Clinical Significance of Dental Anatomy, Histology, Physiology, and Occlusion
Teeth and Supporting Tissues
Dentitions
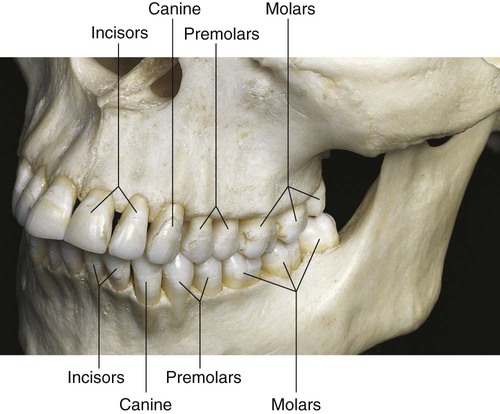
Classes of Human Teeth: Form and Function
Human teeth are divided into classes on the basis of form and function. The primary and permanent dentitions include the incisor, canine, and molar classes. The fourth class, the premolar, is found only in the permanent dentition (Fig. 1-1). Tooth form predicts the function of teeth; class traits are the characteristics that place teeth into functional categories. Because the diet of humans consists of animal and plant foods, the human dentition is called omnivorous.
Incisors
The incisors are located near the entrance of the oral cavity and function as cutting or shearing instruments for food (see Fig. 1-1). From a proximal view, the crowns of these teeth have a relatively triangular shape, with a narrow incisal surface and a broad cervical base. During mastication, incisors are used to shear (cut through) food. Incisors are essential for the proper esthetics of the smile, facial soft tissue contours (e.g., lip support), and speech (phonetics).
Canines
Canines possess the longest roots of all teeth and are located at the corners of the dental arch. They function in the seizing, piercing, tearing, and cutting of food. From a proximal view, the crown also has a triangular shape, with a thick incisal ridge. The anatomic form of the crown and the length of the root make these teeth strong, stable abutment teeth for a fixed or removable prosthesis. Canines not only serve as important guides in occlusion because of their anchorage and position in the dental arches but also play a crucial role (along with the incisors) in the esthetics of the smile and lip support (see Fig. 1-1).
Molars
Molars are large, multi-cusped, strongly anchored teeth located nearest the temporomandibular joint (TMJ), which serves as the fulcrum during function. These teeth have a major role in the crushing, grinding, and chewing of food to the smallest dimensions suitable for swallowing. They are well suited for this task because they have broad occlusal surfaces and multi-rooted anchorage (Fig. 1-2 and 1-3). Premolars and molars are important in maintaining the vertical dimension of the face (see Fig. 1-1).
Structures of Teeth
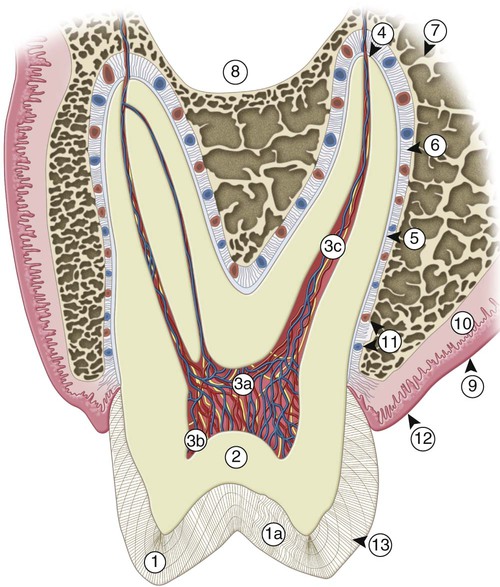
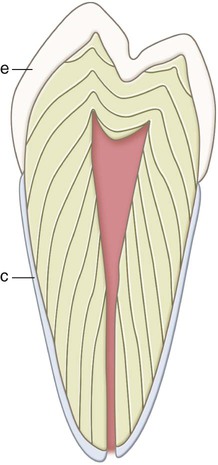
Teeth are composed of enamel, the pulp–dentin complex, and cementum (see Fig. 1-3). Each of these structures is discussed individually.
