Rehabilitation of patients with maxillofacial defects presents a difficult challenge for the oral and maxillofacial surgeon, the maxillofacial prosthodontist, and the anaplastologist. To improve the care for patients with maxillofacial defects, it is important to understand their journey toward rehabilitation from the beginning.
Many of the aggressive lesions that require surgical resection, which can result in devastating defects, are operated on by oncological surgeons, whose primary goal is to limit and/or eliminate the spread of the primary disease to preserve the patient’s life. Although the primary disease may be cured, many of these patients acquire a secondary disease after their surgical resection, so-called postresection defects. These postsurgical defects are devastating in many ways, leading to these patients retracting from their family and society and living a life of seclusion and depression.
A collaborative approach by health care providers in treating this group of patients is imperative. Collaboration of the entire team comprising the oncological surgeon, the maxillofacial surgeon, the maxillofacial prosthodontist, and the anaplastologist must be in concert before initiating the resection procedure. Close communication with the oncological surgeon allows for a more auspicious postresection defect, which in turn allows the anchoring of extraoral implants to support the planned replacement of facial prosthesis.
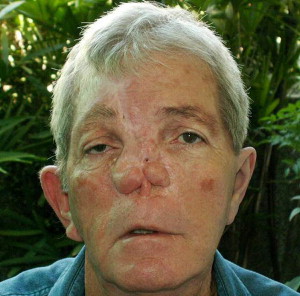
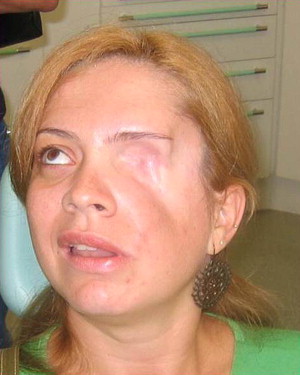
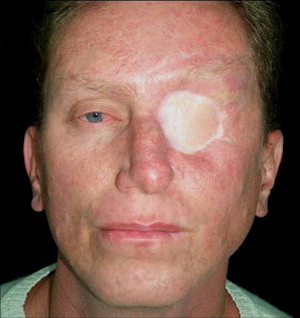
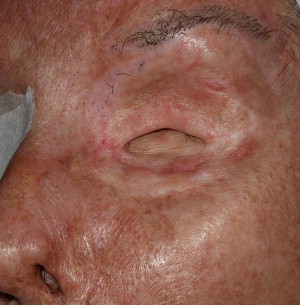
Autogenous tissues used for the reconstruction of these postresection defects are generally not predictable. After multiple surgeries, the resultant reconstruction is often unaesthetic and nonfunctional ( Figs. 1 and 2 ), hence, defects have been reconstructed prosthetically ( Figs. 3 and 4 ) using acrylic and silicone.

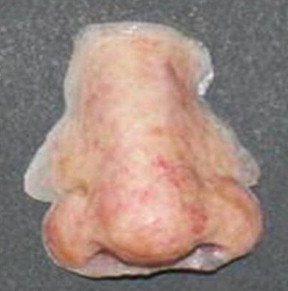
Many of the initial prosthetic reconstructions involved the use of a carrier. In the example of the patient in Fig. 3 , a pair of glasses must be used at all times to stabilize the prosthetic nose.
With the advent of a better material, such as silicone, the carriers were eliminated and skin adhesives were used to retain the prosthesis, as seen in Fig. 4 . However, skin irritations from the chronic use of adhesives as well as the hot and humid temperatures encountered by these patients result in the unpredictable retention of this prosthesis by the patients .
With the use of intraoral titanium fixtures applied in the extraoral region, the treatment of maxillofacial defects has become more predictable . Extraoral implants; maxillofacial implants, have allowed for simplification as well as the predictable treatment of this group of patients. The author in collaboration with Professor PI Brånemark, Maxillofacial Prosthodontist; Dr Marcelo De Oliveri; and corporate support from Nobel Biocare were successful in collaborating with the Food and Drug Administration (FDA) in 2008 in obtaining the Nobel Biocare implants for use in the maxillofacial region.
A protocol for the planning for the locations of the maxillofacial implants has been presented by this team, and the subject of this article is to share the protocol for the treatment of patients with maxillofacial defects.
