Obstructive sleep apnea syndrome (OSAS) is a relatively common disorder characterized by recurrent episodes of upper airway collapse and obstruction during sleep. It results in apneic events (or in the case of partial obstruction, hypopneic events) that lead to loud snoring, hypoxia, and arousals that interrupt the normal sleep cycle. Typically, these nocturnal arousals cause patients to complain of excessive daytime sleepiness, which may interfere with routine daily activities. More importantly, when moderate to severe, OSAS can lead to a wide variety of medical complications, some of which can result in severe consequences or even death.
Of the approximately 70 million Americans who complain of snoring, an estimated 20%—approximately 15 million people—actually have OSAS. Unfortunately, because of the difficulty in obtaining a proper diagnosis, 80% of the cases remain undiagnosed . Previous data from a cohort study (adults aged 30–60 years) showed the lifetime incidence of OSAS to be 9% to 24% for men and 4% to 9% for women. The estimated prevalence (percent of the population affected at any given time) of OSAS is 4% in men and 2% in women . Similar data have been reported from epidemiologic studies .
Risk factors for OSAS include a family history of sleep apnea, a large neck, a recessed chin, any abnormalities in the structure of the upper airway, smoking, alcohol use, and age. In addition to loud snoring and excessive daytime sleepiness, presenting signs and symptoms include high blood pressure and other cardiovascular complications, morning headaches, memory problems, feelings of depression, gastroesophageal reflux, nocturia, and impotence. Excessive weight is a predominant physical characteristic and compounding factor to the prevalence of OSAS. Aside from better recognition, one reason for the increasing number of OSAS diagnoses is the increasing percentage of Americans who are obese. More than 50% of the American population has a body mass index (weight in kg/height in m 2 ) of more than 30, and research has shown a strong correlation between obesity and nocturnal respiratory disturbance . A large population-based study also showed that the level of disturbance is directly proportional to the degree of obesity .
More dentists are getting involved in the diagnosis and management of OSAS. Oral and maxillofacial surgeons are well suited to perform any surgical interventions necessary, from diagnostic nasopharyngoscopy to soft tissue airway procedures (eg, uvulopalatopharyngoplasty, genioglossus muscle advancement, tongue base surgery) to major maxillomandibular advancement telegnathic surgery. General dentists are also becoming more involved, however, specifically in the area of dental appliances to maintain or advance mandibular position during sleep. These appliances have been shown to be as effective as a first-line treatment as the traditional primary treatment method—continuous positive airway pressure . Many people visit their dentist on a more regular basis than their physicians, and dentists have an excellent opportunity to provide screening for the signs and symptoms of OSAS on their health history forms and during the discussion phase of their dental treatment. This screening allows patients to be referred in a timely fashion to an oral and maxillofacial surgeon and formal sleep physician specialist for definitive diagnosis and imaging.
Diagnosis
The initial diagnosis of OSAS is almost always made while taking a good history. Although the history (eg, excessive daytime sleepiness, loud snoring) is a valid screening tool, it is not reliably diagnostic. Up to 30% of patients who have sleep apnea have no signs or symptoms other than loud snoring . Another important assessment tool for the degree of interrupted sleep that a patient experiences is the Epworth Sleepiness Scale, which is a subjective score of a patient’s tendency to fall asleep during specific nonstimulating situations, such as driving a car or watching a movie. Although a history and the Epworth Sleepiness Scale can provide some insight into the likelihood that a patient may have OSAS, it is clear that an objective mechanism for diagnosis is necessary before any treatment begins.
The gold standard for diagnosis of OSAS is the polysomnogram (PSG). Polysomnography is a monitored sleep study that is performed in a sleep laboratory and can be performed during the day or night (depending on the patient’s normal sleep time) to evaluate a patient during the sleep cycle. The typical PSG has 11 parameters, such as electroencephalogram, monitoring of air flow, electroymogram of the chin, legs, and ocular muscles (also known as electro-oculogram), electrocardiogram, and monitoring of oxygen saturation and chest wall movement. These parameters are evaluated by a sleep specialist and are interpreted in multiple ways to determine the extent of the interrupted sleep experienced by the patient. The report from the PSG provides objective data on the type and frequency of any sleep-related breathing abnormalities, the duration and degree of these abnormalities, and any cardiac, neurologic, or pulmonary consequences of these events (eg, cardiac arrythmias).