Enamel
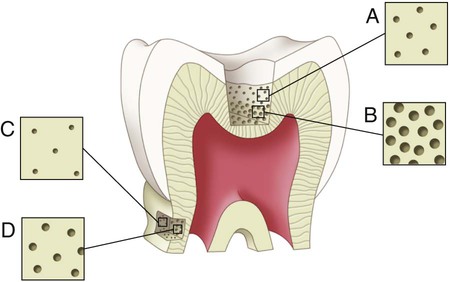
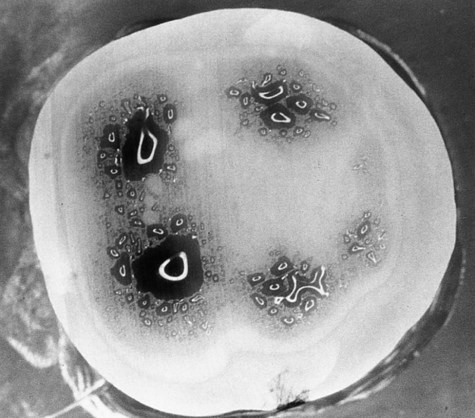
Enamel formation, amelogenesis, is accomplished by cells called ameloblasts. These cells originate from the embryonic germ layer known as ectoderm. Enamel covers the anatomic crown of the tooth and varies in thickness in different areas (see Fig. 1-3). It is thicker at the incisal and occlusal areas of a tooth and becomes progressively thinner until it terminates at the cementoenamel junction (CEJ). The thickness also varies from one class of tooth to another, averaging 2 mm at the incisal ridges of incisors, 2.3 to 2.5 mm at the cusps of premolars, and 2.5 to 3 mm at the cusps of molars. The cusps of posterior teeth begin as separate ossification centers, which form lobes that coalesce. Enamel usually decreases in thickness toward the junction of these developmental features and can approach zero where the junction is fissured (noncoalesced).
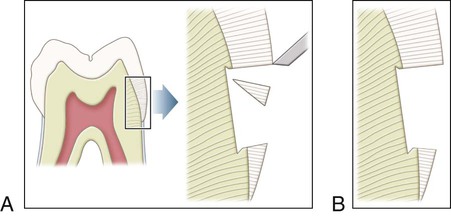
Enamel is the hardest substance of the human body. Hardness can vary over the external tooth surface according to the location; also, it decreases inwardly, with hardness lowest at the DEJ. The density of enamel also decreases from the surface to the DEJ. Enamel is a rigid structure that is both strong and brittle (high elastic modulus, high compressive strength, and low tensile strength) and requires a dentin support to withstand masticatory forces. Dentin is a more flexible substance that is strong and resilient (low elastic modulus, high compressive strength, and high tensile strength), which essentially increases the fracture toughness of the more superficial enamel. Enamel rods that lack dentin support because of caries or improper preparation design are easily fractured away from neighboring rods. For optimal strength in tooth preparation, all enamel rods should be supported by dentin (Fig. 1-4).
Human enamel is composed of rods that, in transverse section, have a rounded head or body section and a tail section, forming a repetitive series of interlocking prisms. The rounded head portion of each prism (5 µm wide) lies between the narrow tail portions (5 µm long) of two adjacent prisms (Fig. 1-5). Generally, the rounded head portion is oriented in the incisal or occlusal direction; the tail section is oriented cervically.
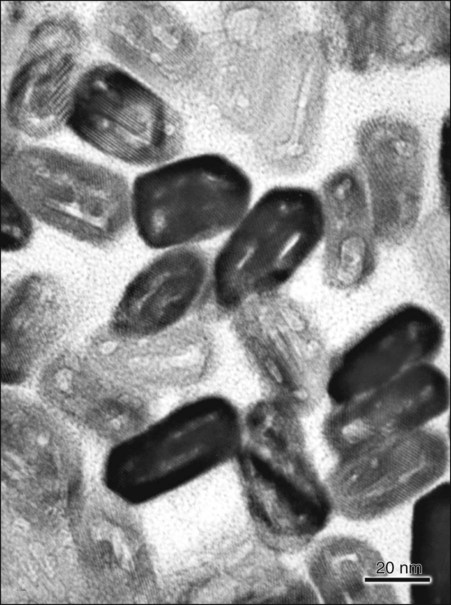
The structural components of the enamel prism are millions of small, elongated apatite crystallites that vary in size and shape. The crystallites are tightly packed in a distinct pattern of orientation that gives strength and structural identity to the enamel prisms. The long axis of the apatite crystallites within the central region of the head (body) is aligned almost parallel to the rod long axis, and the crystallites incline with increasing angles (65 degrees) to the prism axis in the tail region. The susceptibility of these crystallites to acid, from either an etching procedure or caries, may be correlated with their orientation. Although the dissolution process occurs more in the head regions of the rod, the tail regions and the periphery of the head regions are relatively resistant to acid attack. The crystallites are irregular in shape, with an average length of 160 nm and an average width of 20 to 40 nm. Each apatite crystallite is composed of thousands of unit cells that have a highly ordered arrangement of atoms. A crystallite may be 300 unit cells long, 40 cells wide, and 20 cells thick in a hexagonal configuration (Fig. 1-6). An organic matrix or prism sheath also surrounds individual crystals and appears to be an organically rich interspace rather than a structural entity.
