Fig. 7.1
Clinical growth pattern
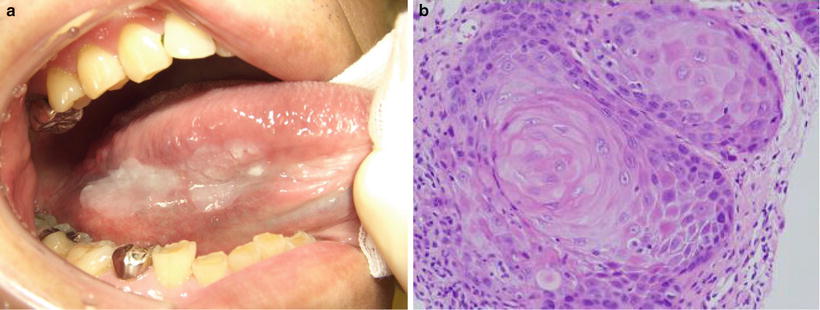
Fig. 7.2
Clinical growth pattern: superficial type of tongue, (a) clinical finding, (b) histological finding; squamous cell carcinoma. Tumor shows superficial growth
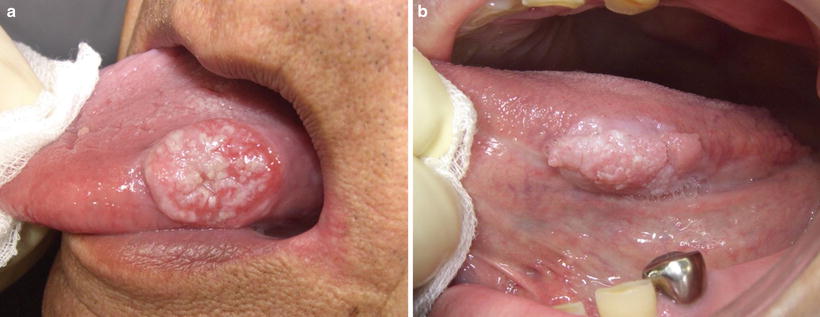
Fig. 7.3
Clinical growth pattern; exophytic type of tongue. Tumor shows exophytic growth
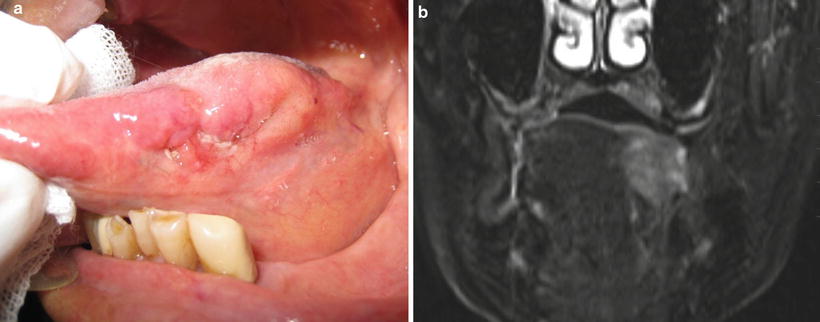
Fig. 7.4
Clinical growth pattern: endophytic type of tongue. (a) Clinical finding, (b) MRI finding. Tumor shows endophytic growth

Fig. 7.5
Mode of tumor invasion
It is necessary to judge a tumor’s progression state and characteristics accurately and to determine the surgical margin using the above-described kinds of information before performing surgical therapy. Practitioners must also predict the functional impairments resulting from resection and furthermore consider reconstruction methods to address them.
7.2 The Safety Margin During Tumor Resection
The oral cavity is located adjacent to vital organs, and hard and soft tissues are mixed in it to form a complex shape. Thus, determining the surgical margin of oral cancer is not simple. There are many reports concerning the establishment of the surgical margin [16–23]. With a safety margin of 5 mm or less from the tumor, the postoperative local recurrence rate is no different from cases with a positive margin [23]. However, the recurrence rate for a margin of 5 mm or less has been observed to be five times higher than a margin of over 5 mm (10 %) [20], and samples show that local tumor recurrence rate drops reliably when there is a distance of 5 mm or more from the tumor margin. From these reports, a safety margin of 10 mm or more at the time of resection can be considered reasonable [21, 24–26]. However, in the oral cavity, local recurrence occurs at a higher frequency for the endophytic and superficial types of clinical growth patterns compared with the exophytic type [1]. Additionally, since recurrence tendencies are high for cases where the histopathological mode of tumor invasion is bad, it is necessary to consider various factors when establishing the safety margin [14–16]. Furthermore, it is important to perform a suitable mandibulectomy in accordance with the invasion into the bone in cases where the oral cancer is adjacent to the mandible [17, 18].
7.3 Vital Staining with Iodine Solution
Vital staining with iodine solution has become an indispensable auxiliary diagnostic method for the detection of early-stage cancer and it is widely used in the upper gastrointestinal region. In the oral cavity, iodine staining allows visualization of epithelial dysplasia and can identify malignant and potentially malignant lesions [27–30] (Figs. 7.6 and 7.7). Further, the accuracy of iodine staining compared with visual examination in discriminating between normal and epithelial dysplastic oral mucosa has been demonstrated [28–31] (Fig. 7.8). In an examination of primary lesion recurrence rate in cases of partial resection that considered the presence or absence of iodine staining, the group that utilized iodine staining was shown to have a significantly lower primary lesion recurrence rate. Consistent with these results, it has also been reported that, for superficial T1 or early T2 lesions, medical cases for which the surgical margin was determined by performing vital staining with iodine solution had a clearly lower local recurrence rate compared with cases where it was not performed (Fig. 7.9). Hence, determining the surgical margin by performing vital staining with iodine solution is useful for superficial lesions. However, there are also cases where the unstained area covers a large range, and so judgments of where to resect to can be difficult for such cases [32, 33] (Figs. 7.10 and 7.11).
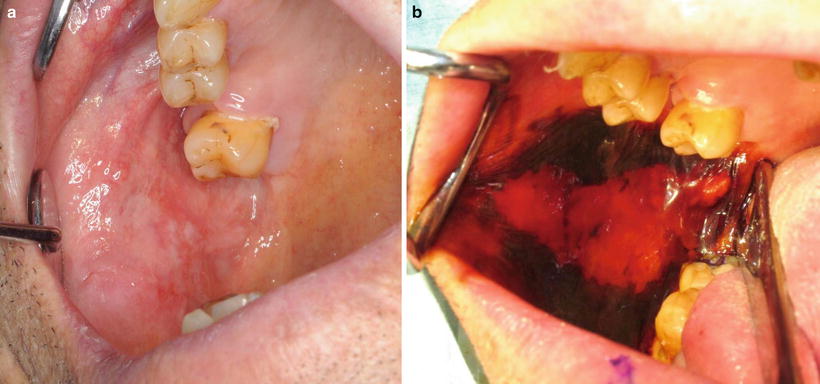
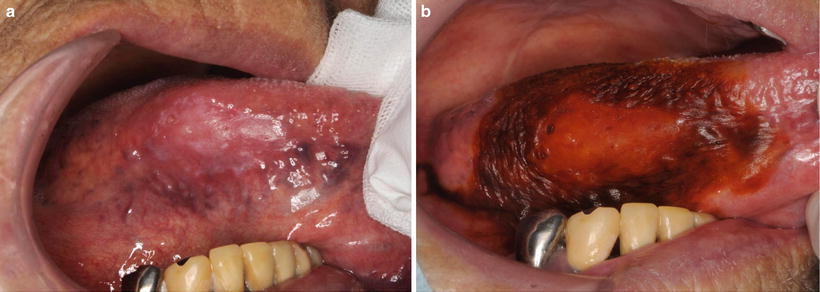
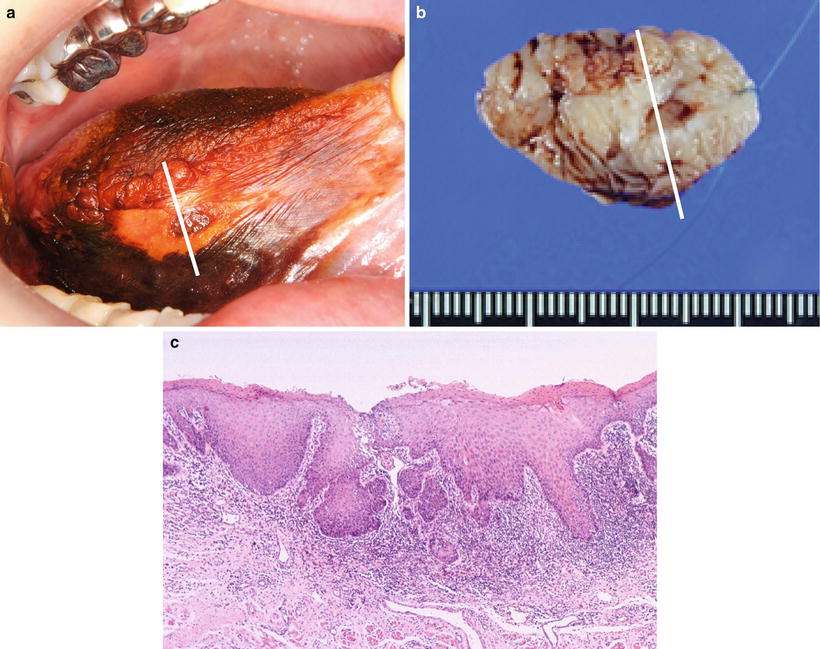
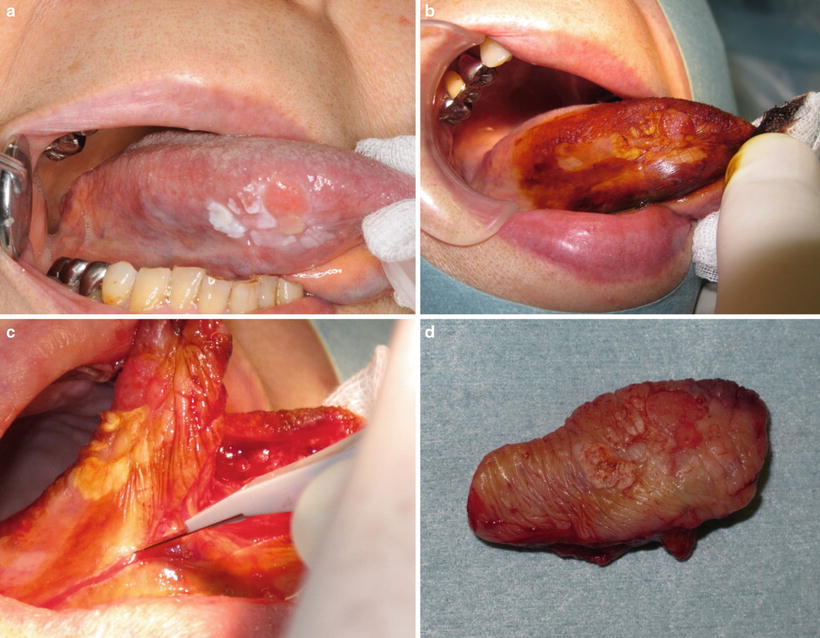
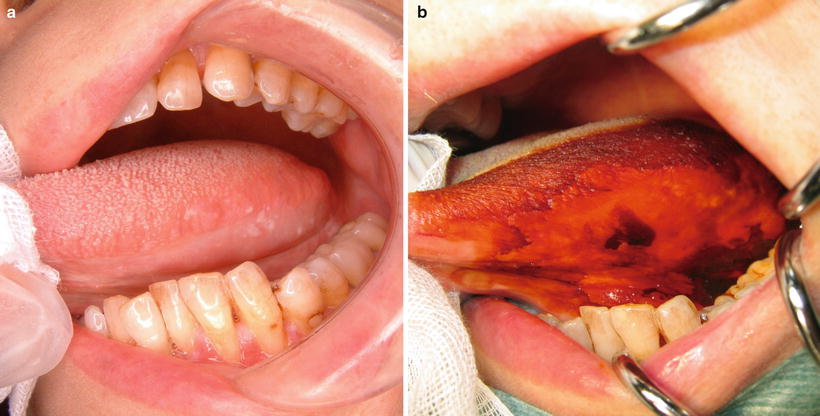
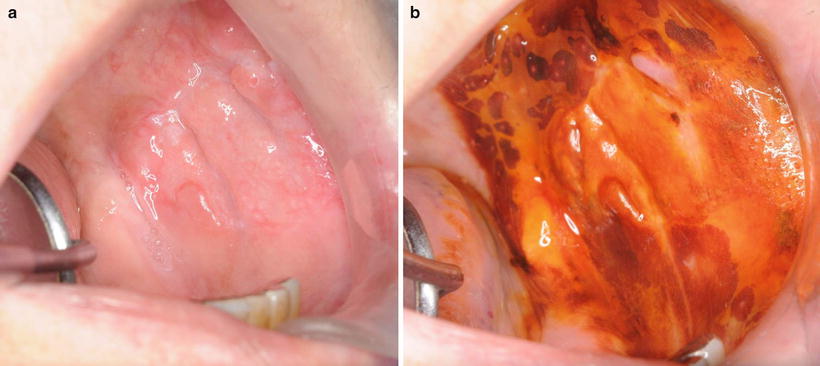

Fig. 7.6
Vital staining with iodine solution: (a) prestaining and (b) poststaining. Dysplastic lesion of buccal mucosais unstained

Fig. 7.7
Vital staining with iodine solution: (a) prestaining and (b) poststaining. Dysplastic lesion is unstained

Fig. 7.8
Vital staining with iodine solution. (a) Precancerous lesion is unstained; (b) surgical material. (c) HE staining; early invasive carcinoma is found a part of the epithelial dysplasia

Fig. 7.9
Vital staining with iodine solution: (a) early carcinoma with precancerous lesion, (b) vital staining with iodine solution, (c) excision of lesion, and (d) removal tissue

Fig. 7.10
Vital staining with iodine solution: (a) prestaining and (b) poststaining. The unstained area covers a large range

Fig. 7.11
Vital staining with iodine solution. (a) Prestaining and (b) poststaining. The unstained area of buccal mucosa covers a large and irregular range
7.4 Tongue Carcinomas
Looking at the tongue anatomically, it is organized into the intrinsic and extrinsic muscles of the tongue. The intrinsic muscles are the superior longitudinal muscle, the inferior longitudinal muscle, the transverse muscle, and the vertical muscle; the extrinsic muscles are the genioglossus muscle, the hyoglossus muscle, and the styloglossus muscle. For surgery, the practitioner judges the invasion status of these muscle groups and the tumor to determine the surgical margin. Also, resection at a shallow depth is not problematic for tumors with superficial or exophytic clinical growth patterns, but performing vital staining with iodine solution is useful to judge the extent of dysplastic epithelium in the tumor periphery. When resecting endophytic tumors, careful attention should be paid to the deep parts of the surgical margin [17]. It is commonly necessary to resect not only the intrinsic but also the extrinsic muscles of the tongue. Furthermore, the tongue is adjacent to the floor of the mouth, the lower gingiva and alveolus, and the buccal mucosa; because a tumor can easily invade these tissues, accurate diagnosis of the extent of tumor progression at the time of surgery is important [34–38]. The histopathological malignancy of such tumors must also be considered (Fig. 7.12). It is unnecessary to take a wide safety margin for tumors with low malignancy, but a safety margin of 1 cm or more must be taken for those with high malignancy or invasiveness. Therefore, the surgical margin for a tongue carcinoma will change depending on the primary lesion’s size, the depth of invasion [17], and the invasive state and histopathological malignancy with regard to the floor of the mouth [34], the base of the tongue, and the mandible [35–38]. Furthermore, it may be necessary to perform neck dissection concurrently via a pull-through operation because tongue carcinomas result in a high frequency of cervical lymph node metastasis.
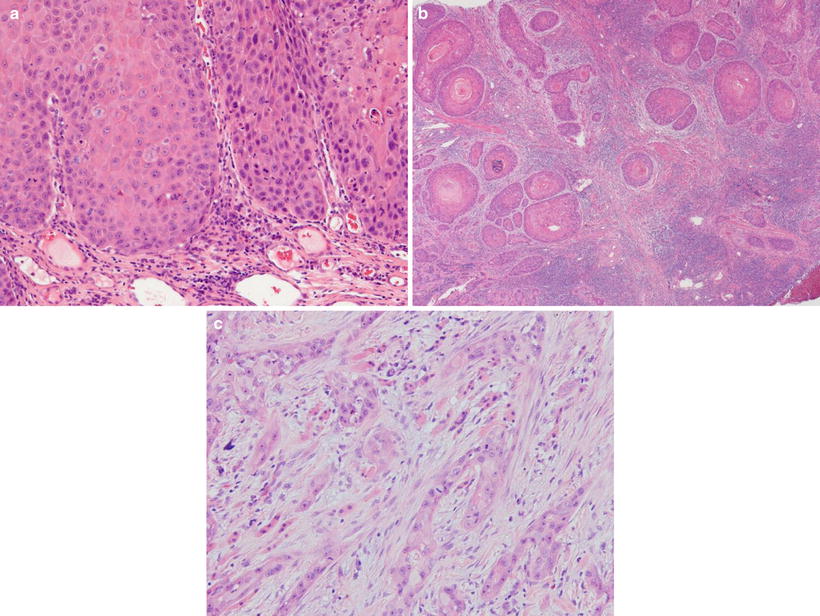