Definitions
An OSA event is defined as a cessation of breathing during sleep that lasts for 10 seconds or more, is associated with at least a 2% drop in oxygen saturation, and leads to an arousal from sleep on electroencephalogram (although the definitions vary somewhat by clinical center). Hypopnea is a period of time during which breathing is reduced by at least 50% with a similar decrease in oxygen saturation and arousal. These are laboratory diagnoses. OSAS is defined as having these objective events and having signs and symptoms from these abnormalities, such as daytime somnolence and snoring. The PSG provides objective data in the form of an apnea-hypopnea index, which is the total number of apneic and hypopneic events divided by the number of hours of sleep. Some laboratories also provide a respiratory disturbance index (RDI), which is the total number of apneic and hypopneic events plus the respiratory effort related arousals, which are arousals during sleep that do not meet the strict apneic or hypopneic criteria (eg, lasting less than 10 seconds or not associated with a significant drop in oxygen saturation). A normal apnea-hypopnea index is evidenced by fewer than five events per hour, whereas OSA is defined as more than five apneic or hypopneic events. Depending on the degree of associated desaturation, between 5 and 20 events per hour may be classified as mild to moderate sleep apnea, which can be associated with increased risks such as hypertension, stroke, and cardiac disease. In moderate to severe OSA, when the RDI exceeds 20 (and in some patients may approach 100 events per hour), a dramatic increase in morbidity and mortality may be present . A list of potential physical and mental consequences is shown in Box 1 .
-
Excessive daytime sleepiness
-
Difficulties with concentration and short-term memory
-
Depression
-
Social withdrawal
-
Nocturnal awakening
-
Insomnia
-
Nocturia
-
Loud snoring
-
Weight gain
-
Truncal/visceral obesity
-
Menopausal status in women
-
Hypertension
-
Pulmonary hypertension
-
Stroke
-
Insulin resistance
-
Diabetes mellitus
-
Serum lipid abnormalities
-
Serum lipid abnormalities
-
Elevated C-reactive protein
-
Elevated liver enzymes
Although the PSG provides clinicians with an objective and reproducible mechanism to define the presence and extent of OSAS, it does nothing to elucidate the actual cause or site of the obstruction. Once the diagnosis is made, it is equally important to determine the site and level of the obstruction so that appropriate nonsurgical and surgical treatment planning can be undertaken. OSAS may be caused by obstruction anywhere in the upper airway, from the tip of the nose to the larynx. Many cases of OSAS are caused by obstruction at multiple levels, either simultaneously or in an alternating pattern. Box 2 lists some of the pathologic areas of obstruction. In some cases, however, the problem is not a pathologic entity but excessive mobility of the tissues within the airway, allowing for collapse during inspiration. It is important that the likely cause and site of obstruction be elucidated so that targeted treatment may be instituted.