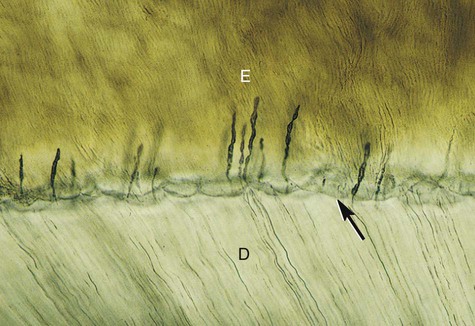
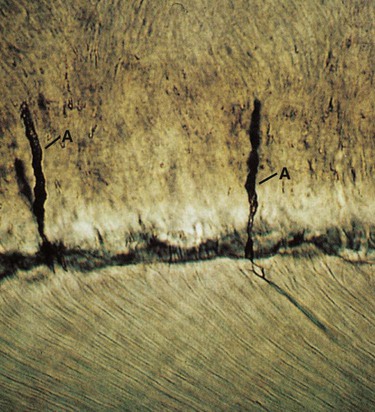
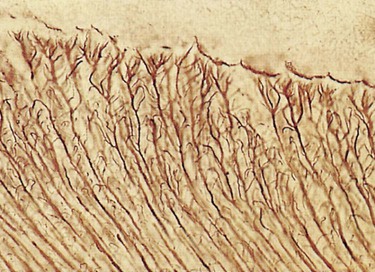
Enamel rods follow a wavy, spiraling course, producing an alternating arrangement for each group or layer of rods as they change direction in progressing from the dentin toward the enamel surface, where they end a few micrometers short of the tooth surface. Enamel rods rarely run a straight radial course, as there is an alternating clockwise and counterclockwise deviation of the rods from the radial course at all levels of the crown. They initially follow a curving path through one third of the enamel next to the DEJ. After that, the rods usually follow a more direct path through the remaining two thirds of the enamel to the enamel surface. Groups of enamel rods may entwine with adjacent groups of rods, and they follow a curving irregular path toward the tooth surface. These constitute gnarled enamel, which occurs near the cervical regions and the incisal and occlusal areas (Fig. 1-7). Gnarled enamel is not subject to fracture as much as is regular enamel. This type of enamel formation does not yield readily to the pressure of bladed, hand-cutting instruments in tooth preparation.
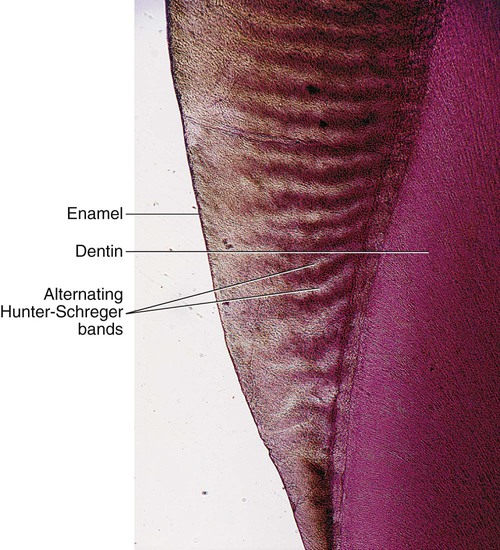
The changes in direction of enamel prisms that minimize fracture in the axial direction produce an optical appearance called Hunter-Schreger bands (Fig. 1-8). These bands appear to be composed of alternate light and dark zones of varying widths that have slightly different permeability and organic content. These bands are found in different areas of each class of teeth. Because the enamel rod orientation varies in each tooth, Hunter-Schreger bands also have a variation in the number present in each tooth. In anterior teeth, they are located near the incisal surfaces. They increase in numbers and areas of teeth, from canines to premolars. In molars, the bands occur from near the cervical region to the cusp tips. The orientation of the enamel rod heads and tails and the gnarling of enamel rods provide strength by resisting, distributing, and dissipating impact forces.
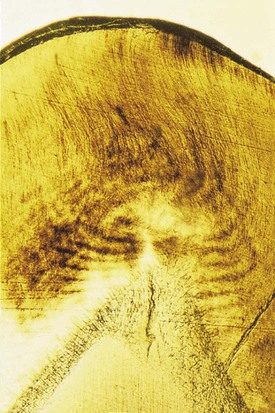
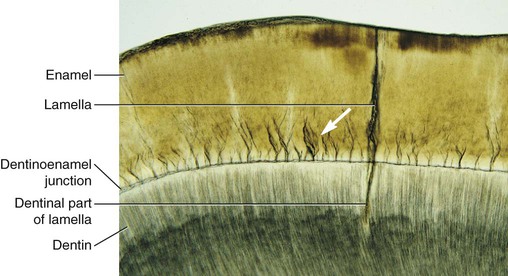
Enamel tufts are hypomineralized structures of the enamel rods and the inter-rod substance that project between adjacent groups of enamel rods from the DEJ (Fig. 1-9). These projections arise in dentin, extend into enamel in the direction of the long axis of the crown, and may play a role in the spread of dental caries. Enamel lamellae are thin, leaf-like faults between enamel rod groups that extend from the enamel surface toward the DEJ, sometimes extending into dentin (see Fig. 1-9). They contain mostly organic material, which is a weak area predisposing a tooth to the entry of bacteria and dental caries. Enamel rods are formed linearly by successive apposition of enamel in discrete increments. The resulting variations in structure and mineralization are called incremental striae of Retzius and can be considered growth rings (see Fig. 1-3). In horizontal sections of a tooth, the striae of Retzius appear as concentric circles. In vertical sections, the lines traverse the cuspal and incisal areas in a symmetric arc pattern, descending obliquely to the cervical region and terminating at the DEJ. When these circles are incomplete at the enamel surface, a series of alternating grooves, called imbrication lines of Pickerill, are formed. The elevations between the grooves are called perikymata; these are continuous around a tooth and usually lie parallel to the CEJ and each other.
