ceramic-metal crowns.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses