Fig. 10.1
The Jadad score
Positive characteristics are:
-
Patients randomization: was the study described as randomized (this includes words such as randomly, random, and randomization)?
-
Correct choice and description of the randomization method: was the method used to generate the sequence of randomization described and appropriate (table of random numbers, computer-generated, etc.)?
-
Double blind clinical trial: was the study described as double blind?
-
Correct and described performance of the blinding: was the method of double blinding described and appropriate (identical placebo, active placebo, dummy, etc.)?
-
Predefined description of lost to follow-up: was there a description of withdrawals and dropouts?
Negative characteristics are:
-
Not correct choice of the randomization method: deduct one point if the method used to generate the sequence of randomization was described and it was inappropriate (patients were allocated alternately, or according to date of birth, hospital number, etc.).
-
Not correct blinding allocation: deduct one point if the study was described as double blind, but the method of blinding was inappropriate (e.g., comparison of tablet vs. injection without employing the double dummy technique).
Once we have understood the number and the importance of evaluation methods in clinical research, we have to move into meta-analysis which is the main part of EB movement.
10.3 Meta-Analysis as a New Approach in Clinical Research
Meta-analysis is a secondary instrument of research aimed to summarize data from various instruments of primary research, in particular from clinical studies.
Precisely, it consists of a series of mathematic- statistical methods designed to integrate the outcomes derived from clinical studies, with the goal of obtaining a unique quantitative evaluation index to draw conclusions stronger than those retrieved from every single study.
Meta-analysis has become popular in the medical research field where the available information is generally the result of various clinical researches, designed with similar protocols.
Some studies are often always limited when considered individually, either in the numerosity or in the definition of goals, in order to obtain crystalline conclusions, and above all, generalized statements in relation to the effect of the treatment. The possibility to gather results from various studies represents an interesting alternative approach that strengthens our knowledge on the effect of the treatment. Let us check some examples of meta-analysis in the medicine and dentistry fields.
10.3.1 Thrombolytic Therapy and Lidocaine in Myocardial Infarction
One of the main examples of the impact of meta-analysis is, for instance, the debate on the use of thrombolytic therapy in myocardial infarction. The large volume of published randomized, controlled trials has led, in fact, to a need for meta-analyses to track therapeutic advances in this field, in order to look at trends in efficacy and to determine whether this treatment appears to be significantly effective or deleterious. The investigators conducted the first RCT of thrombolytic therapy in the late 1950s, enrolling 23 patients and finding approximately half the number of deaths in treated patients as in control patients. With this very small sample size, the confidence interval (CI) was extremely wide. With the further advent of new trials in the 1970s and with the randomization number of over 2,500 patients, the CI no longer overlaps the line of no-effect, suggesting therefore that chance is no longer an explanation of the differences between treatment and control. By 1990, with the randomization of nearly 50,000 patients, the CI around the odd ratios of approximately 0.75 (an odds reduction of 0.25) are very narrow (Table 10.1).
Table 10.1
RCTs on thrombolytic therapy
|
Years
|
Number of trials
|
Number of patients
|
p value
|
|---|---|---|---|
|
1960–1970
|
4
|
316
|
n.s.
|
|
1970–1980
|
19
|
5,451
|
<0.01
|
|
1980–1990
|
47
|
42,387
|
<0.00001
|
As the evidence was accumulating that thrombolytic therapy was working, a considerable variability in expert opinion still existed. A consensus for administering thrombolytic therapy was not reached before 1990 (Table 10.2).
Table 10.2
Textbook/review recommendations on thrombolytic therapy
|
Years
|
Not mentioned
|
Experimental therapy
|
Routine/specific
|
|---|---|---|---|
|
1960–1970
|
26
|
0
|
0
|
|
1970–1980
|
35
|
4
|
1
|
|
1980–1990
|
21
|
17
|
40
|
The opposite example regards the use of prophylactic lidocaine to prevent lethal ventricular arrhythmias in patients presenting with myocardial infarction.