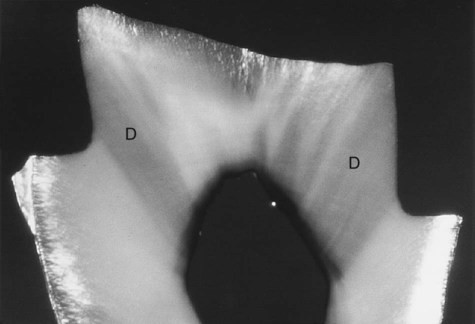
The interface of enamel and dentin (dentinoenamel junction, or DEJ) is scalloped or wavy in outline, with the crest of the waves penetrating toward enamel (Fig. 1-10). The rounded projections of enamel fit into the shallow depressions of dentin. This interdigitation may contribute to the firm attachment between dentin and enamel. The DEJ is also a hypermineralized zone approximately 30 µm thick.
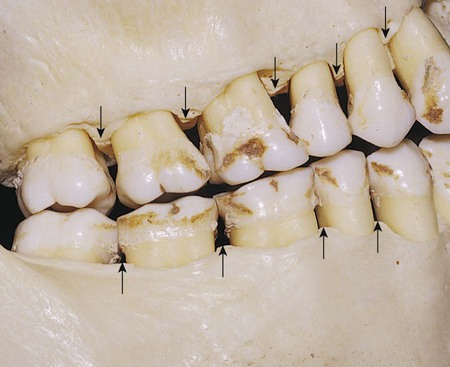
The occlusal surfaces of premolars and molars have grooves and fossae that form at the junction of the developmental lobes of enamel. These allow movement of food to the facial and lingual surfaces during mastication. A functional cusp that opposes a groove (fossa) occludes on enamel and inclines on each side of the groove and not in the depth of the groove. This arrangement leaves a V-shaped escape path between the cusp and its opposing groove for the movement of food during chewing. Failure of the enamel of the developmental lobes to coalesce results in a deep invagination of the enamel surface and is termed fissure. Non-coalesced enamel at the deepest point of a fossa is termed pit. These fissures and pits act as food and bacterial traps that predispose the tooth to dental caries (Fig. 1-11).
Pulp–Dentin Complex
The pulp is circumscribed by the dentin and is lined peripherally by a cellular layer of odontoblasts adjacent to dentin. Anatomically, the pulp is divided into (1) coronal pulp located in the pulp chamber in the crown portion of the tooth, including the pulp horns that are directed toward the incisal ridges and cusp tips, and (2) radicular pulp located in the pulp canals in the root portion of the tooth. The radicular pulp is continuous with the periapical tissues by connecting through the apical foramen or foramina of the root. Accessory canals may extend from the pulp canals laterally through the root dentin to the periodontal tissues. The shape of each pulp conforms generally to the shape of each tooth (see Fig. 1-3).
The pulp contains nerves, arterioles, venules, capillaries, lymph channels, connective tissue cells, intercellular substance, odontoblasts, fibroblasts, macrophages, collagen, and fine fibers.1 The pulp is circumscribed peripherally by a specialized odontogenic area composed of the odontoblasts, the cell-free zone, and the cell-rich zone.
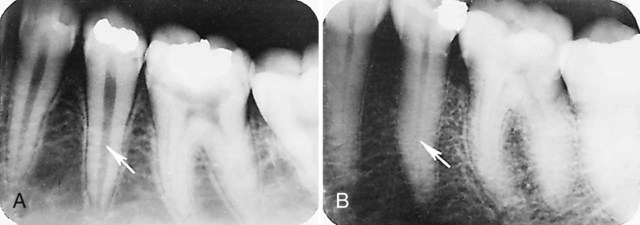
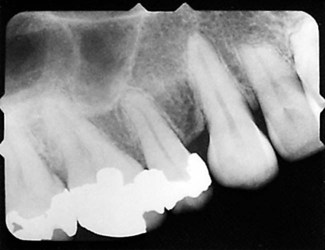
Knowledge of the contour and size of the pulp cavity is essential during tooth preparation. In general, the pulp cavity is a miniature contour of the external surface of the tooth. Pulp cavity size varies with tooth size among individuals and even within a single person. With advancing age, the pulp cavity usually decreases in size. Radiographs are an invaluable aid in determining the size of the pulp cavity and any existing pathologic condition (Fig. 1-12). A primary objective during operative procedures must be the preservation of the health of the pulp.