Fig. 7.12
Mode of tumor invasion: (a) YK-2, cords, less marked borderline; (b) YK-3, groups of cells, no distinct borderline; and (c) YK-4C, diffuse invasion, cord-like-type invasion
Footnote
A “pull-through operation” refers to the surgical technique in which—in consideration of the lymph vessels running from the primary lesion to the submandibular tissue or metastatic lymph nodes of the cervical region—the primary lesion is resected en bloc with dissected cervical tissue and pulled out from the submandibular region through the floor of the mouth [39–41] (Figs. 7.13 and 7.14). A pull-through operation is conducted in addition to resection of the primary lesion in cases of N1 and N2b carcinomas of the tongue or floor of the mouth, cases of cervical metastatic lymph node metastasis with multiple lesions, cases of distant metastasis, and N2a/N3 cases. On the other hand, if the primary lesion is late T2 (3–4 cm) or greater and resection through the intraoral approach is difficult, the pull-through operation can be conducted at the same time as neck dissection [42, 45]. For T1 or early T2 at N0, resection of the primary lesion through the intraoral approach will not be problematic for the most part. However, when neck dissection is conducted concurrently because cervical metastasis is suspected upon imaging, the appropriateness of the pull-through operation becomes an issue. The majority opinion mostly supports conducting the pull-through operation, but there are also those who deem it unnecessary.
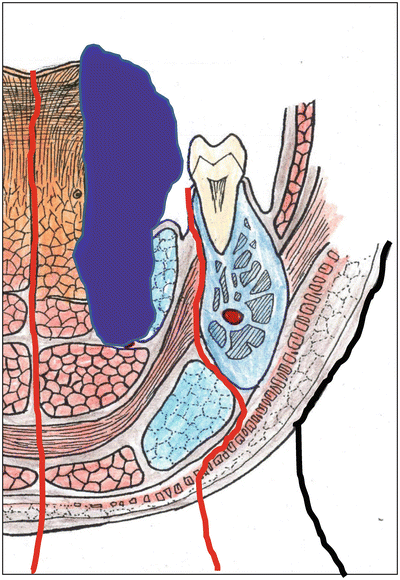
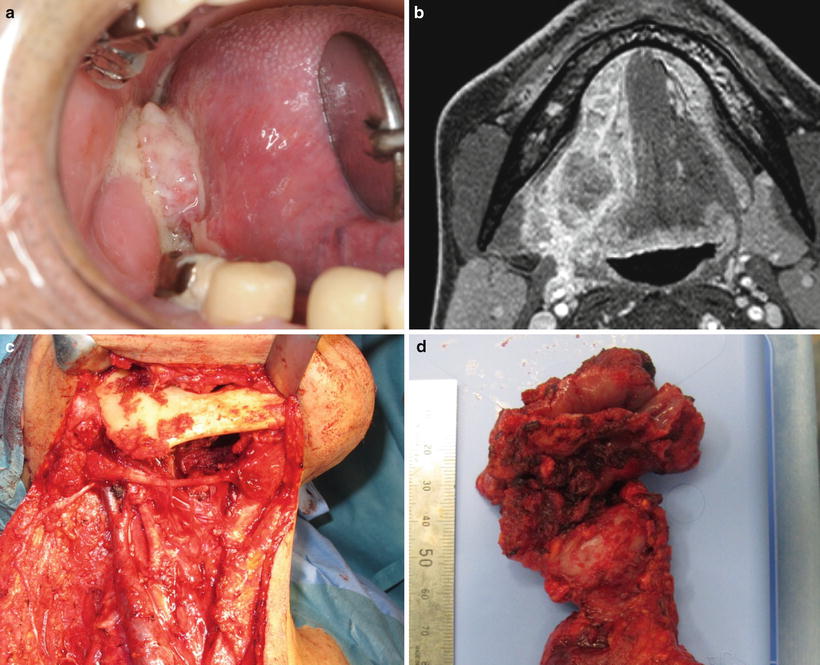

Fig. 7.13
Schema of pull-through operation; primary lesion is resected en bloc with dissected cervical tissue

Fig. 7.14
Pull-through operation. (a) Tongue carcinoma (T2N1M0) and (b) MRI; tumor occupies the part of less than half of the oral tongue. (c) Hemiglossectomy with radical neck dissection. (d) Primary lesion is resected en bloc with dissected cervical tissue
7.4.1 Surgical Resection of Tongue Carcinomas (Figs. 7.15 and 7.16)
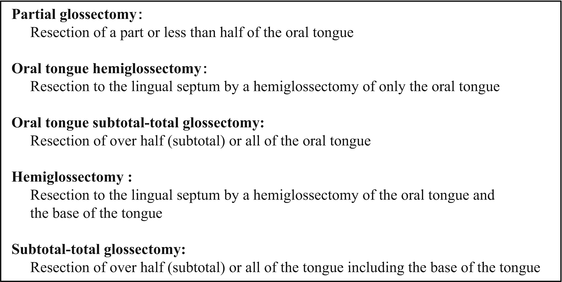
Fig. 7.15
Surgical resection of tongue carcinomas
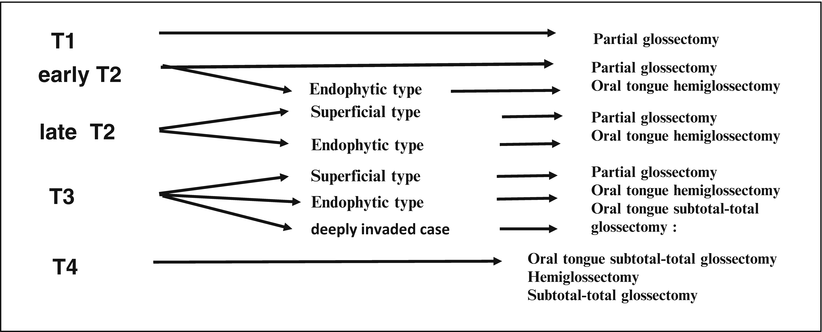
Fig. 7.16
Algorithm of the surgical treatment of tongue carcinoma
1.
Partial Glossectomy Performed for T1/Early T2 N0
Partial glossectomy refers to the resection of a part or less than half of the oral tongue (Fig. 7.17). The surgery is performed via the intraoral approach. It is important to bear in mind not only the horizontal safety margin but also the vertical safety margin at the time of resection. It is also necessary to perform resection taking into account the tumor’s clinical growth pattern and histopathological malignancy. Resection of deep muscle tissue will be considerable for tumors that have deeply invaded the muscle layers. Hence, sufficient postoperative hemostasis on the wound and primary suturing are conducted (Fig. 7.18). A skin graft should be performed when there is a possibility that suturing will cause functional disturbance of the tongue. Impairment of oral function after partial glossectomy is mild.
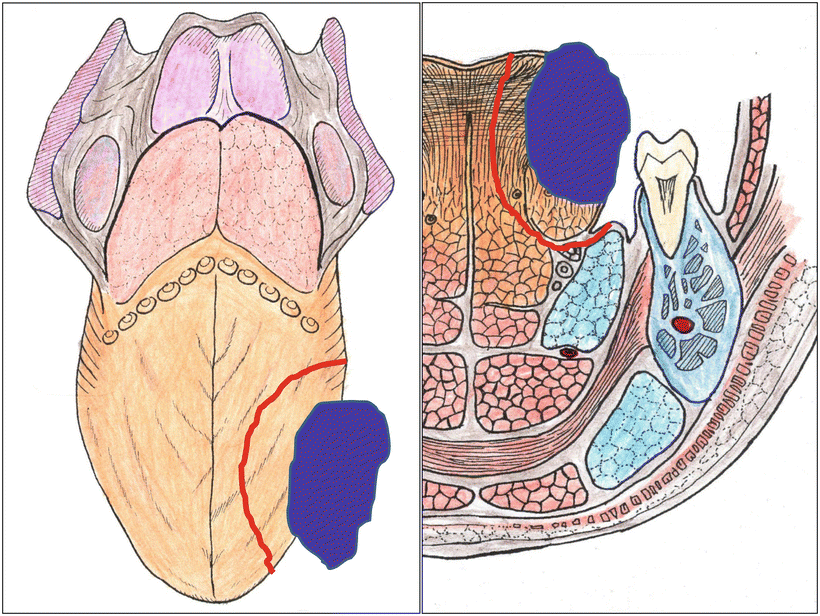
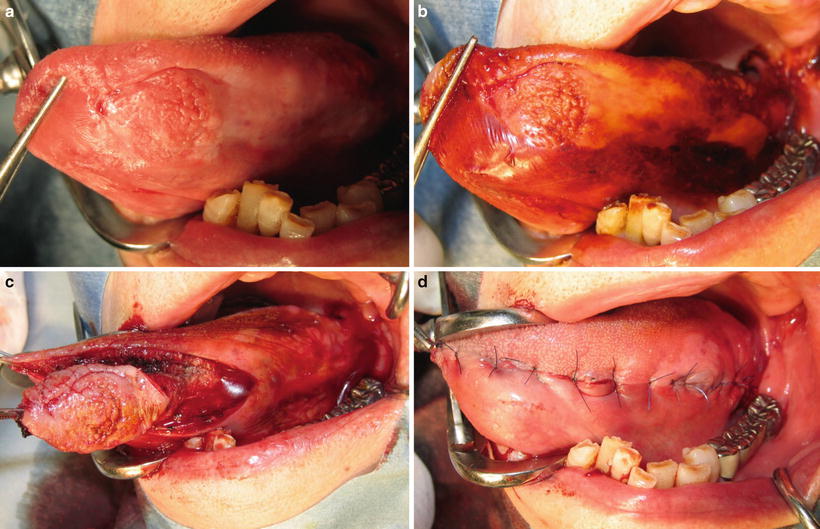