The prosthesis determines the number and position of the implants
When considering the placement of maxillofacial implants in any type of maxillofacial defect, the fixtures should be placed with the planned prosthetic framework in mind. The angle of the implants should allow an emergence profile, allowing for a proper bar design without interfering with the ideal sculpture of the prosthesis. Misplaced implants may cause a poor aesthetic outcome. Proper spacing and angles of the implants is necessary to allow the manipulation of the prosthetic screws using the necessary drivers.
Pretreatment Criteria to Consider
The following points are a few of the preoperative considerations that allow predictable outcomes for the treatment of patients with simple as well as complex maxillofacial defects.
- 1.
Collaboration with oncological surgeons before the resection of maxillofacial lesions
- 2.
Planning the resection to allow adequate residual bony volume for the support of maxillofacial implants
- 3.
Consideration for the thickness of the overlying soft tissues
- 4.
Close planning with the maxillofacial prosthodontist and anaplastologist, allowing for the proper emergence profile of the prosthesis.
Preresection collaboration
Planning with the oncological surgeon before resection for patients with maxillary tumors is essential to maximize the residual volume of zygomatic bone remaining after the tumor ablation surgery. The zygomatic bone serves as an essential bony landmark in treatment planning of the anchorage positions for implants during the reconstructive phase of the patient’s care ( Fig. 5 ).
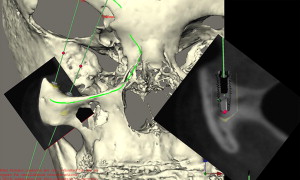
Communication of the contours of soft tissue reconstruction immediately after the resection of the tumors is also important. To allow adequate room for the emergence profile of the abutments as well as the retaining components of the maxillofacial prosthesis, considerations of the depth of the defect is critical. In cases of orbital exenteration, the free flap reconstruction must allow a residual concave defect. If the depth of the defect is not adequate, preprosthetic soft tissue surgery may be necessary in consultation with the maxillofacial prosthodontist and/or the anaplastologists. Close cooperation between the oncological surgeon and the maxillofacial surgeon is paramount ( Fig. 6 ).
Residual bony volume
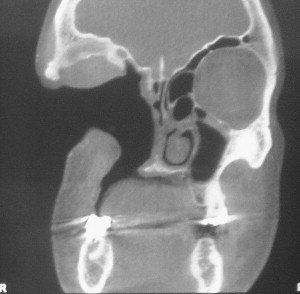
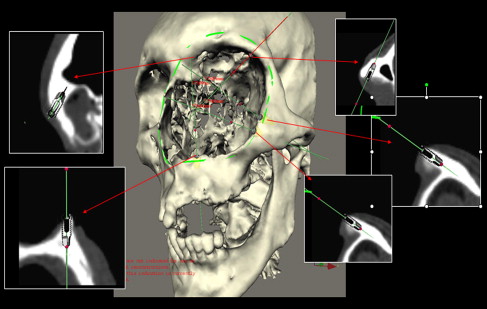
Adequate residual bony volume for the placement of maxillofacial implants is obvious. The conventional preoperative evaluation has relied on traditional radiographs and/or 2-dimensional (2D) computed tomography scans for planning the implant positions ( Figs. 7 and 8 ). The surgeons would use the limited information gained from these radiographs and, in conjunction with the intraoperative trial and error, locate at times the extremely limited bony volume for the placement of maxillofacial implants in the attempt to avoid hollow spaces, such as the frontal, ethmoidal, and maxillary sinuses. The advent of computer-guided software has allowed the adaptation of this technology for the treatment of maxillofacial defects.

The Maxillofacial Concept Software allows collaboration between the surgeon, the maxillofacial prosthodontist, and the anaplastologist in planning the treatment of the patient with the maxillofacial defects before initiation of the treatment. The 2D Digital Imaging and Communications in Medicine files of the patients are converted into 3-dimensional (3D) format, allowing better visualization of the remaining osseous tissues by the surgical team. The patient’s soft tissues can also be reformatted and superimposed onto the reconfigured 3D bony volume showing the topography of the patient’s remaining facial and or intraoral architecture.