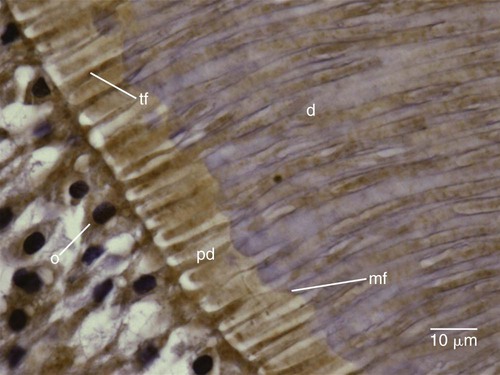
Dentin formation, dentinogenesis, is accomplished by cells called odontoblasts. Odontoblasts are considered part of pulp and dentin tissues because their cell bodies are in the pulp cavity, but their long, slender cytoplasmic cell processes (Tomes fibers) extend well (100–200 µm) into the tubules in the mineralized dentin (Fig. 1-13).
Because of these odontoblastic cell processes, dentin is considered a living tissue, with the capability of reacting to physiologic and pathologic stimuli. Odontoblastic processes occasionally cross the DEJ into enamel; these are termed enamel spindles when their ends are thickened (Fig. 1-14). They may serve as pain receptors, explaining the enamel sensitivity experienced by some patients during tooth preparation.
Dentin forms the largest portion of the tooth structure, extending almost the full length of the tooth. Externally, dentin is covered by enamel on the anatomic crown and cementum on the anatomic root. Internally, dentin forms the walls of the pulp cavity (pulp chamber and pulp canals) (Fig. 1-15). Dentin formation begins immediately before enamel formation. Odontoblasts generate an extracellular collagen matrix as they begin to move away from the adjacent ameloblasts. Mineralization of the collagen matrix, facilitated by modification of the collagen matrix by various noncollagenous proteins, gradually follows its secretion. The most recently formed layer of dentin is always on the pulpal surface. This unmineralized zone of dentin is immediately next to the cell bodies of odontoblasts and is called predentin. Dentin formation begins at areas subjacent to the cusp tip or incisal ridge and gradually spreads to the apex of the root (see Fig. 1-15). In contrast to enamel formation, dentin formation continues after tooth eruption and throughout the life of the pulp. The dentin forming the initial shape of the tooth is called primary dentin and is usually completed 3 years after tooth eruption (in the case of permanent teeth).
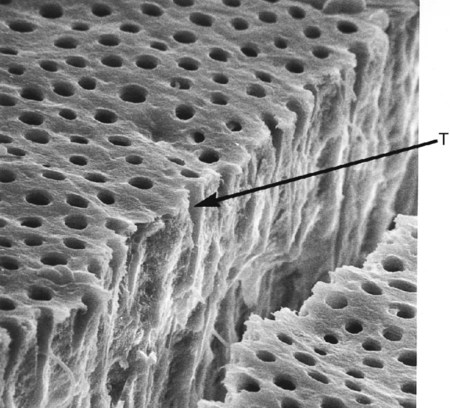
The dentinal tubules are small canals that extend through the entire width of dentin, from the pulp to the DEJ (Figs. 1-16 and 1-17). Each tubule contains the cytoplasmic cell process (Tomes fiber) of an odontoblast and is lined with a layer of peri-tubular dentin, which is much more mineralized than the surrounding intertubular dentin (see Fig. 1-17).
The surface area of dentin is much larger at the DEJ or dentinocemental junction than it is on the pulp cavity side. Because odontoblasts form dentin while progressing inward toward the pulp, the tubules are forced closer together. The number of tubules increases from 15,000 to 20,000/mm2 at the DEJ to 45,000 to 65,000/mm2 at the pulp.2 The lumen of the tubules also varies from the DEJ to the pulp surface. In coronal dentin, the average diameter of tubules at the DEJ is 0.5 to 0.9 µm, but this increases to 2 to 3 µm near the pulp (Fig. 1-18).