Fig. 7.17
Schema of partial glossectomy

Fig. 7.18
Partial glossectomy. (a) Tongue carcinoma (T1), (b) vital staining with iodine solution, (c) partial glossectomy, and (d) primary suturing
Note: Sometimes the term “early T2” is used for tongue carcinomas with a maximum diameter of between 2 and 3 cm, and the term “late T2” (or “advanced T2”) for those with a maximum diameter of between 3 and 4 cm; for tongue carcinomas there are some who consider the classification significant.
2.
Partial Glossectomy Performed for Late T2/T3 (Superficial Type)
Because the surgical margin and resection depth become deep for these tumors, it is important that the safety margin be sufficiently verified. There are also cases where primary suturing cannot be performed, and a skin graft or, depending on the case, flap transplantation becomes necessary (Fig. 7.19). There are also cases where a safe resection cannot be made via the intraoral approach, and surgery must be performed via the extraoral approach.
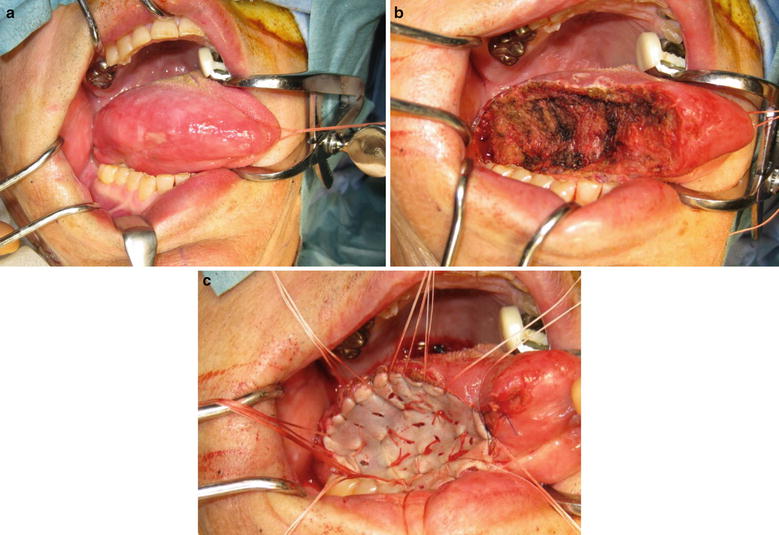

Fig. 7.19
Partial glossectomy. (a) Tongue carcinoma (T2), (b) partial glossectomy, and (c) surgical defect is covered with shin graft
3.
Partial Glossectomy, Oral Tongue Hemiglossectomy, Oral Tongue Subtotal-Total Glossectomy, Hemiglossectomy, and Subtotal-Total Glossectomy Performed for Late T2/T3/T4 N0
Oral tongue hemiglossectomy refers to a resection to the lingual septum by a hemiglossectomy of only the oral tongue (Figs. 7.20 and 7.21). Oral tongue subtotal-total glossectomy refers to a resection of over half (subtotal) or all of the oral tongue (Fig. 7.22). Hemiglossectomy refers to a hemiglossectomy that includes the base of the tongue (Figs. 7.23 and 7.24). Subtotal-total glossectomy refers to a resection of over half (subtotal) or all of the tongue including the base of the tongue (Fig. 7.25).
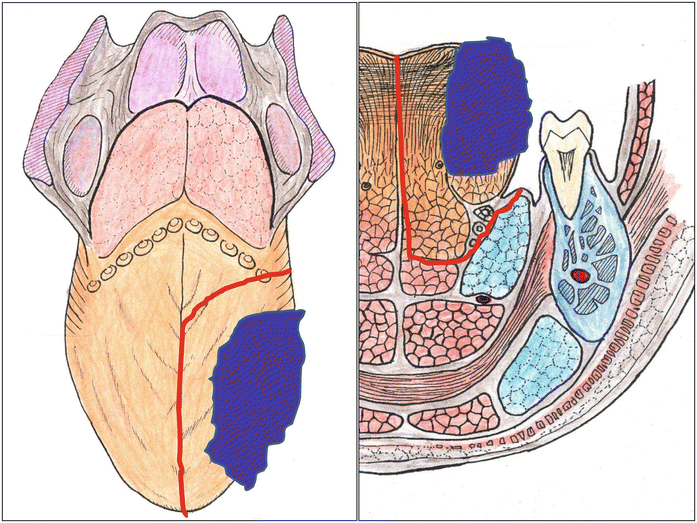
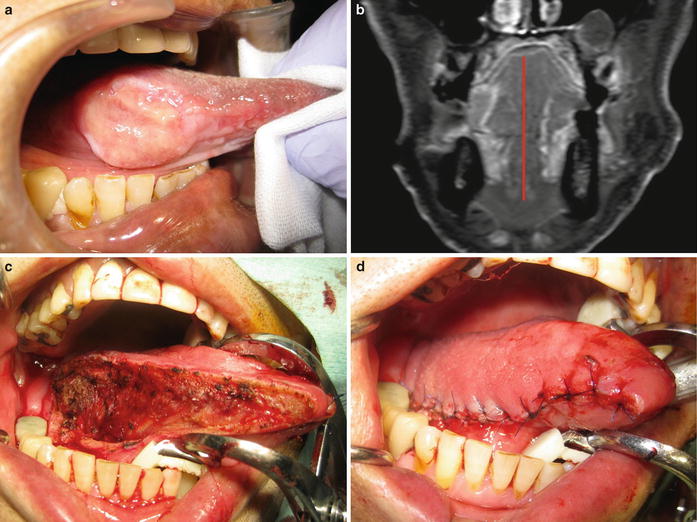
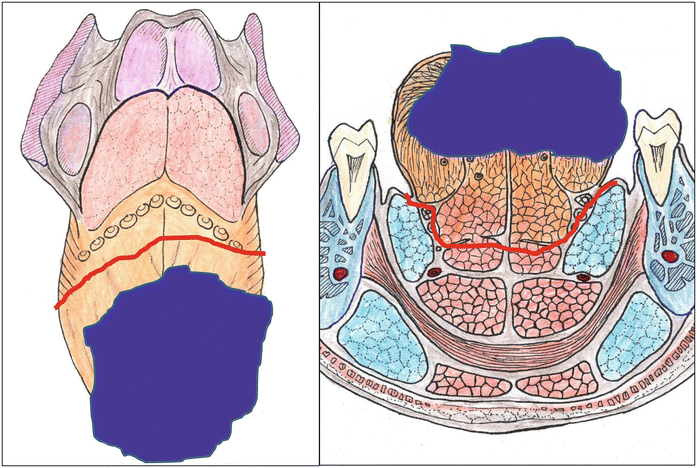
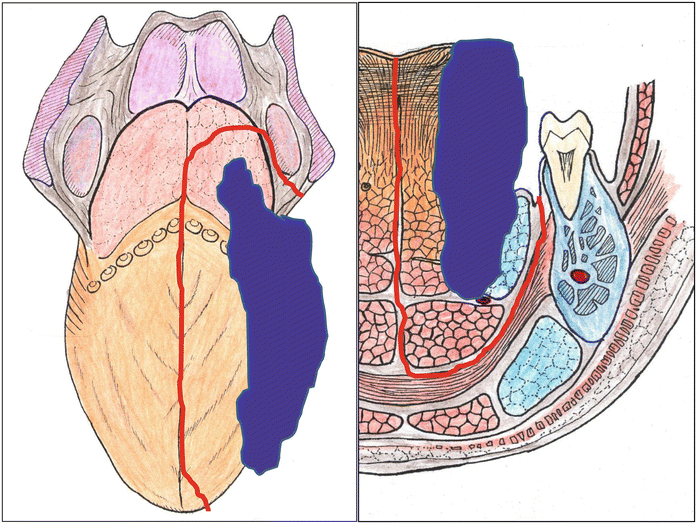
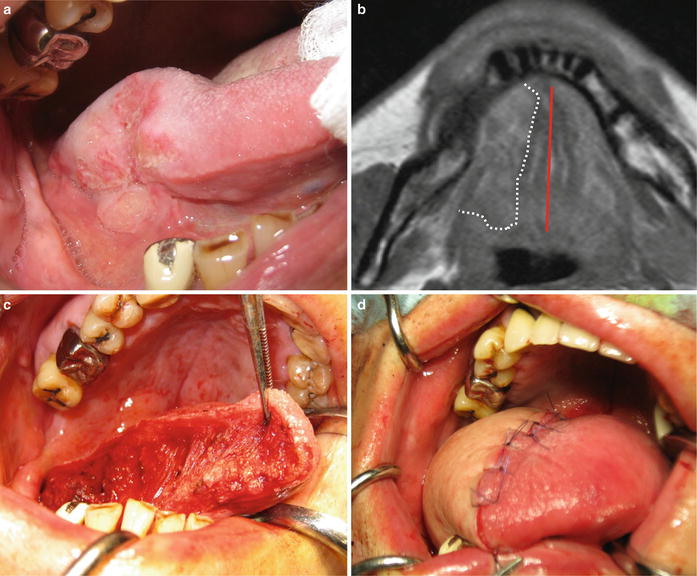
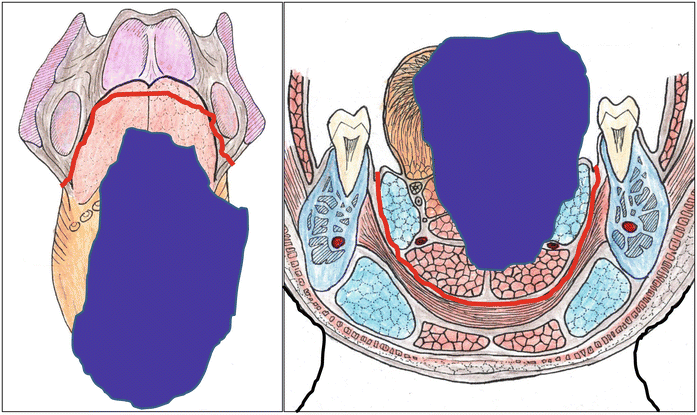

Fig. 7.20
Hemiglossectomy of the oral tongue

Fig. 7.21
Hemiglossectomy of the oral tongue. (a) Tongue carcinoma (T2) and (b) MRI; tumor occupies the part of less than half of the oral tongue. (c) Resection to the lingual septum of the oral tongue. (d) Primary suturing

Fig. 7.22
Subtotal-total glossectomy of the oral tongue. Resection of over half (subtotal) or all of the oral tongue

Fig. 7.23
Hemiglossectomy that includes the base of the tongue

Fig. 7.24
Hemiglossectomy. (a) Tongue carcinoma (T3) and (b) MRI; tumor includes the base of the tongue. (c) Resection to the lingual septum by a hemiglossectomy with base of the tongue. (d) Reconstruction with forearm flap

Fig. 7.25
Subtotal-total glossectomy: resection of over half (subtotal) or all of the tongue including the base of the tongue
There are cases where performing these resections via the intraoral approach is possible, but performing them via the extraoral approach is preferable in order to reliably ensure the safety margin of the deep parts of the tumor.
For cases in which the extraoral approach is used, preventative neck dissection is sometimes performed concurrently (via the pull-through method) to sufficiently resect deep tissue or to accompany soft tissue reconstructive surgery [42–45].
Oral tongue hemiglossectomy method is as follows. (1) From the neck, use the fingers to separate the left and right geniohyoid muscles and genioglossus muscles and divide the lingual septum into left and right. (2) From inside the oral cavity, make an incision from the exact center of the floor of the mouth to the exact center of the tongue and detach the lingual septum. (3) From the neck, cut the geniohyoid and mylohyoid muscles on the affected side near the submental hyoid bone. (4) From inside the oral cavity, detaching parallel with the lingual septum in the center of the tongue dorsum, cut the intrinsic muscles of the tongue and the styloglossus muscle posteriorly, cut the genioglossus muscle anteriorly, and resect the floor of the mouth.
4.
Partial Glossectomy Performed Concurrently with Neck Dissection for Cases of T1/Early T2 N1–3 (via the Pull-Through Method)
For late T2/T3/T4N1–3, neck dissection is performed concurrently with resection of the primary lesion. This involves a compound operation of partial glossectomy, hemiglossectomy, oral tongue subtotal-total glossectomy, etc., and the mandible and surrounding tissues, depending on the extent of tumor invasion. Concerning tumor progression towards the mandible, if the distance of the tumor from the mandible is 1 cm or more, then a mandibulectomy is not necessary5.
5.
Surgical Treatment of T4 Cases [46–48]
(a)
In cases of invasion into the floor of the mouth or into the mandibular bone marrow, the practitioner resects the mandible while concurrently ensuring the safety margin. One performs a marginal mandibulectomy if the tumor has yet to reach the mylohyoid muscle. In this situation, if the tumor is limited to the cortical bone on the lingual side of the mandible, one resects the cortical bone on the lingual side by sagittal splitting osteotomy (Fig. 7.26). One resects the upper portion of the bone if the tumor has only infiltrated the upper portion of the mandible (Fig. 7.27).
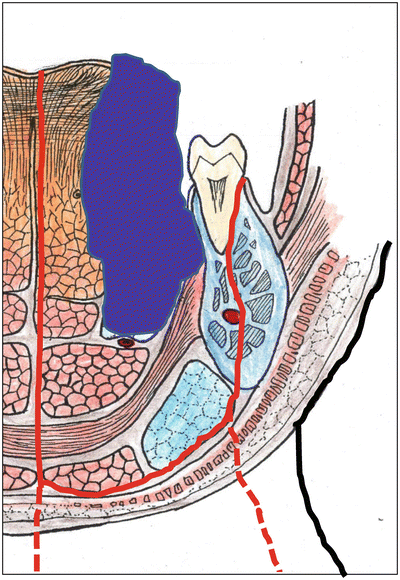
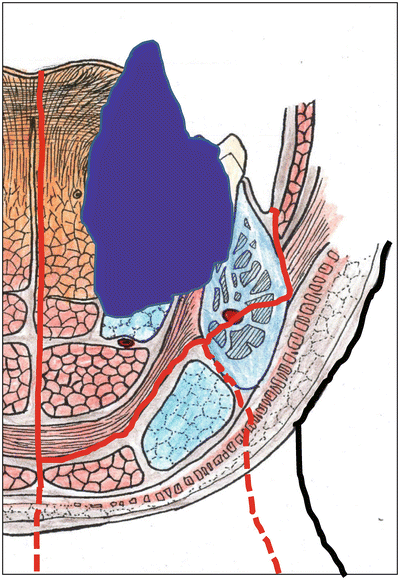

Fig. 7.26
Command operation. Hemiglossectomy with the cortical bone on the lingual side by sagittal splitting osteotomy and/or neck dissection

Fig. 7.27
Command operation. Hemiglossectomy with marginal mandibulectomy and/or neck dissection
A segmental mandibulectomy is performed in cases where the tumor exceeds the mylohyoid muscle or invades into the mandibular bone marrow, into the submandibular space, or into the extrinsic muscles of the tongue (Figs. 7.28, 7.29, and 7.30).
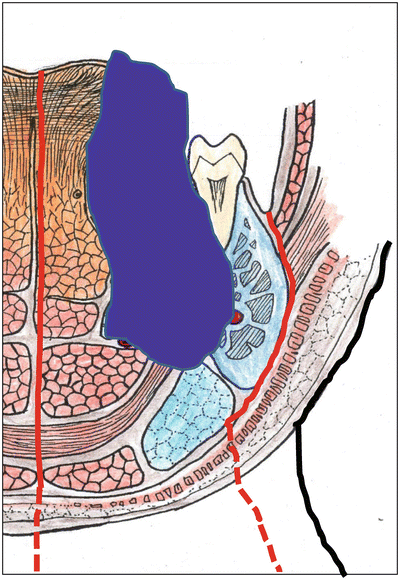
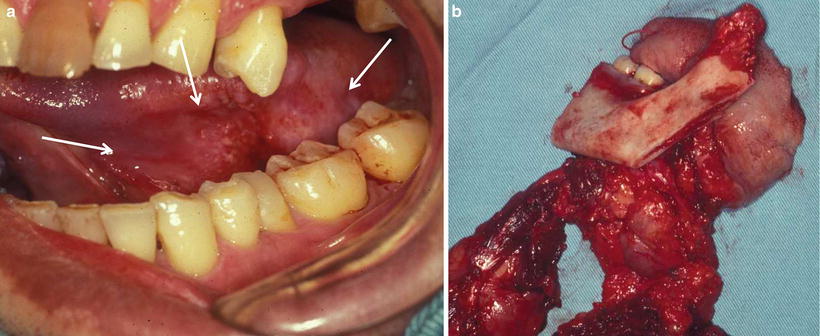
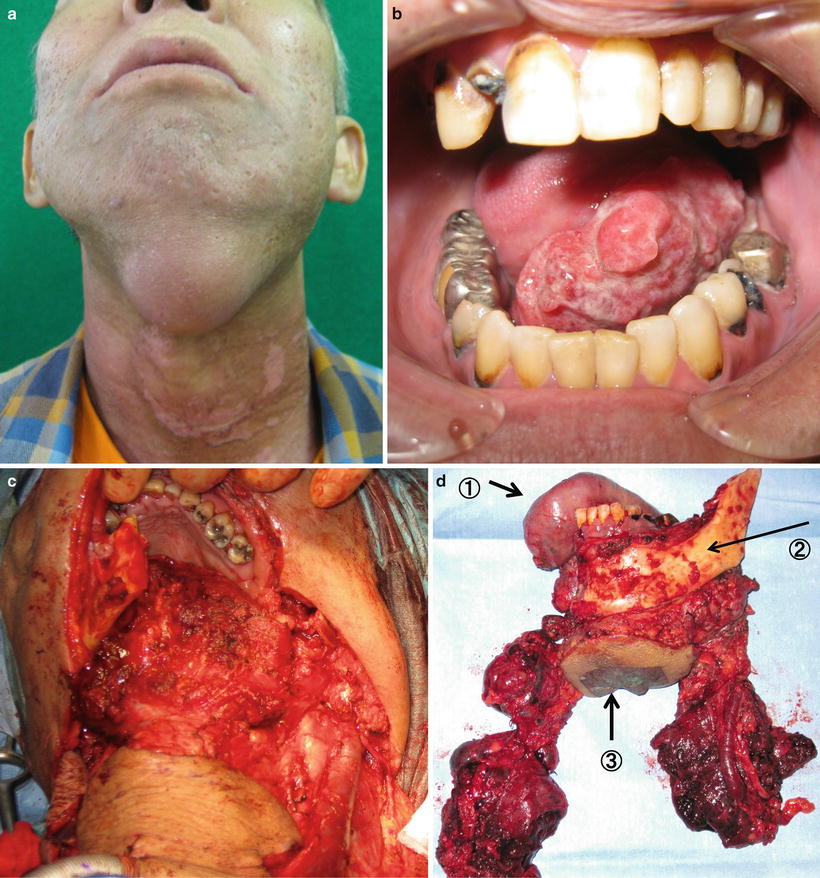

Fig. 7.28
Command operation. Hemiglossectomy with segmental mandibulectomy and/or neck dissection

Fig. 7.29
Command operation of tongue carcinoma. (a) Tongue carcinoma invades the mandible (T4N2cM0). (b) Total glossectomy with segmental mandibulectomy and bilateral neck dissection

Fig. 7.30
Tongue carcinoma with mandibular invasion and cervical metastasis: (a) cervical metastasis and (b) tongue carcinoma invades into the mandible. (c) Hemiglossectomy, segmental mandibulectomy, bilateral neck dissection, and skin resection of submental region. (d) Surgical specimen: (1) tongue, (2) mandible, and (3) skin
(b)
In cases involving invasion posterior to the lingual margin to the base of the tongue, the base of the tongue, the palatoglossal arch, and the tonsillar fossa are resected in addition to a total glossectomy. Furthermore, if the tumor has invaded inferiorly, then a laryngectomy is also performed (Fig. 7.31).
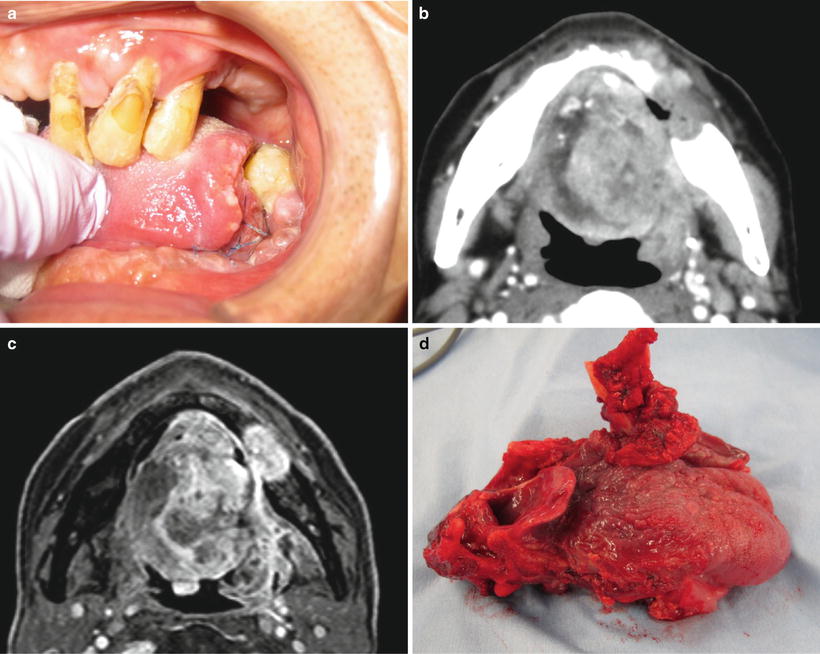

Fig. 7.31
Total glossectomy. (a) Tongue carcinoma (T4) and (b) CT; tumor invades the base of the tongue and mandible. (c) MRI; tumor invades the base of the tongue and mandible. (d) Total glossectomy with laryngectomy
Determining the surgery indicated is difficult in cases where invasion is observed in the masticatory muscle space, the pterygoid process, the base of the skull, or the internal carotid artery and its surroundings.
7.5 Surgical Therapy for Lower Gingival Carcinomas
Lower gingival carcinomas invade the mandible relatively quickly in many cases, causing bone destruction. In the establishment of surgical margins, because the tumor progression into the bone cannot be directly probed physically, it is diagnosed via diagnostic imaging techniques such as dental X-ray, panoramic X-ray, CT, and MRI scans. It is important to have a complete picture of the depth and type of bone resorption and invasion to adjacent structures and to establish a treatment plan [49–51].
7.5.1 Surgical Methods for Lower Gingival Carcinomas (Fig. 7.32)