-
Nose
-
Deviated septum
-
Polyposis
-
Septal hematoma
-
Septal dislocation
-
Rhinitis
-
Turbinate hypertrophy
-
Nasopharynx
-
Carcinoma
-
Adenoidal hypertrophy
-
Lymphoma
-
Choanal stenosis or atresia
-
Pharyngeal flap
-
Papillomatosis
-
Mouth and oropharynx
-
Hypertrophied tonsils
-
Elongated and/or thickened palate and uvula
-
Lymphoma of tonsils, lingual cyst
-
Lingual tonsillar hypertrophy
-
Macroglossia: acromegaly
-
Micrognathia: congenital or acquired
-
Lipoma of the neck
-
Hunter syndrome
-
Hurler syndrome
-
Head and neck burns
-
Papillomatosis
-
Hypopharynx
-
Floppy or elongated epiglottis
-
Mass of the valecula
-
Epiglottic edema
-
Excessive compliance of the pharyngeal walls
-
Larynx
-
Edema of epiglottis
-
Vocal cord paralysis
-
Laryngomalacia collapse of aryepiglottic folds
Definitions
An OSA event is defined as a cessation of breathing during sleep that lasts for 10 seconds or more, is associated with at least a 2% drop in oxygen saturation, and leads to an arousal from sleep on electroencephalogram (although the definitions vary somewhat by clinical center). Hypopnea is a period of time during which breathing is reduced by at least 50% with a similar decrease in oxygen saturation and arousal. These are laboratory diagnoses. OSAS is defined as having these objective events and having signs and symptoms from these abnormalities, such as daytime somnolence and snoring. The PSG provides objective data in the form of an apnea-hypopnea index, which is the total number of apneic and hypopneic events divided by the number of hours of sleep. Some laboratories also provide a respiratory disturbance index (RDI), which is the total number of apneic and hypopneic events plus the respiratory effort related arousals, which are arousals during sleep that do not meet the strict apneic or hypopneic criteria (eg, lasting less than 10 seconds or not associated with a significant drop in oxygen saturation). A normal apnea-hypopnea index is evidenced by fewer than five events per hour, whereas OSA is defined as more than five apneic or hypopneic events. Depending on the degree of associated desaturation, between 5 and 20 events per hour may be classified as mild to moderate sleep apnea, which can be associated with increased risks such as hypertension, stroke, and cardiac disease. In moderate to severe OSA, when the RDI exceeds 20 (and in some patients may approach 100 events per hour), a dramatic increase in morbidity and mortality may be present . A list of potential physical and mental consequences is shown in Box 1 .
-
Excessive daytime sleepiness
-
Difficulties with concentration and short-term memory
-
Depression
-
Social withdrawal
-
Nocturnal awakening
-
Insomnia
-
Nocturia
-
Loud snoring
-
Weight gain
-
Truncal/visceral obesity
-
Menopausal status in women
-
Hypertension
-
Pulmonary hypertension
-
Stroke
-
Insulin resistance
-
Diabetes mellitus
-
Serum lipid abnormalities
-
Serum lipid abnormalities
-
Elevated C-reactive protein
-
Elevated liver enzymes
Although the PSG provides clinicians with an objective and reproducible mechanism to define the presence and extent of OSAS, it does nothing to elucidate the actual cause or site of the obstruction. Once the diagnosis is made, it is equally important to determine the site and level of the obstruction so that appropriate nonsurgical and surgical treatment planning can be undertaken. OSAS may be caused by obstruction anywhere in the upper airway, from the tip of the nose to the larynx. Many cases of OSAS are caused by obstruction at multiple levels, either simultaneously or in an alternating pattern. Box 2 lists some of the pathologic areas of obstruction. In some cases, however, the problem is not a pathologic entity but excessive mobility of the tissues within the airway, allowing for collapse during inspiration. It is important that the likely cause and site of obstruction be elucidated so that targeted treatment may be instituted.