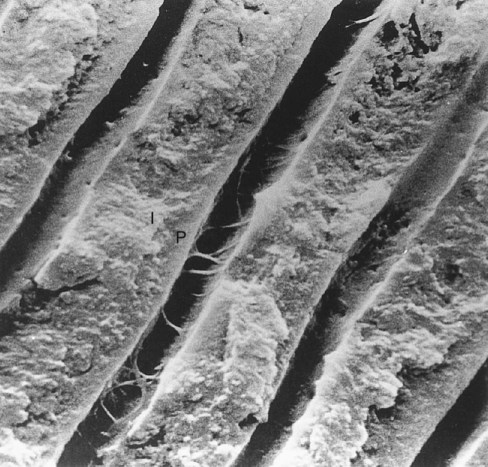
The course of the dentinal tubules is a slight S-curve in the tooth crown, but the tubules are straighter in the incisal ridges, cusps, and root areas (Fig. 1-19). The ends of the tubules are perpendicular to the DEJ. Along the tubule walls are small lateral openings called canaliculi. As the odontoblastic process proceeds from the cell in the pulp to the DEJ, lateral secondary branches extend into the canaliculi and can communicate with the lateral extensions of adjacent odontoblastic processes. Near the DEJ, the tubules divide into several terminal branches, forming an intercommunicating and anastomosing network (Fig. 1-20).
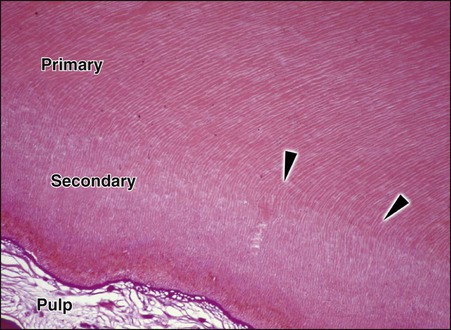
After the primary dentin is formed, dentin deposition continues at a reduced rate even without obvious external stimuli, although the rate and amount of this physiologic secondary dentin vary considerably among individuals. In secondary dentin, the tubules take a slightly different directional pattern in contrast to primary dentin (Fig. 1-21). Secondary dentin forms on all internal aspects of the pulp cavity, but in the pulp chamber, in multi-rooted teeth, it tends to be thicker on the roof and floor than on the side walls.3
When moderate stimuli are applied to dentin, such as caries, attrition, and some operative procedures, the affected odontoblasts may die. Replacement odontoblasts (termed secondary odontoblasts) of pulpal origin then begin to form reparative dentin (tertiary dentin). The reparative dentin usually appears as a localized dentin deposit on the wall of the pulp cavity immediately subjacent to the area on the tooth that has received the injury (a dentin deposit underneath the affected tubules) (Fig. 1-22). Being highly atubular, the reparative dentin is structurally different from the primary and secondary dentin.
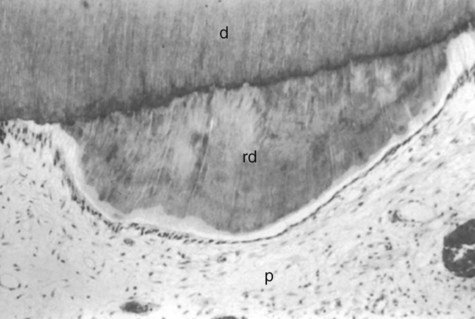
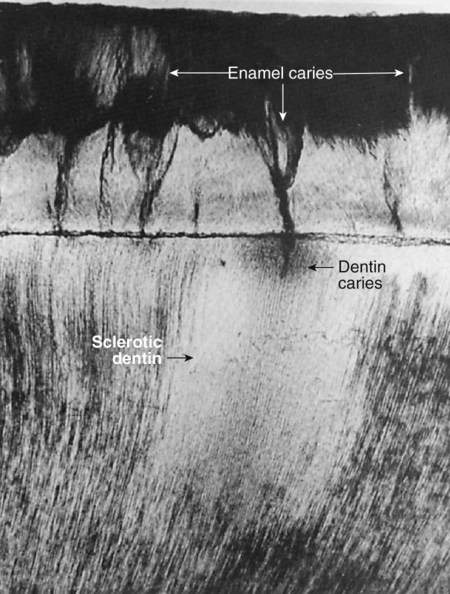
Sclerotic dentin results from aging or mild irritation (e.g., slowly advancing caries) and causes a change in the composition of the primary dentin. The peritubular dentin becomes wider, gradually filling the tubules with calcified material, progressing pulpally from the DEJ (Fig. 1-23). These areas are harder, denser, less sensitive, and more protective of the pulp against subsequent irritations. Sclerosis resulting from aging is called physiologic dentin sclerosis; sclerosis resulting from a mild irritation is called reactive dentin sclerosis. Reactive dentin sclerosis often can be seen radiographically in the form of a more radiopaque (lighter) area in the S-shape of the tubules.