Fig. 7.32
Surgical resection of lower gingival carcinoma
Concerning the treatment standards for T4 in “General Rules for Clinical and Pathological Studies on Oral Cancer” the T classification is currently widely utilized as the diagnostic criteria of the mandibular canal in Japan [1]: i.e., an invasion reaching the mandibular canal is classified as T4. This differs from the UICC classification, whereby T4 refers to invasion beyond the alveolar bone. Surgical treatment of a lower gingival carcinoma in principle requires a mandibulectomy, and the basic operative procedure differs depending on the surgical margin.
A gingivectomy is a method during which only the gingival mucosa and periosteum are resected, and the alveolar bone is not. This method is indicated for precancerous lesions and in situ carcinoma of the gingiva.
Marginal mandibulectomy refers to a resection of the only lingual cortex and/or alveolar process (Figs. 7.33, 7.34, and 7.35). Segmental mandibulectomy refers to a resection of under half (subtotal) of the mandible without condyle (Figs. 7.36 and 7.37). Hemi-mandibulectomy refers to a resection of half of the mandible with one-side condyle (Fig. 7.38). Subtotal mandibulectomy(with/without condylectomy)refers to a resection of over half (subtotal) of the mandible with/without condyle (Fig. 7.39). Total mandibulectomy refers to a resection of all of the mandible including the both side condyle.
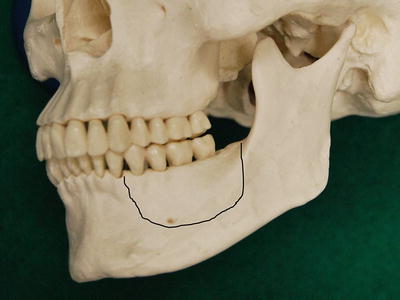
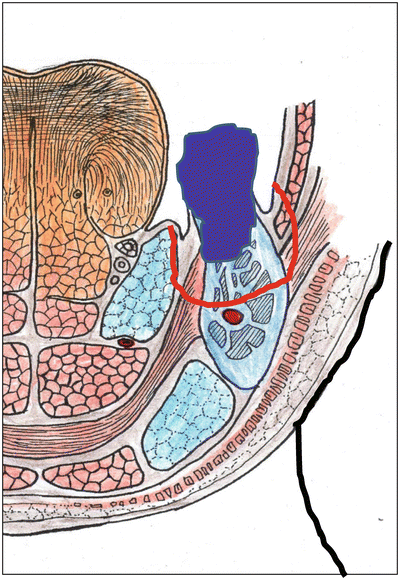
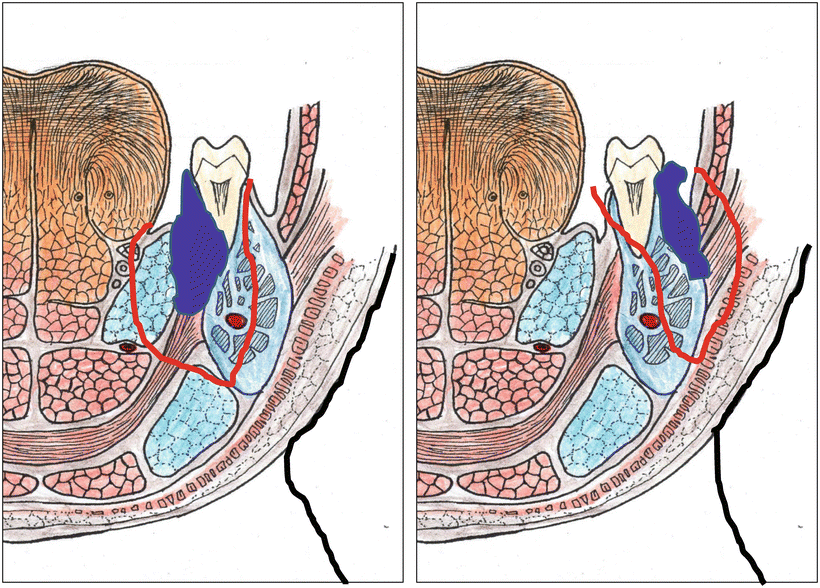
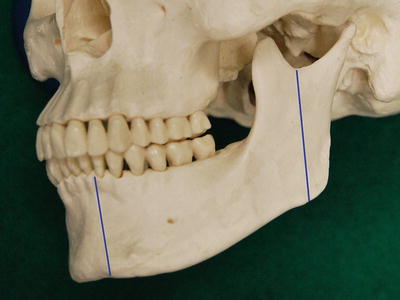
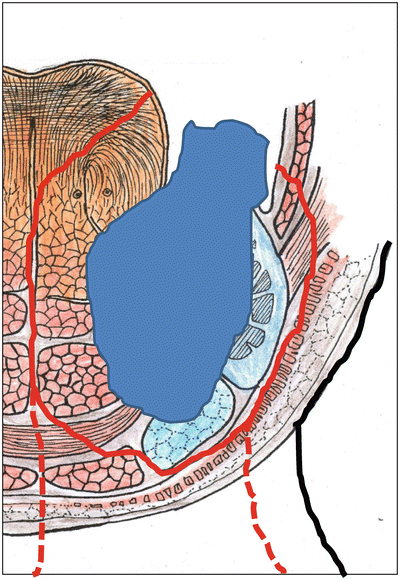
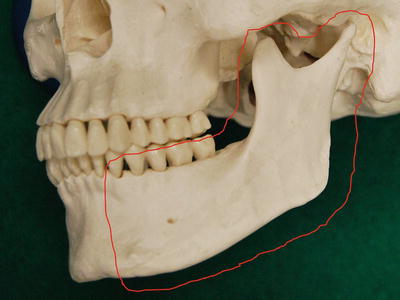
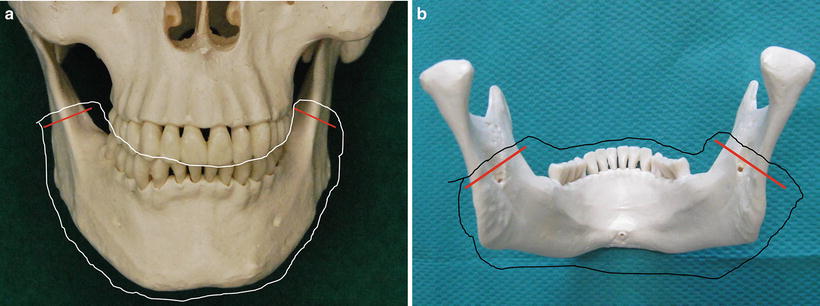

Fig. 7.33
Surgical resection of lower gingival carcinoma. Marginal mandibulectomy

Fig. 7.34
Schema of marginal mandibulectomy

Fig. 7.35
Schema of marginal mandibulectomy (sagittal splitting)

Fig. 7.36
Surgical resection of lower gingival carcinoma. Segmental mandibulectomy

Fig. 7.37
Schema of segmental mandibulectomy

Fig. 7.38
Surgical resection of lower gingival carcinoma. Hemi-mandibulectomy

Fig. 7.39
Subtotal mandibulectomy
Marginal mandibulectomy is performed for cases of T1 where the invasion into the bone is light. For T2 and T3, the relationship between mandibular resorption type (Fig. 7.40) and depth is especially important [52]. In these cases, marginal resection or segmental resection of the mandible can be chosen, but that judgment is determined by the extent of tumor progression [42–45, 49–52] (Figs. 7.41, 7.42, 7.43, and 7.44).
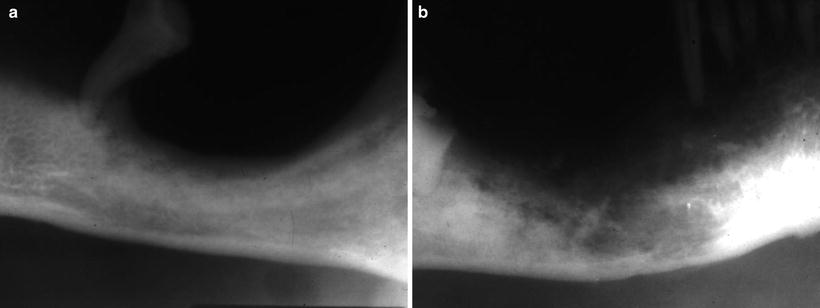
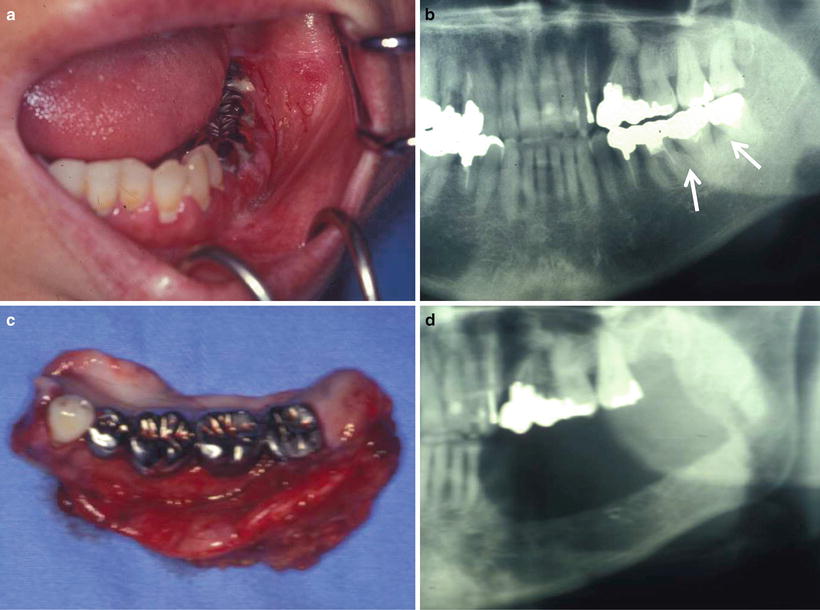
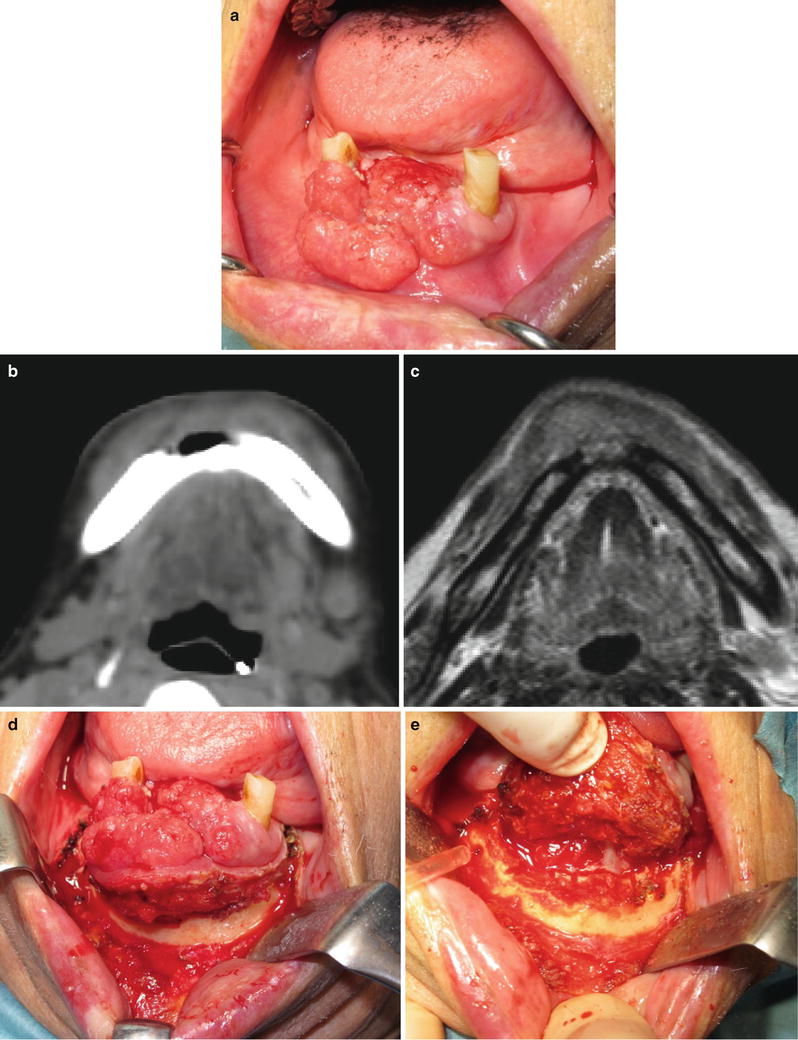
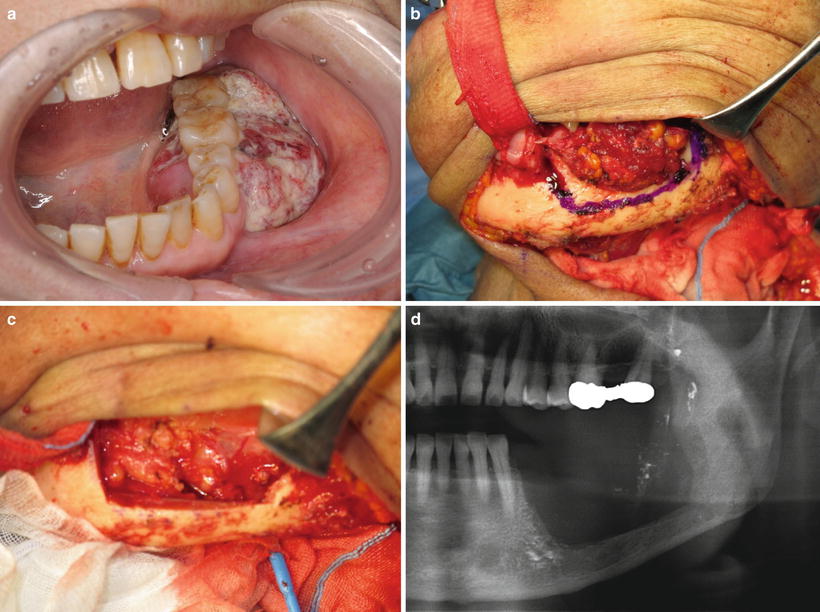
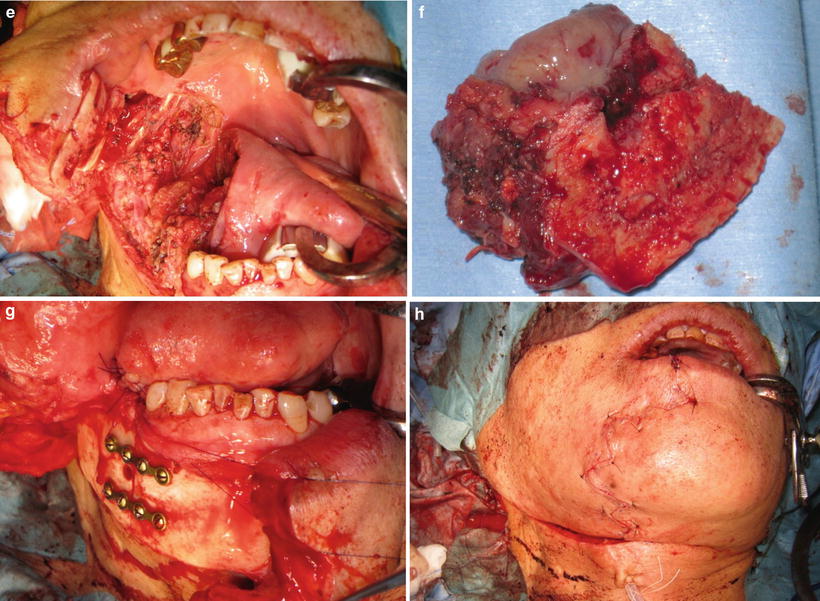

Fig. 7.40
Mandibular absorption type. (a) Expansive type and (b) invasive type

Fig. 7.41
Marginal mandibulectomy. (a) Clinical finding, gingival carcinoma (T2N0M0). (b) Tumor invades only the alveolar bone. (c) Surgical specimen. (d) X-ray finding

Fig. 7.42
Marginal mandibulectomy (intraoral approach). (a) Clinical finding; gingival carcinoma (T2N0M0). (b) CT finding; tumor invades just the alveolar bone. (c) MRI finding; tumor invasion remains in the alveolar bone. (d) Resection of soft tissue. (e) Removal of the tumor with alveolar bone

Fig. 7.43
Marginal mandibulectomy (extraoral approach). (a) Clinical finding; gingival carcinoma (T3N0M0). (b) Cut line for marginal mandibulectomy. (c) Removal of tumor (marginal mandibulectomy). (d) X-ray finding


Fig. 7.44
Marginal mandibulectomy (sagittal splitting). (a) Clinical finding; lower gingival carcinoma. (b) Excision line on the lower lips. (c) Sagittal splitting of mandible. (d) Removal of tumor that invades into the tongue. (e) Tumor is removed. (f) Removal of tissue. (g) Bone fixation by miniplate. (h) Postoperative condition
In cases of T4a, for invasions reaching the mandibular canal, into the buccal space or subcutaneous fat, into the submandibular space, and into the extrinsic muscles of the tongue, segmental resection (Fig. 7.45), hemi-mandibulectomy (Fig. 7.46), subtotal mandibulectomy, and total mandibulectomy can be performed. For these cases, making an excision on the lower lips and expanding them increase the practitioner’s field of vision and an accurate resection can be performed (Fig. 7.47
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