-
Nose
-
Deviated septum
-
Polyposis
-
Septal hematoma
-
Septal dislocation
-
Rhinitis
-
Turbinate hypertrophy
-
Nasopharynx
-
Carcinoma
-
Adenoidal hypertrophy
-
Lymphoma
-
Choanal stenosis or atresia
-
Pharyngeal flap
-
Papillomatosis
-
Mouth and oropharynx
-
Hypertrophied tonsils
-
Elongated and/or thickened palate and uvula
-
Lymphoma of tonsils, lingual cyst
-
Lingual tonsillar hypertrophy
-
Macroglossia: acromegaly
-
Micrognathia: congenital or acquired
-
Lipoma of the neck
-
Hunter syndrome
-
Hurler syndrome
-
Head and neck burns
-
Papillomatosis
-
Hypopharynx
-
Floppy or elongated epiglottis
-
Mass of the valecula
-
Epiglottic edema
-
Excessive compliance of the pharyngeal walls
-
Larynx
-
Edema of epiglottis
-
Vocal cord paralysis
-
Laryngomalacia collapse of aryepiglottic folds
Imaging
Diagnostic imaging techniques have improved significantly the understanding of the pathophysiology of OSAS. They allow for evaluation and treatment planning of surgical and nonsurgical therapeutic interventions that are specific to a patient’s likely source of obstruction. Several different imaging tools are currently being used in the diagnosis and treatment planning of OSAS. They range from direct examination using mirror or endoscopic visualization of the nasal and hypopharyngeal airway to sophisticated new techniques, such as three-dimensional MRI and three-dimensional cone beam tomography. It is important to understand that each modality has advantages and disadvantages, and it is up to individual clinicians to decide which imaging technique would best be used for any individual patient.
Panorex
Panoramic radiography ( Fig. 1 ) is commonly used in assessing the hard tissue of the maxilla and mandible in dental offices. Panoramic radiography allows for identification of hard tissue anatomy, such as the location of impacted teeth, the location of the inferior alveolar nerve canal, evaluation of the maxillary sinuses, and the anatomy of the condylar head, all of which may be useful in ruling out anatomic abnormalities that are causing obstruction or, more commonly, any anatomic issues that could affect hard tissue surgical procedures, such as maxillomandibular telegnathic surgery. Little diagnostic information specific for the diagnosis of OSAS is obtained from the panorex; however, it does provide preoperative documentation of existing anatomy important in the surgical treatment planning, and it provides a postoperative assessment of surgical intervention and anatomic changes.
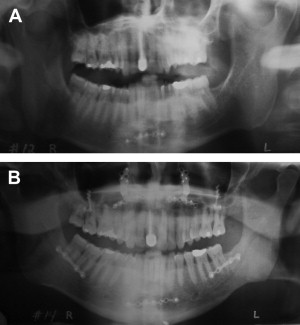
Rose and colleagues used the panoramic radiograph to monitor the dental condition for patients who have OSAS who were being treated with oral protrusive devices. Healthy oral conditions with sufficient dental retention for the device are essential for successful outcomes using dental appliances for OSAS. They concluded that periodontal disease and unwanted orthodontic movement of the teeth could result from such devices, which limits the indication of this therapeutic approach in some patients. They suggest that to maximize treatment success and minimize dental side effects, close collaboration with dental colleagues and routine panoramic radiographs is necessary in treating patients who have OSAS with an oral protrusive device .
Lateral cephalometric radiography
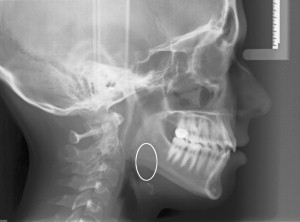
Plain film radiography is the most widely used modality for evaluating the hard and soft tissue anatomy of patients who have OSAS . Lateral cephalometric radiography is used routinely by orthodontists and oral and maxillofacial surgeons for evaluating the facial skeleton. Advantages of lateral cephalometric radiography are that it is inexpensive and minimally invasive, it exposes patients to low-dose radiation (0.005 mSv) , and it is easily obtained in the office setting. Standard positioning of the patient during the lateral cephalogram is with the patient’s left side toward the film (the patient is facing to his or her left), with Frankfort horizontal (defined as the plane from orbitale to porion) paralleling the horizon, and teeth in centric relation at the end of expiration . The reason for this positioning is that it is a standard, predictable, and reproducible head position that is consistent with the patient’s natural head position at rest ( Fig. 2 ).