Here the randomized trial evidence never supported therapy (Table 10.3), and in fact, suggested a possible increase in mortality. Nevertheless, some experts continue to recommend this practice until almost 1990 [67] (Table 10.4).
Table 10.3
RCTs on prophylactic lidocaine
|
Years
|
Number of Trials
|
Number of patients
|
p value
|
|---|---|---|---|
|
1960–1970
|
0
|
0
|
n.a.
|
|
1970–1980
|
12
|
1,986
|
n.s.
|
|
1980–1990
|
3
|
6,759
|
n.s.
|
Table 10.4
Textbook/review recommendations on prophylactic lidocaine
|
Years
|
Not mentioned
|
Experimental therapy
|
Routine/specific
|
|---|---|---|---|
|
1960–1970
|
5
|
0
|
21
|
|
1970–1980
|
6
|
1
|
31
|
|
1980–1990
|
16
|
1
|
75
|
As regards myocardial infarction, fibrinolytic therapy would have been adopted and lidocaine would have been abandoned 10–15 years before if meta-analysis had been adequately recognized, allowing thousands of patients to benefit a more effective and secure therapy.
10.3.2 The Impact of Polyol-Containing Chewing-Gums on Dental Caries
Some meta-analyses have also been conducted in dentistry. Their role is to try to achieve some evidences from different studies executed in different centers in order to answer a common clinical question. Once a question is answered with a strong methodological base, it is time to stop searching in this context and to move on daily clinical practice. Our case deals with a recent meta-analysis conducted on the impact of sugar substitutes (called polyols) on dental caries [5].
It is important to affirm that dental caries in the United States is considered the most common chronic childhood disease and that more than 90% of children have experienced caries at some point in their lives [42, 75].
Many preventive programs (individual hygiene procedure and dental office procedure) have been developed and a reduction of overall caries burden could be more beneficial from a public health perspective [9].
Also, lots of studies have been executed on the effect of sugar substitutes known as “polyols” or “sugar alcohols” that are nonfermentable sugars commonly used in chewing-gum (xylitol, sorbitol, mannitol). Typical meta-analysis methods have been used to retrieve and evaluate the type and quality of studies. From a starting number of 231 eligible articles, 19 articles were finally selected for this meta-analysis. At the end of their analysis, they conclude that polyol-containing chewing-gum does reduce dental caries. Even if some gaps in literature are still present around dose-response relationship and the efficacy of different polyols, there are some consistent evidences to support the use of xylitol and sorbitol-containing chewing-gum as part of normal hygiene to prevent dental caries.
As we can see this is not only important for the individual patient, but also, and most importantly, for a possible reduction of these lesions in general population with lots of benefits for public health care services. Such kind of answer must be obtained only through the meta-analysis of well-designed and well-conducted studies.
Despite these positive examples, there are some concerns about the utility of meta-analysis regarding different points: there are few doubts about the fact that putting together results derived from different studies may give serious problems as regard the interpretation and methodological issues. As far as end points and study design are concerned, we will often have to analyze and interpret results obtained from numerous dissimilar studies. For instance, not all studies will be able to give the same input on the definition of treatment efficacy, while some will be underpowered due to the low number of treated patients or the loss of them during follow-up.
One of the most difficult issues is to try to give the correct importance to different results coming from similar studies that have identical end points but different conclusions.
Now we are going to accurately analyze the way meta-analyses are composed, and what is EB movement’s basic process.
10.4 Randomized Clinical Trials (RCT) and Their Role in Medicine Progress
The strengths of EBM movement have been to try to give biology and medicine a more predictable approach, providing clear answers to open-ended questions.
In medicine and dentistry, learning and acquiring professional competences have been once based on experts’ word (e.g., various specialists’ personal experience, reference books, congress cultural exchanges). The non-systematic observations based on clinical experience were thought to be valid in order to form our knowledge on prognosis, diagnosis, and effectiveness of treatment.
The knowledge of the biological and physiological mechanisms, the good sense, and the clinical experience seemed to be sufficient in order to guide the specialist in clinical and the evaluation of new therapies or diagnostic techniques.
The field experts had to produce for consensus the guidelines for the treatment and the diagnosis of several pathologies.
With the advent of EBM, all has changed: the importance of the personal experience has been limited and it has been proposed that the decisions and the knowledge about diagnostic tests, prognosis, and treatments had to be based on all validated and statistically significant data derived from well-designed studies. The clinical experience was still fundamental; however, the clinical observation had to be made in a systematic, reproducible, and quantitative way (research methodology).
The knowledge of the biological mechanisms is necessary, but not sufficient to foretell the “best” clinical practice at our time in which new mechanisms of molecular biology always widen our knowledge on diseases on the basis of patients’ characteristics. Formation and medical practice must not only be based, therefore, on the doctor’s ability to retrieve the information on the best possible EB on the most important results of clinical research, but also on the ability to consult the medical literature and databases for dealing those cases that go far beyond clinical routine.
In particular, EBM’s efforts focused on three fundamental points such as:
-
Searching the information with the maximum efficiency.
-
Interpreting the articles that bring back the inform ation.
-
Estimating validity.
Above all, for an efficient research of the information, EBM makes reference to research via scientific and computer science articles taking advantage of data collection, like Cochrane Collaboration, Embase, and Medline.
The main topic of EBM literature consists of therapy rather than diagnosis. The therapeutic procedures (drugs, surgical techniques, etc.) must always be estimated through RCTs, while the consequent meta-analyses quantitative and qualitative synthesis of multiple RCT.
These are RCTs, that is, extremely common scientific experiments performed on man in order to estimate the effectiveness of new instruments, drugs, or techniques, in respect of those used once in clinical practice, which represented the “gold standard.” In RCTs, a comparison between gold standard and innovative therapy was studied to determine the most effective treatment.
RCTs are used in all scientific research fields; they are experimentations, in which the investigator proposes to the patient through an informed consent the possibility to be involved in a study to improve therapeutic results or to reduce the side effects of a specific treatment. Consequently, positive perturbations such as therapeutic, preventive, and rehabilitative interventions can be performed.
It is important to affirm that before any clinical trial is carried out, result of nonclinical investigations or previous human studies should be sufficient to indicate that the drug is acceptably safe for the proposed investigation in humans [55].
RCTs are carried out in dentistry as in any other medical specialty. In this case, RCTs are a guarantee of an advanced quality of result that will further be evaluated. They are structured in the same way, although the patients’ recruitment phase and the collection of a high and statistically significant number of patients turn out to be more difficult.
The necessary number of RCT participants is determined through a statistic analysis before the beginning of the study based on the primary goal to reach and the quantification of a possible increase of the benefit in order to assure a statistic significance to the study.
The main characteristics of RCTs are (Fig. 10.2):
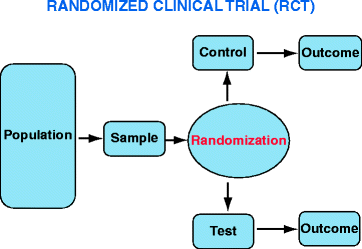

Fig. 10.2
Description of RCT design
-
There are two or more groups of study: Test group (new therapy) and Control group (placebo or gold standard therapy).
-
Participants are randomly distributed in Test and Control groups in order to reduce the influence of known or unknown confusing factors.
This allows to increase the probability that the two groups are similar and that possible differences in study outcomes are due only to the type of procedure that has been attributed to the patient and not to known or unknown disease characteristics that they are canted in a group of treatment.
Other characteristics of RCT studies are perspective longitudinal design of the study, the necessity of an ethical committee approval, and beyond the necessity of having the patient’s informed consent of as mentioned above.
RCT studies should be used in cases in which drug effectiveness, surgical techniques, screening interventions, sanitary education, and health care system organization are evaluated.
According to the degree of the staff’s knowledge of participant distribution into Test and Control groups, it is possible to distinguish four RCT types:
-
Open RCT: both the subjects and the clinicians know which group participants belong to.