Human dentin is composed of approximately 50% inorganic material and 30% organic material by volume. The organic phase is approximately 90% type I collagen and 10% noncollagenous proteins. Dentin is less mineralized than enamel but more mineralized than cementum or bone. The mineral content of dentin increases with age. This mineral phase is composed primarily of hydroxyapatite crystallites, which are arranged in a less systematic manner than are enamel crystallites. Dentinal crystallites are smaller than enamel crystallites, having a length of 20 to 100 nm and a width of about 3 nm, which is similar to the size seen in bone and cementum.3 Dentin is significantly softer than enamel but harder than bone or cementum. The hardness of dentin averages one-fifth that of enamel, and its hardness near the DEJ is about three times greater than near the pulp. Dentin becomes harder with age, primarily as a result of increases in mineral content. Although dentin is a hard, mineralized tissue, it is flexible, with a modulus of elasticity of approximately 18 gigapascals (GPa).4 This flexibility helps support the more brittle, nonresilient enamel. Often small “craze lines” are seen in enamel, indicating minute fractures of that structure. The craze lines usually are not clinically significant unless associated with cracks in the underlying dentin. Dentin is not as prone to fracture as is the enamel rod structure. The ultimate tensile strength of dentin is approximately 98 megapascals (MPa), whereas the ultimate tensile strength of enamel is approximately 10 MPa. The compressive strength of dentin and enamel are approximately 297 and 384 MPa, respectively.4
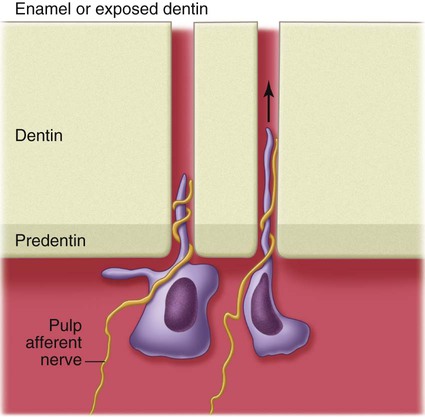
Sensitivity is encountered whenever odontoblasts and their processes are stimulated during operative procedures, even though the pain receptor mechanism appears to be within the dentinal tubules near the pulp. Physical, thermal, chemical, bacterial, and traumatic stimuli are transmitted through the dentinal tubules, although the precise mechanism of the transmissive elements of sensation has not been conclusively established. The most accepted theory of pain transmission is the hydrodynamic theory, which accounts for pain transmission through rapid movements of fluid within the dentinal tubules.5 Because many tubules contain mechanoreceptor nerve endings near the pulp, small fluid movements in the tubules arising from cutting, drying, pressure changes, osmotic shifts, or changes in temperature account for most pain transmission (Fig. 1-24).
Dentinal tubules are filled with dentinal fluid, a transudate of plasma. When enamel or cementum is removed during tooth preparation, the external seal of dentin is lost, allowing tubular fluid to move toward the cut surface. Pulpal fluid has a slight positive pressure that forces fluid outward toward any breach in the external seal. Permeability studies of dentin indicate that tubules are functionally much smaller than would be indicated by their measured microscopic dimensions as a result of numerous constrictions along their paths (see Fig. 1-17).6 Dentin permeability is not uniform throughout the tooth. Coronal dentin is much more permeable than root dentin. There also are differences within coronal dentin (Fig. 1-25).7 Dentin permeability primarily depends on the remaining dentin thickness (i.e., length of the tubules) and the diameter of the tubules. Because the tubules are shorter, more numerous, and larger in diameter closer to the pulp, deep dentin is a less effective pulpal barrier compared with superficial dentin (Fig. 1-26).
Cementum
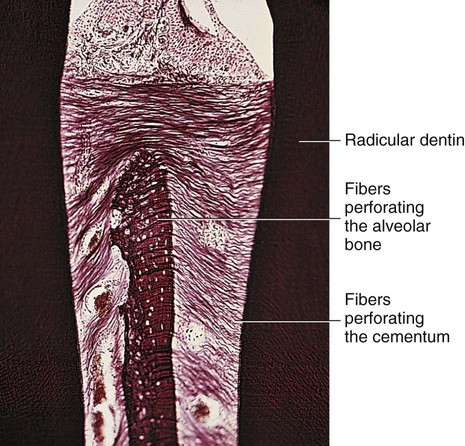
Cementum is a thin layer of hard dental tissue covering the anatomic roots of teeth and is formed by cells known as cementoblasts, which develop from undifferentiated mesenchymal cells in the connective tissue of the dental follicle. Cementum is slightly softer than dentin and consists of about 45% to 50% inorganic material (hydroxyapatite) by weight and 50% to 55% organic matter and water by weight. The organic portion is composed primarily of collagen and protein polysaccharides. Sharpey’s fibers are portions of the principal collagenous fibers of the periodontal ligament embedded in cementum and alveolar bone to attach the tooth to the alveolus (Fig. 1-27). Cementum is avascular.