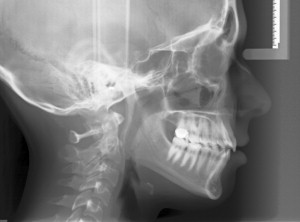
Many analyses are used to evaluate lateral cephalometric radiographs, predominately for use in orthognathic surgery. Specific measurements for assessing the airway are useful in evaluating patients with OSA :
- •
Soft palate length (PNS-P): the distance from posterior nasal spine (PNS) to the tip of the soft palate
- •
Posterior airway space (PAS): the distance from the base of the tongue to the posterior pharyngeal wall along the line from B-point through gonion
- •
Minimum posterior airway space: the narrowest point in the pharynx
- •
Mandibular plane to hyoid distance (MP-H): the distance determined by measuring a perpendicular line from the mandibular plane to the hyoid bone
- •
Bony nasopharynx (PNS-Ba): PNS to basion
- •
Bony oropharynx (PNS-AA): PNS to anterior border of the atlas (C2)
- •
Tonsillar size (T): perpendicular distance at the maximum convexity of the tonsil
- •
Tonsillar pharyngeal ratio (TP): the ratio of the width of the tonsil (T) to the depth of the pharyngeal space (PAS).
Patients who have OSAS have abnormal cephalometric anatomy, which may be a function of the hard and/or the soft tissue ( Figs. 3 and 4 ) .

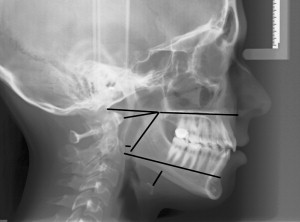
Yu and Fujimoto and colleagues examined the radiographic findings of 62 obese and nonobese patients who have OSAS. Their data showed that patients who have OSAS consistently have upper airway narrowing at the soft palate (short minimum posterior airway space), a longer soft palate (long PNS-P), an inferiorly positioned hyoid bone (long MP-H), narrowing of the bony nasopharynx (shorter PNS-Ba), and narrowing of the bony oropharynx (shorter PNS-AA). In comparing the obese patient and nonobese patients, the soft tissue differences were much more significant in obese patients , which indicated that adipose deposition in the upper airway may aggravate the severity of OSAS.
Because patients who have OSAS have abnormal cephalometric anatomy, researchers have studied whether the cephalometric measures may indicate a higher risk for severity of OSAS disease. Partinen and colleagues evaluated 157 patients who have OSAS by obtaining lateral cephalometric radiographs and PSG data. The results showed two important correlations: (1) that patients with OSAS consistently have longer mandibular plane to hyoid distance (MP-H) and more narrow posterior airway space (PAS), and (2) that when MP-H distance is more than 24 mm and PAS width is 5 mm or less, the RDI, as determined by the polysomnogram, is elevated. This study indicated that the severity of a low hyoid and a narrow posterior airway space is consistent with a higher risk for elevated RDI. Naganuma and colleagues obtained cephalometric and endoscopic studies on 64 patients previously diagnosed with OSAS or snoring. Statistically significant correlations were found between the apnea hypopnea index and minimum posterior airway space and mandibular plane to hyoid (MP-H) distance. The apnea hypopnea index increased with a minimum posterior airway space distance of less than 7 mm and an MP-H distance of more than 27.4 mm .
When assessing patients who have OSAS, it is important to distinguish between adult and the pediatric patients. OSAS is becoming a more frequently diagnosed disease in children, and studies have found the prevalence to be 2% to 4% in the US population . Similar to adults, there are many physiologic causes for OSAS in children, yet one significant difference is that adenotonsillar hypertrophy is the most common cause of OSAS in children . Li and Wong and colleagues measured TP ratio on standard lateral cephalometric radiographs and performed overnight PSG in 35 children. The tonsillar size is determined by measuring the perpendicular distance at the maximum convexity of the tonsil ( Fig. 5 ). The TP ratio is the tonsillar width (T) divided by the pharyngeal depth (PAS). All children in the study had some degree of tonsillar hypertrophy, and the TP ratio ranged from 0.65 to 0.8, meaning that on average the pediatric airways were 65% to 80% occluded by the tonsils. A significant correlation was found between the TP ratio and the RDI, which indicated a positive relationship between the amount of airway occlusion by the tonsils and the severity of OSAS .