-
Single blind RCT: only the clinicians know which group participants belong to.
-
Double blind RCT: only the staff involved in the randomization procedure know which group participants belong to through a code, but neither the clinicians who execute the procedure nor the patients know the type of treatment.
-
Triple blind RCT: only the staff involved in the randomization procedure know which group participants belong to through a code, but neither the clinicians who execute the procedure, nor the patients nor those who analyze the outcomes know the type of treatment.
In the hierarchy of evidence that influences health care practice, RCTs are largely considered to be the top individual unit of research. They are considered the most reliable form of scientific evidence because they eliminate spurious causality and bias.
One example of a very important RCT is the one regarding the use of hormone replacement therapy (HRT) in postmenopausal women that was thought to reduce cardiovascular diseases, and indeed, was found not only to increase these issues, but also and more dramatically to increase the risk of breast cancer.
10.4.1 Hormone Replacement Therapy (HRT)
Despite decades of accumulated observational evidence, the balance of risks and benefits for hormone used in healthy postmenopausal women remained uncertain, and in common clinical practice, a wide use of such therapy was administered without any real EB data.
To assess the major health benefits and risks of the most commonly used combined hormone preparation in the United States, a randomized trial was finally performed. Overall health risks exceeded benefits from the use of combined estrogen plus progestin for an average 5.2-year follow-up among healthy postmenopausal US women as recently reported by Rossouw et al. [78] in their analysis of data from Women’s Health Initiative randomized controlled trial.
Thus, in today’s health care system, all the new procedures must have been previously estimated through RCTs to be approved and be applied in clinical practice. Such studies are used in order to determine not only the effects of a therapy, but also to detect and underline possible side effects.
10.4.2 RCT: What they Can Say and What they Cannot Say. Are Observational Studies Still Needed?
The onset of EB has generated many criticisms: too rigid, and not able to fit medical flexibility for the complexity of the subject as well as for cultural and ethical reasons.
One of Smith and Pell’s famous articles clearly explained this EB question. A systematic review on the parachute effect in preventing death and major trauma related to gravitational challenge using EBM instruments was performed [88]. Obviously, there were no publications available in Medline, Embase, and Cochrane Library databases and the outcomes were considered insignificant; the authors ironically declared that the parachute was not safe enough to be widely used, since there were no RCTs present in Literature. In addition, the strongest advocates of EBM movement were invited to take part in a randomized, placebo-controlled, crossover, double blind trial on the use of parachute. This would have for sure resulted in a disaster; other types of experimental studies exist for such purpose.
Scientific studies can be led as experiments or observations. In the first case, the researcher will establish the parameters of the study, while in the second case the researcher works only as an observer. Transversal or longitudinal studies determine the duration of the study. As regards, longitudinal studies can be carried out on the basis of previously existing data (retrospective studies) or data collected over time (prospective studies).
The so-called observational studies are based on the researcher’s external observation of reality in which potentially harmful etiologic factors are investigated, such as bad habits (alcohol, smoke, etc.) and precarious environmental conditions (X ray, passive smoke, etc.). There is no sample randomization, but self-selection.
In EBM movement, observational studies have a minor evidence value and can be classified in:
1.
Study group or cohort (or prospective). Two groups are confronted: the study group is exposed to treatment/causal agent, while the second group is that of control. Both groups are followed over time (prospective study) in order to detect the incidence of a pathology.
2.
Case/control study (or retrospective). Two groups are controlled: one is affected by a pathology, while the other is not. Both groups are estimated on the base of the collected data in the past (retrospective or historical study) in order to discover an eventual causal agent of the pathology.
3.
Cross-sectional studies (or of prevalence). The observation is limited to a determined period of time.
In this type of observational studies, drugs are prescribed according to the officially-registered commercial purpose. The patient’s assignment to a specific therapeutic strategy is not decided in advance by an experimental protocol, but belongs to routine clinical practice. The decision to prescribe drugs does not depend on the patient’s inclusion in the study. No additional diagnostic or monitoring procedure is applied to the patients, while epidemiological methods are used for the analysis of the collected data (in compliance with the Italian law: Law Decree 24 June 2003 no.11).
To be considered as observational studies, drugs must satisfy the following conditions:
1.
The drug must be prescribed according to the officially-registered commercial purpose.
2.
The prescription of the drug under investigation must be part of the routine clinical practice.
3.
The decision to prescribe the drug to the subject must not depend on the patient’s inclusion in the study (where possible).
4.
The diagnostic and assessment procedures must correspond to the common practice (in compliance with the Italian Law: Gazzetta Ufficiale n.76 of – the 31/03/2008 AIFA – Determination 20 March 2008).
From this dissertation we clearly understand that observational studies guarantee a minor level of evidence. Therefore, are observational studies still necessary? Let us now see some examples both in medical and dental fields in order to answer this question.
10.4.2.1 Smoking
Can we perform a clinical controlled experimentation on active smoking subjecting two samples accurately selected and randomized to an intense and long-lasting exposure in order to estimate the pulmonary consequences?
It is clear that no study of this type could be performed since our ethics forbids it. How could we intentionally expose a patient to such risky and well-known carcinogenic factor?
10.4.2.2 Caries Lesions and Mutans streptococci Infection
Also, in dentistry, it seems difficult to be able, for example, to perform RCT on the correlation between caries and diet or daily habits; we cannot imagine a study in which a population is subjected to an intense and frequent carbohydrate daytime exposure and another population in which such exposure is simply absent, that is, ineffective from the caries point of view. Undoubtedly, we would worsen the oral health of the study group intentionally, obtaining outcomes not so different from those derived from longitudinal or retrospective observational studies [58].
It is therefore important to affirm that even if the reliability and the importance of RCTs as a model of research from a methodological point of view are undeniable, they are not universally applicable in medicine. Indeed this field requires a certain degree of flexibility in order to adapt to a myriad of not only biological, but also cultural and deontological variables, which are hardly compatible with such a rigid and unmodifiable method as RCTs.
Even though observational studies constitute a lower level of evidence for EBM purists, these studies should be promoted in surgery as a source of evidence along with the development of standards for meta-analyses of nonrandomized studies [107].
It has been shown that high-quality nonrandomized (observational) studies and high-quality RCTs can produce similar answers [8]. Despite the fact that clinical surgical research is mainly based on nonrandomized studies, there is no agreement on the methodological standards for such studies [28, 87].
Few attempts have been made to develop instruments allowing surgeons to evaluate published nonrandomized studies, and thus, quantify the strength of their results before applying their conclusions to “individual patients.”
A systematic review [59] showed that study estimates of effectiveness may be valid in this case if confounding factors are controlled for, but a formal agreement on the standards of nonrandomized studies and meta-analyses of such studies is needed.
10.5 “Best Available Research Evidence” in the Field of Dentistry
EBD is an approach to oral health care that requires the judicious integration of systematic assessments of clinically relevant scientific evidence, relating to the patient’s oral and medical condition and history, with the dentist’s clinical expertise and the patient’s treatment needs and preferences. EB care is now regarded as the “gold standard” in health care delivery worldwide [2].
But how can we reach this evidence in the dental arena?
Niederman [71] proposed in 1998 the following simple but simultaneously clear example:
“Professional or home bleaching can be a solution when aging is responsible for yellowed teeth.”
-
Problem: yellowed teeth due to aging…
-
Procedure: …professional bleaching…
-
Comparison: …or indicate a home bleaching…
-
Result: …to whiten teeth.
Once the problem has been defined, the solution must be searched in literature.
As already said, EBD is based on the information available online. As regards, there are some websites that deal with EBD. Among these, some databases can be used for retrieving information:
1.
EviDents [48] is an EBD browser developed by the Forsyth Institute of Boston, in association with PubMed databases. This system is very practical since the clinician can formulate the problem and select the specific clinical field, the age range, Public Health studies, or Systematic reviews, to identify a solution. Furthermore, the clinician can focus his own research on diagnosis, Prognosis, treatment, and so on.
2.
The US National Library of Medicine [65] allows an easier research thanks to RCT filters. The information is already selected and displays clinically relevant articles. PubMed is the most widely-used clinical basis worldwide. The website also offers a filter designed for the research of clinical study category, that is, systematic reviews [53].
3.
The Cochrane Library: [104] Cochrane is an International Organization made of 50 staff groups (over 11,500 people) and dedicated to the systematic collection of research information on the effects of health care interventions in order to: (1) make this information rapidly and conveniently available to the largest number of people; (2) put this information in a more comprehensive perspective; and (3) carry out research on methodologies to improve systematic review. The Cochrane collaboration is committed: (1) to find the best way to make this information available; and (2) to do so in a way that helps those who have to make decision both for individuals and populations.
The Cochrane Library includes 3,625 systematic Cochr ane revisions, and it is widely consulted all over the world, especially in English-speaking countries (Fig. 10.3). The Organization has officially created a group of study specialized in oral health in 1994 (managed by Prof. Shaw at Manchester University Hospital), which produces systematic reviews about diagnosis, prevention, and therapy of oral and maxillofacial diseases.
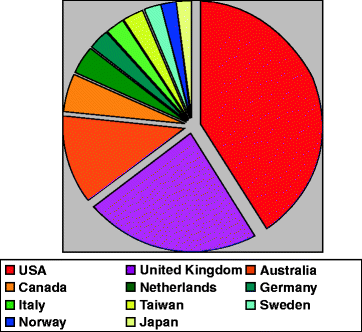

Fig. 10.3
Access on Cochrane database in 2008
There are also browsers specialized in meta-analysis:
There are reviews that gather many studies considered relevant in terms of evidence.
The goal of all those websites is to produce and spread sound knowledge derived from gold standard research methodology that insures a high level of quantitative and qualitative clinical outcomes.
In developing appropriate treatment plans, dentists should combine the patient’s treatment needs and preferences with the best available scientific evidence, in conjunction with the dentist’s clinical expertise. To keep pace with other health professions in building a strong EB foundation, dentistry will require significant investments in clinical research and education to evaluate the best currently available evidence in dentistry and to identify new information needed to help dentists provide optimal care to patients [10].
10.5.1 How Evidence-Based Dentistry (EBD) Has Changed Clinical Practice
The American Dental Association defined EBD as an approach to oral health care that requires the judicious integration of systematic assessments of clinically relevant scientific evidence relating to patients’ oral and medical condition and history, along with the dentists’ clinical expertise and the patients’ treatment needs and preferences [4].
Information derived from clinical trials is considered more reliable than information based on intuition, authority, or custom. There is a hierarchy when considering the levels of evidence (LOE). Systematic reviews of randomized controlled trials are considered to be at the highest level, whereas expert opinion is considered the lowest level of evidence [34]. This is only partially correct from our point of view.
Dentistry is a special field where the best evidence is not always applicable to the single patient. As the base of our work, we must align to protocols and guidelines for therapies and prognosis, but we also have to consider patient’s preferences and values as well as costs and personal esthetic sense both for us and the patient.
This may be exemplified by a case of a patient with four asymptomatic impacted third molars. Of the oral surgeons consulted, all of whom were working under a fee-for-service plan, 80% recommended the removal of all four teeth compared with 45% of general dentists working under a fee-for-service plan, and 27% of general dentists working under a capitation plan.
With the intention to turn over a new leaf, we go through the field of EBID and the decision itself has a great importance in everyday practice. A model for the factors influencing decisions in healthcare was described in 2000 by Chapman and Sonnenberg [17] and has been adapted to discuss decision-making in dentistry. The decision-making model describes two major components, the normative and the descriptive, which are involved in decision-making (Fig. 10.4).
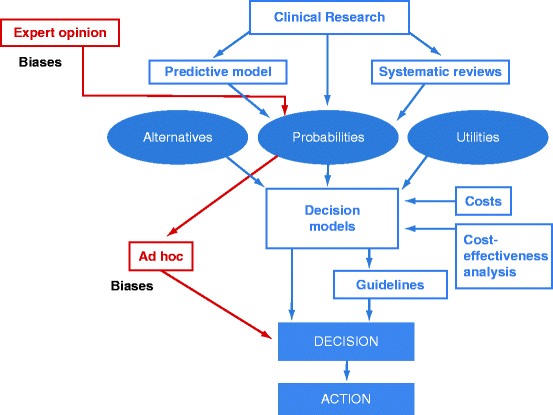

Fig. 10.4
Overall scheme of dental decision-making (adapted from Chapman and Sonnenberg [17]). The normative components of decision-making are marked in blue and the descriptive components are marked in red
The normative aspect of decision-making relies on quantitative information derived from systematic reviews and predictive models on the probabilities and uncertainties of treatment outcomes. Clinical outcomes, such as survival of a tooth or success of a restoration, are assessed on the basis of the utility they offer to the patient and their costs. Normative analyses allow quantitative comparisons of alternative therapies and can identify optimal treatments for multiple attributes. The descriptive aspect in decision-making involves cognitive processes and biases of both providers and patients that translate the normative information into clinical action. Decision-making in health care occurs at three broad levels: the level of lawmakers and governmental regulators, the level of insurance plans that determine coverage and reimbursement for healthcare, and provider and patient’s levels [34].
Thus, we enter in the field of EB practice. This term is defined as the conscientious, explicit, and judicious use of current best evidence in making decisions about the care of individual patients. It is currently defined as integration of best research evidence with clinical expertise and patient’s needs.
This is the secret to obtain real answers. In our opinion best research evidence refers to clinically relevant research, especially from patient-centered clinical research. Clinical expertise is the ability to use clinical skills and experience to rapidly identify each patient’s unique health state and diagnosis, individual risks and benefits of potential interventions, and personal values and expectations. Patient’s values refer to unique preferences, concerns, and expectations that each patient brings to a clinical encounter and that must be integrated into clinical decisions if they are to serve the patient [37].
The results of systematic reviews of databases referred to dental care have to be considered from this point of view.
10.6 Problems in Applying EBM to Surgery: Performing Surgical Procedure is Different from Administering Drugs
Even if EB movement has widely spread over the last years, some researchers do not agree to consider this scientific movement as entirely positive. As regard the fact that Holmes et al. compare EBM with fascism in their article is a clear demonstration. They affirm that EBM has had an evolution that corresponds to that of a microfascism: EBM has been colonized (territorialized) by an all-encompassing scientific research paradigm – that of postpositivism – but also and foremost in showing the process by which a dominant ideology comes to exclude alternative forms of knowledge, thereby acting as a fascist structure [43]. In fact, perhaps the comparison remains excessive since EBM has met considerable success in scientific research and literature. However, 96% of the articles do not satisfy the inclusion criteria of the EB research [95].
What is the meaning of this datum? Does it imply that only 4% of articles are significant because they are correctly executed? That 96% of studies conducted in medical research are not reliable?
Surgery (and dentistry) allows us to understand this methodological problem in the field of research and is always considered a qualitatively lower branch compared to medicine, for the humble origins of its tradition. Extraordinarily effective in its convenience, the ancestral art-surgery has always been limited to a subordinate role compared to medicine.
Also, while on the one hand, in all ancient civilizations the professional medical figure was represented by the noble priest, the astrologer, the philosopher or the esoteric magician, shaman, sorcerer, on the other hand, there was the surgeon. The latter belonged to some vulgar category even if he could treat some diseases and explain many of them. Many doctors do not like the idea of treating patients soiling one’s hands (the word “surgery” comes from Greek words cheir (hand) and ergon (work), i.e., handiwork): a drug administration is more elegant than a surgical procedure. However, surgical procedures and drug administration are different.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