The maxillofacial prosthodontist and the anaplastologist can evaluate the thickness of the soft tissues and guide the surgical team regarding the best location for the implants, and consequently the abutments and the prosthetic framework design.
By using the information provided by the Maxillofacial Concept Software, the team can plan the position of the implants that best complies with the surgical as well as the prosthetic principles.
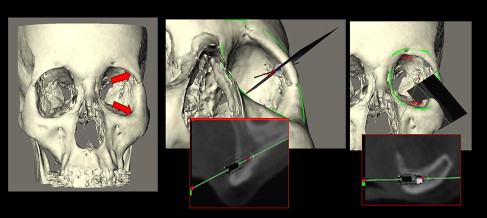
The surgical team preoperatively can now position the implants, evaluate their trajectory ( Fig. 9 ), select final abutments ( Fig. 10 ), and modify the treatment plan several times until the best implant positions for the support of the maxillofacial prosthesis leading to the most transparent transition line and aesthetic emergence profile is reached ( Fig. 11 ). The final treatment plan can then be taken to the operating room, and the virtual surgery can be translated to the actual procedure for the patient ( Fig. 12 ).
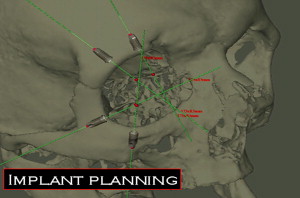
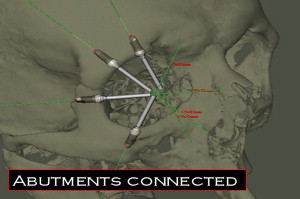

Overlying soft tissues
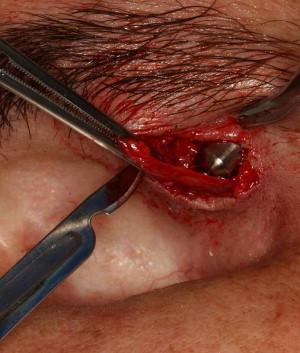
The presence of healthy overlying soft tissues is important. Whether cutaneous or mucosal tissue, the clinicians must be aware of the thickness of the tissues to allow a maintainable environment around the prosthetic substructures for the patient and the maxillofacial prosthodontist. The management of the periabutment soft tissues is very similar to the intraoral management of soft tissues surrounding abutments. Palacci flaps are performed where necessary to allow a maintainable depth of the soft tissue around the maxillofacial abutments ( Fig. 13 ).
Transition line
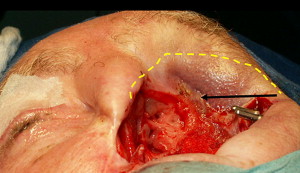
The transition line of the prosthesis with the patient’s skin should be as subtle as possible, to allow a lifelike appearance. In any type of defect, the fixtures should be placed with the planned prosthetic framework in mind. The angle of the implants, the trajectory, and its long axis during its placement should allow for an adequate emergence profile, allowing for a proper bar design without interfering with the ideal sculpturing of the prosthesis ( Fig. 14 ). Misplaced implants may cause a poor aesthetic outcome. Proper spacing and angles of the implants is also necessary to allow the manipulation of the prosthetic screws and drivers needed during the fabrication and the delivery of the maxillofacial prosthesis ( Fig. 15 ).
Surgical considerations
Armamentarium
The treatment of patients with maxillofacial defects may involve the intraoral structures. The type of implants used for the treatment of the various clinical cases should be defined.
Extraoral implants
The extraoral implants used to date are the machined-surface, regular-platform, 3.75-mm and 4.0-mm Brånemark implants manufactured by Nobel Biocare, Zurich, Switzerland. (Note: extraoral implants used for the bone-anchored hearing aid [BAHA] appliance or to support a prosthetic ear are not the regular platform Brånemark implants). The implants used for ear prostheses are specially designed implants to support the BAHA appliance.
Intraoral implants
The intraoral implants used to date are also the machined-surface, regular-platform Brånemark implants. When the zygomatic bone was used to anchor implants, several approaches were considered. For patients having had partial or total maxillectomies, regular-platform implants were used in the remaining portion of the zygomatic body to allow for the contralateral point stabilization of the prosthetic framework.
In cases for which the maxillary sinus and the maxillary residual arch were intact, the Brånemark zygomatic implant with the 45° angulated platform was used.
Preparing the osteotomy
Most cases are treated in the operating room under general anesthesia. After sterile preparation and draping of the patient, 2% lidocaine with 1:100,000 epinephrine is administered to the surgical field to allow better control of homeostasis. Sharp dissection through the epithelium, connective tissue, and the periosteum exposes the residual bony defect. The drilling sequence to prepare the osteotomy is the same as for conventional intraoral implants. The surgeon has to judge clinically the quality of the bone while preparing the osteotomy.
Orbital Defects
Placement of 2 to 3 implants is generally adequate for the support of the substructure and the orbital prosthesis. The preferred site is the lateral-supraorbital rim, if it has not been resected. However, implants may be placed in the residual periorbital bony rim.
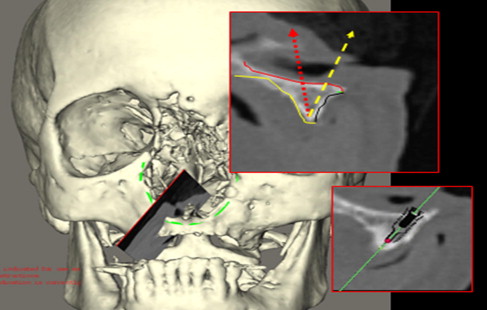
A special consideration for treatment planning of the orbital prosthesis is the available depth of the orbital defect. Inadequate orbital depths ( Fig. 16 ) do not allow the tipping of the trajectory of the implant into the center of the orbit, resulting in inadequate space for the fabrication of the substructure as well as the orbital prosthesis. Virtual planning of the implants is performed ( Fig. 17 ), and the orbital depth is established intraoperatively by debulking the orbital contents ( Fig. 18 ). The implants are placed as planned at 35 N cm ( Fig. 19 ). The immediate postoperative implant positioning is in concert with the proposed virtual positioning of the implants ( Figs. 20 and 21 ).
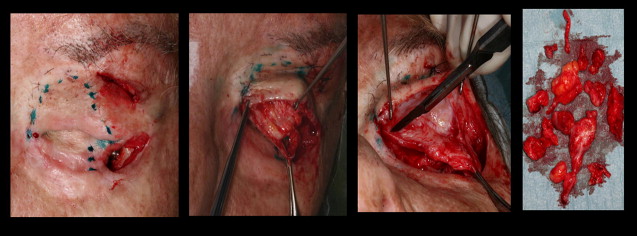
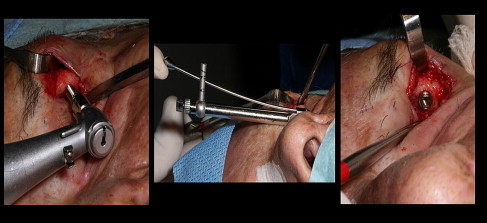
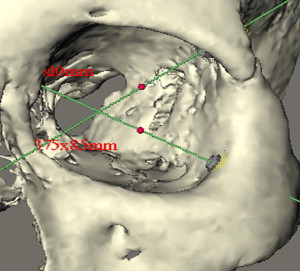
The computer-guided treatment planning allows for the positioning of the implants in the appropriate bony volume as well as with the proper trajectory for reconstruction of the maxillofacial prosthesis ( Fig. 22 ). After exposure of the orbital rim, a round bur is used to identify the position of the implant ( Fig. 23 ). A paralleling pin is placed on completion of the 2-mm osteotomy to better evaluate the trajectory of the implants ( Fig. 24 ). The final osteotomy size is 3 mm, to be followed by the insertion of the implant. Most procedures follow the 1-step protocol whereby the temporary healing abutment is placed at the time of the implant surgery with adaptation of the soft tissues around the temporary healing abutment ( Figs. 25 and 26 ). Once the second planned implant is placed, the position of the actual surgery can be compared with the position of the implants in the virtual surgery using the computer-guided treatment-planning software. Triple antibiotic ointment and pressure dressings complete the surgical treatment ( Figs. 27 and 28 ). An osseointegration period of 4 to 6 months is allowed, followed by the fabrication of the substructure and the prosthetic eye ( Figs. 29 and 30 ).