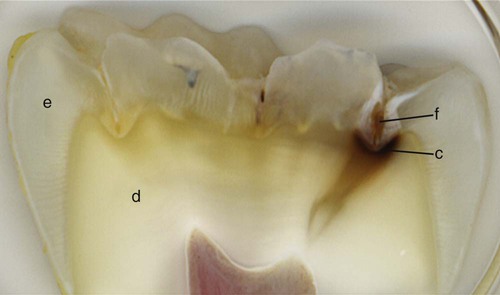
The cementodentinal junction is a relatively smooth area in the permanent tooth, and attachment of cementum to dentin is firm, but this is not understood completely yet. Cementum joins enamel to form the CEJ. In about 10% of teeth, enamel and cementum do not meet, and this can result in a sensitive area. Abrasion, erosion, caries, scaling, and restoration finishing and polishing procedures can denude dentin of its cementum covering, which can cause the dentin to be sensitive to various stimuli (e.g., heat, cold, sweet substances, sour substances). Cementum is capable of repairing itself to a limited degree and is not resorbed under normal conditions. Some resorption of the apical portion of the root can occur, however, if orthodontic pressures are excessive and movement is too fast (Fig. 1-28).
Physiology of Tooth Form
Function
Contours
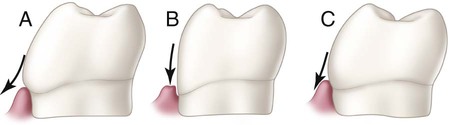
Facial and lingual surfaces possess a degree of convexity that affords protection and stimulation of supporting tissues during mastication. The convexity generally is located at the cervical third of the crown on the facial surfaces of all teeth and the lingual surfaces of incisors and canines. The lingual surfaces of posterior teeth usually have their height of contour in the middle third of the crown. Normal tooth contours act in deflecting food only to the extent that the passing food stimulates (by gentle massage) and does not irritate supporting tissues. If these curvatures are too great, tissues usually receive inadequate stimulation by the passage of food. Too little contour may result in trauma to the attachment apparatus. These tooth contours must be considered in the performance of operative dental procedures. Improper location and degree of facial or lingual convexities can result in serious complications, as illustrated in Figure 1-29, in which the proper facial contour is disregarded in the placement of a cervical restoration on a mandibular molar. Over-contouring is the worst offender, usually resulting in increased plaque retention that leads to a chronic inflammatory state of the gingiva.
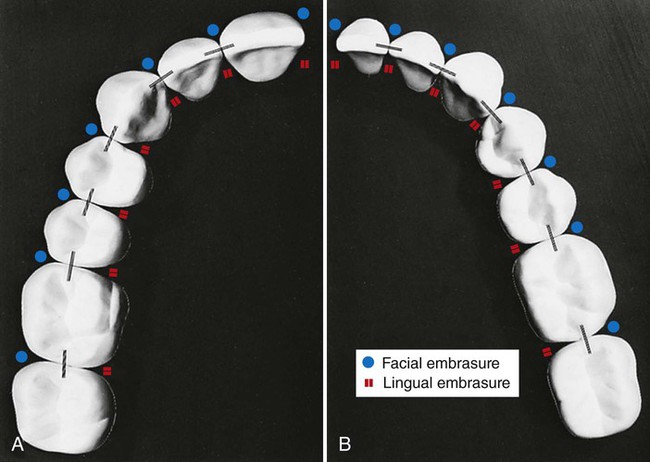
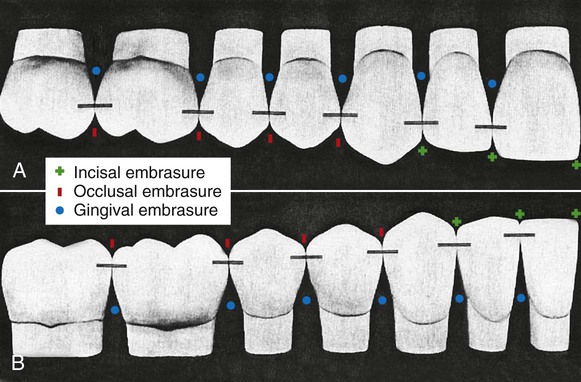
The proper form of the proximal surfaces of teeth is just as important to the maintenance of periodontal tissue as is the proper form of facial and lingual surfaces. The proximal height of contour serves to provide (1) contacts with the proximal surfaces of adjacent teeth, thus preventing food impaction, and (2) adequate embrasure space apical to the contacts for gingival tissue, supporting bone, blood vessels, and nerves that serve the supporting structures (Fig. 1-30).
Proximal Contact Area
When teeth erupt to make proximal contact with previously erupted teeth, initially a contact point is present. The contact point increases in size to become a proximal contact area as the two adjacent tooth surfaces abrade each other during physiologic tooth movement (Figs. 1-31 and 1-32).
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses