Evaluation of Periodontal Indices
Periodontal indices are often used for evaluation of dental implants. A comparison of natural teeth and implants for each criterion provides insight into their differences in the health-disease continuum. After one understands the basis for evaluation, these criteria may then be used to establish a health-disease implant quality scale related to patient treatment.
Mobility
Natural Tooth vs. Implant Support Systems.
A dental implant and a natural tooth differ in many ways. In general, a natural tooth is better designed to reduce the biomechanical forces distributed to the tooth/restoration and the crestal bone region. The periodontal attachment apparatus, biomechanical design of the tooth root and material, nerve and blood vessel complex, occlusal material (enamel), and surrounding type of bone blend to decrease the risk of occlusal overload to the natural tooth system.10
Tooth movement.
A tooth exhibits normal physiologic movements in vertical, horizontal, and rotational directions. The amount of movement of a natural tooth is directly related to its surface area and root design. The factors that dictate the movement include: the number and length of the root surface, the root diameter, shape, position, health of the periodontal attachment apparatus, and the bone density primarily influence a tooth’s mobility. A healthy tooth exhibits zero clinical mobility in a vertical direction. Studies have shown an initial vertical tooth movement to be about 28 µm and is the same for anterior and posterior teeth.11 The vertical movement of a rigid, fixated implant has been measured to be approximately 2 to 3 µm under a 10-lb force and is due mostly to the viscoelastic properties of the underlying bone.12
Muhlemann found that horizontal tooth movement may be divided into initial mobility and secondary movement.13 The initial mobility is observed when there is a light force applied, which occurs immediately, and is a consequence of the periodontal ligament (PDL). Initial horizontal tooth mobility is greater than initial vertical movement. A very light force (500 g) may horizontally move a tooth. The initial horizontal mobility of a healthy, “nonmobile” posterior tooth is less than that of an anterior tooth and ranges from 56 to 75 µm, which is two to nine times the vertical movement of the tooth. Initial horizontal mobility is even greater in anterior teeth and ranges from 70 to 108 µm in health (Fig. 18.2).14

The secondary tooth movement described by Muhlemann occurs after the initial movement when greater forces are applied. When an additional force is applied to the tooth, a secondary movement is also observed, which is related directly to the amount of force. The secondary tooth movement is related to the viscoelasticity of the bone and measures as much as 40 µm under considerably greater force (Fig. 18.3).13
Implant movement.
The term rigid fixation is used to describe the absence of clinical mobility of an implant tested with vertical or horizontal forces less than 500 g. However, rigid fixation is a clinical term. Osseointegration is a histologic term that is defined as bone in direct contact with an implant surface at the magnification of a light microscope (Fig. 18.4).15 Over the years, these two terms have been used interchangeably, and implant abutment support is most predictable with rigid fixation. Lack of implant mobility (IM) does not always coincide with a direct bone-implant interface. However, when observed clinically, rigid fixation usually means that at least a portion of the implant is in direct contact with bone, although the percentage of bone contact cannot be specified. When an implant has mobility, there is most likely connective tissue between the implant and bone.

In some situations, the presence of implant mobility may be difficult to discern. The absence of clinically observable movement does not indicate the true absence of any movement. For example, a “nonmobile” posterior natural tooth actually moves horizontally 56 to 73 µm. The human eye does not perceive this movement. The anterior teeth, which often have slight clinically observable movement, actually move approximately 0.1 mm. A healthy implant may move less than 73 µm; it appears as zero clinical mobility (rigid fixation).
Similar to a natural tooth, the implant-bone interface exhibits more lateral than apical movement. Sekine et al evaluated the movement of endosteal implants with rigid fixation and found a range of 12 to 66 µm of movement in the labiolingual direction.12 Komiyama reported 40 to 115 µm of implant movement in the mesiodistal direction under a force of 2000 g (≈4.5 psi) and a labiolingual range of 11 to 66 µm.16 The greater implant movement in the mesiodistal dimension corresponds to the lack of cortical bone between the implants in this direction compared with the thicker lateral cortical plates present in the labiolingual dimension. Rangert et al suggested that part of this implant movement may also be due to component flexure of the implant abutment and screw.17 The mobility of implants varies in direct proportion to the load applied and the bone density and reflects the elastic deformation of bone tissue.
Sekine et al applied a gradually increasing load over a 2-second period to a tooth and an implant. The teeth moved immediately with a light load (primary tooth movement) and less with an additional load (secondary tooth movement). The implant did not move when the tooth had its primary tooth movement. A heavier force caused the implant to gradually move, similar to the secondary tooth movement (see Fig. 18.3).12 These mobility characteristics corroborate the findings of Fenton et al, who applied a 500-g load for 4 seconds to maxillary anterior teeth and osseointegrated implants.18 Whereas the implants were displaced a mean of 10 µm with a rapid elastic return (less than 1 millisecond), the teeth showed a mean displacement of 57 µm with a prolonged viscoelastic return.
Evaluating Dental Implant Mobility
Mirror handles.
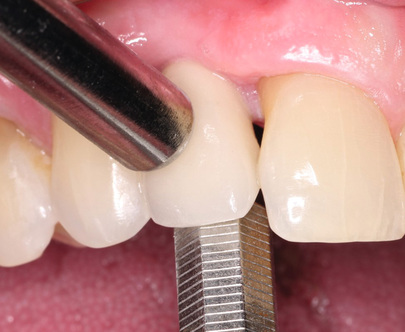
Increased tooth mobility may be caused by many factors including occlusal trauma or bone loss. Increased tooth mobility alone is not a criterion of periodontal health or pathology. Unlike a tooth, for which mobility is not a primary factor for longevity, mobility is a primary determining factor for implant health.15 Rigid fixation of an implant is also an excellent indicator of the implant health status because it is an easy, objective test. As such, rigid fixation is usually the first clinical criterion evaluated for a dental implant. The techniques to assess rigid fixation are similar to those used for natural tooth mobility. Two rigid instruments apply a labiolingual force of approximately 500 g, and no observed mobility indicates rigid fixation (Fig. 18.5).3
The amplitude of tooth mobility may be rated from 0 to 4, where 0 is normal mobility from physiologic movement; 1 is detectable increased mobility; 2 is visible mobility up to 0.5 mm; 3 is severe mobility up to 1 mm; and 4 is extreme mobility, including vertical movement.3 This same gradient may be used for oral implants with slight modification. As Table 18.1 depicts, IM-0 corresponds to the absence of clinical mobility, IM-1 demonstrates detectable increased movement, IM-2 is visible mobility movement up to 0.5 mm, IM-3 is severe horizontal mobility greater than 0.5 mm, and IM-4 is visible horizontal and vertical movement. The IM scale was used frequently for plate (blade) form implants or disc implants because a clinical goal was for slight mobility when joining the device to natural teeth. However, the ideal goal for root form implants should always be rigid fixation and IM-0 status (no movement).
TABLE 18.1
Clinical Implant Mobility Scale
| Scale | Description |
| 0 | Absence of clinical mobility with 500 g in any direction |
| 1 | Slight detectable horizontal movement |
| 2 | Moderate visible horizontal mobility up to 0.5 mm |
| 3 | Severe horizontal movement greater than 0.5 mm |
| 4 | Visible moderate to severe horizontal and any visible vertical movement |
Periotest.
The Periotest (Gulden-Medizinteknik) is a computer-mechanical device, developed by Schulte, that measures the dampening effect or attenuation degree against objects by developing a force of 12 to 18 N against a pistonlike device, which then measures the distance the piston recoils into the chamber after striking an object.19 A soft surface or mobile object gives higher recordings than a hard or rigid object. The recordings range from negative 8 to positive 50 numbers.
Teeth with zero clinical mobility have typical Periotest ranges from 5 to 9. The degree or absence of clinical movement around an implant corresponds to values ranging from −8 to +9, or a range of 17 units. The bone density around the implant may be correlated with Periotest numbers. Whereas softer bone types give higher numbers, harder bone around the implant results in lower numbers. A nondestructive resonance frequency analysis technique to measure implant stability and osteointegration has also been introduced to the profession and provides similar valuable information as to the clinical movement and bone density around implants (Table 18.2).20,21
TABLE 18.2
Periotest Values vs. Clinical Interpretation
| Periotest Value | Clinical Interpretation |
| −8 to 0 | Satisfactory integration |
| +1 to +9 | Clinical examination recommended because integration is not sufficient for prosthetic loading |
| +10 to +50 | Integration insufficient, progressive bone loading recommended |
Osstell.
A second device exists to evaluate the implant-bone interface that is nondestructive and noninvasive. The Osstell (Ostell) is based on resonance frequency analysis (RFA) and was developed by Huang.22 The Osstell has been shown to have quantitative and reproducible measurements on the presence of integration, immediate load feasibility, and follow-up evaluation at the prediction of an implant failure. RFA is a technique that is based on continual excitation of the implant interface through the use of dynamic vibration analysis (piezo effect). A specialized transducer, which contains two piezoceramic elements, is either attached directly to the implant or abutment. The first piezo element generates an excitation signal that is a sinusoidal wave (5–15 kHz), leading to vibration of a whole transducer-implant-tissue complex. The oscillation response is measured by the second piezo element.23
The RFA technique measures implant stability as a function of stiffness of the bone/implant complex. The health of the implant is measured on an implant stability quotient (ISQ) that is calculated on a scale from 1 to 100. The full integration of an implant is most usually measured in the range from 45 to 85 ISQ. Measurements of less than 45 are indicative of implant failure, whereas an ISQ value of 60 to 70 indicates success.24 Studies have shown that the Osstell is more reliable than the Periotest in evaluating and measuring implant stability in hard and soft tissue interfaces (Fig. 18.6 and Table 18.3).25

Plaque Biofilm
The differences between tooth and implant biologies make dental implants more susceptible to inflammation and bone loss in the presence of bacterial plaque accumulation.26 Biofilms are the primary causative factor of periodontal disease processes. Sticky masses of bacteria with a polysaccharide matrix, water, and bacteria accumulate on hard and soft surfaces in the oral cavity and can be disturbed and removed with mechanical or chemical obliteration. If undisturbed, mature plaque will form. Current chemotherapeutics cannot penetrate thick biofilm, and rough surfaces have been found to hold more biofilm than smooth surfaces.27 Bacteria will migrate from teeth to implants and from implant to implant. Similar to teeth, clinical findings of failing implants include inflammation, pockets, and progressive bone loss. Another similarity lies in the bacteria responsible for periodontitis and peri-implantitis.
When evaluating the peri-implant microbiota, Lee et al. compared microbial changes between patients with a history of periodontal or peri-implant infections and implants that have been in function for a length of time.28 This study found a history of periodontitis had a greater impact on the peri-implant microbiota than implant loading time. The major influence on the peri-implant microbiota was, however, the microbiota on remaining teeth. Porphyromonas gingivalis and Bacteroides forsythus (Tannerella forsythia), red complex periodontal pathogens, colonized several implants, although all implants were successfully osteointegrated. It is important to educate patients about their responsibility to decrease plaque effectively, especially if they have a history of periodontal disease.
Plaque biofilm development and maturation have similarities for natural teeth and dental implants. The gingival sulcus in periodontal health and the perimucosal attachment of a successful dental implant are essentially similar. In a study by Mombelli and Mericske-Stern of the plaque from 18 edentulous patients with successful dental implants, facultative anaerobic cocci (52.8%) and facultative anaerobic rods (17.4%) were reported.
However, the pathogens P. gingivalis and spirochetes were absent, and minimal (7.3%) gram-negative rods were present.29 Generally, pellicle—a naturally occurring glycoprotein in the saliva—first adheres to the intraoral structure, whether it be a tooth or an implant. Gram-positive cocci bacteria are the first “early colonizers,” beginning with single cocci and progressing to streptococci forms (Box 18.3).
Without appropriate oral hygiene measures (e.g., brushing, flossing, interdental cleaning), additional bacterial colonies including gram-negative rod-shaped bacteria synergistically grow with the established gram-positive bacteria. The gram-negative bacteria are frequently facultative or strict anaerobic bacteria and are considered “late colonizers.” Many, if not the majority, of these gram-negative bacteria are black pigmented and are classified under a number of genera (e.g., Bacteroides, Prevotella, Porphyromonas, Fusobacterium).
Plaque biofilm reported to be associated with failing dental implants also consists largely of gram-negative rods.30 Clinically, failing dental implants are characterized by soft tissue inflammation, increased probing depths, increased mobility, and peri-implant radiolucency.
Specific pathogens in implant pockets greater than 6 mm include Actinobacillus actinomycetemcomitans (Aggregatibacter actinomycetemcomitans), Prevotella intermedia, and P. gingivalis in more than one third of the sites, as confirmed by DNA analysis.31
In other studies on plaque biofilm around dental diseases and failing implants, differences in bacterial type have been reported.32,33 Mombelli did not detect spirochetes in plaque samples from well-maintained and clinically healthy implants. Rams et al noted higher proportions of staphylococci (15.1%)19 than usually found in gingivitis (0.06%) and periodontitis (1.2%) sites.34 This finding suggests that staphylococci may be more significant in developing peri-implantitis lesions than previously recognized.
Changes involve both the hard and soft tissues surrounding an implant. The implant may exhibit all the signs of peri-implant mucositis, as well as exudate, increased pocket depth, and bone loss. If left untreated, significant bone loss, infection, and mobility could result, leading to the failure of an initially integrated implant.
Comparisons of plaque biofilms have been reported in a limited study of Brånemark and ITI (Straumann Institute) implants and are remarkably similar in controlled studies. Mombelli et al compared 10 patients with Brånemark implants and 10 patients with ITI implants and sampled the deepest pockets around the implants.35 After 3 and 6 months, several periodontal pathogens were cultured and isolated, including P. gingivalis, P. intermedia, Fusobacterium nucleatum, and various spirochetes. None of the implants were colonized by A. actinomycetemcomitans. Longer investigations by Leonhardt et al extended these microflora reports on dental implants in 19 patients. At 3 years, the osteointegrated implants were colonized predominantly by P. gingivalis, P. intermedia, and A. actinomycetemcomitans.36
Natural dentitions with dental implants appear to increase the risk for implant infections, compared with completely edentulous patients. This suggests that natural teeth may serve as a reservoir for periodontal pathogens that may extend their growth to contiguous implants in the same oral cavity.37 Quirynen and Listgarten reported that proportions of coccoid forms (65.8%), motile rods (2.3%), and spirochetes (2.1%) in implant pocket areas were similar to the microorganisms in natural teeth (55.6%, 4.9%, and 3.6%, respectively). Fully edentulous patients exhibited more coccoid forms (71.3%), fewer motile rods (0.4%), and no spirochetes.38 They also concluded that microflora in partially edentulous implant patients were potentially more pathogenic than they were in fully edentulous patients. Implants with longevity of more than 3 or 4 years appear to have greater numbers of bacteria than implants in place for 1 or 2 years.
Clinical Significance
Plaque biofilms are the main reason peri-implantitis treatments fail. Host tissue is unable to reform attachments onto implant surfaces if titanium surface is not thoroughly cleaned. Bacterial deposits produce toxins that prevent fibroblast and osteoblast growth, which prevents proximal regeneration onto implant surface. To ensure the most optimal condition for treatment success, plaque biofilms need to be thoroughly removed. It is important to note that while it is impossible to guarantee 100% sterility of exposed implant surfaces, the body is capable of removing small amounts of bacterial deposit via cellular defense mechanisms. Carefully removing macro deposits of plaque biofilm and irrigating with antimicrobial solution is generally sufficient to allow a favorable environment for new attachment formation.
In essence, the patient should have a full-mouth debridement to reduce overall oral load of bacterial colonies while making an effort to remove all plaque biofilms on exposed implant surfaces. This gives the body a chance to reform a healthy-associated colonization of biofilm.
Percussion
Percussion often is used on teeth to determine which tooth is sensitive to function or if pulpal necrosis is beginning. In the past, percussion was used to evaluate the presence of rigid fixation for osseointegrated implants.15 However, percussion is an indicator neither of clinical health nor of rigid fixation for osseointegrated implants. The ringing sound that occurs on percussion only corresponds to the presence of some amount of bone at the interface because 2 mm of bone and 16 mm of bone-implant interface sound almost identical.
Clinical Significance
Percussion may be used to diagnose pain or tenderness with an implant but is misleading if used to determine the status of rigid fixation.
Probing Depths
Probing depths around teeth is an excellent proven means to assess the past and present health of natural teeth. The increasing sulcus depth around natural teeth is directly related to disease and bone loss. However, probing depth indices used to evaluate dental implants are more controversial because implant sulcus depth and health are not always directly related. A thorough appreciation of the soft tissue interface is needed for the clinician to understand the inherent differences between natural teeth and dental implants.
Soft Tissue Interface Around Teeth and Implants
For a natural tooth the surrounding soft tissue has an average biologic width of 2.04 mm between the depth of the sulcus and the crest of the alveolar bone. It should be noted the biologic “width” is actually a height dimension with a greater range in the posterior region compared with the anterior and may be greater than 4 mm in height.39 With respect to natural teeth, the biologic width is composed of a connective tissue attachment (1.07 mm average) above the bone and a junctional epithelial attachment (JEA) (0.97 mm average) at the sulcus base, with the most consistent value among individuals being the connective tissue attachment (Fig. 18.7).

The sulcular regions around an implant and around a tooth are similar in many respects. The rete peg formation within the attached gingiva and the histologic lining of the gingiva within the sulcus are similar in implants and teeth.40 A free gingival margin forms around a tooth or implant with nonkeratinized sulcular epithelium, and the epithelial cells at its base are also similar to teeth and implants, with junctional epithelial cells for both. However, a fundamental difference characterizes the base of the gingival complex around teeth. Whereas a tooth has two primary regions that make up the biologic width, an implant only has one (Fig. 18.8).

When probing next to a tooth, the probe not only measures the sulcus depth but also penetrates and measures the JEA.41 The junctional epithelial “attachment” of a tooth is not a true attachment. It attaches to teeth via hemidesmosomes, but these attachments are light in general and are easily disrupted. A periodontal probe, plaque, or impression material can easily separate the hemidesmosomal attachment. At the apex of the sulcus is the junctional epithelium, which is a few cells thick and supported by the connective tissue attachment zone (see Fig. 18.8).
The connective tissue attachment zone of the “biologic width” around a tooth prevents the probe from penetrating deeper into the sulcus and allows gingival fibers of the connective tissue attachment zone to establish direct connection with the cementum of the natural tooth. It acts as a physical barrier to the bacteria in the sulcus to the underlining periodontal tissues. Eleven different gingival fiber groups compose the connective tissue attachment zone observed around a natural tooth and tissue: dentogingival (coronal, horizontal, and apical), alveologingival, intercapillary, transgingival, circular, semicircular, dentoperiosteal, transseptal, periosteogingival, intercircular, and intergingival.3 At least six of these gingival fiber groups insert into the cementum of the natural tooth: the dentogingival (coronal, horizontal, and apical), dentoperiosteal, transseptal, circular, semicircular, and transgingival fibers. In addition, some crestal fibers from the periodontal fiber bundles also insert into the cementum above the alveolar bone. These Sharpey fibers form a true attachment to the tooth. They prevent a periodontal probe from invading the PDL space and delay the ingress of plaque.
James and Schultz were the first to begin a systematic study to investigate the biologic seal phenomenon of the soft tissue around dental implants.40 Hemidesmosomes from the JEA region help form a basal lamina-like structure on the implant, which can act as a biologic seal. However, collagenous components of the linear body cannot physiologically adhere to or become embedded into the implant body.26 The hemidesmosomal seal has a circumferential band of gingival tissue to provide mechanical protection against tearing. However, the mucopolysaccharide layer is less adherent to an implant surface than a natural tooth root. The hemidesmosome of the natural tooth has a lamina lucida and a lamina densa, which are part of the basement membrane. The hemidesmosome next to an implant has a lamina lucida, lamina densa, and sublamina lucida (which is less adherent).42
The biologic width for implants has been reported by Cochran et al to be 3.3 mm, but unlike the biologic width dimension for teeth, they also included the sulcus depth.43 In a typical implant gingival region, only two of the gingival fiber groups found around a tooth (circular and periosteogingival fibers) and no periodontal fibers are present.44 These fibers do not insert into the implant body below the abutment margin as they do into the cementum of natural teeth. Instead, the collagen fibers around an implant run parallel to the implant surface, not perpendicular, as with natural teeth.45 Hence, the implant only has a junctional epithelial “attachment” system.
The gingival and periosteal fiber groups are responsible for the connective tissue attachment component of the biologic width around teeth, and these are not present around the transosteal region of an implant. The “biologic width” around the abutment-implant connection should not be similarly compared with the connective tissue attachment of a tooth. The biologic seal around dental implants can prevent the migration of bacteria and endotoxins into the underlying bone. It is unable, however, to constitute an attachment component of the biologic width similar to the one found with natural teeth (Fig. 18.9).
Consequences of Probing Around an Implant
A dental probe introduced into an implant sulcus may proceed through the junctional epithelial close approximation of tissue and the probe may proceed to the crestal bone (Fig. 18.10).

The connective tissue zone for an implant has only two fiber groups, and neither of them inserts into the implant. As a result, with an implant the probe goes beyond the sulcus, through the JEA, and through the type III collagen connective tissues and reaches closer to the bone.46 Because the probe penetrates deeper next to an implant compared with a tooth, one should take care not to contaminate the implant sulcus with bacteria from a diseased periodontal site (Table 18.4).
TABLE 18.4
Comparison of Tooth and Implant Support Structures
| Structure | Tooth | Implant |
| Connection to bone | Cementum, bone, periodontium | Osseointegration, bone functional ankyloses |
| Junctional epithelium | Hemidesmoses and basal lamina (lamina lucida and lamina densa zones) | Hemidesmosomes and basal lamina (lamina lucida, lamina densa, and sublamina lucida zones) |
| Connective tissue |
12 groups: six insert perpendicular to tooth surfaces ↓ collagen, ↑ fibroblasts |
Only two groups: parallel and circular fibers; no attachments to the implant surface ↑ collagen, ↓ fibroblasts |
| Biologic width | 2.04–2.91 mm | 3.08 mm (includes sulcus) |
| Vascularity | Greater; supraperiosteal, and periodontal ligament | Less periosteal |
| Probing depth | 3 mm in health | 2.5–5.0 mm (depending on previous soft tissue depth) |
| Bleeding on probing | More reliable | Less reliable |
The benefit of probing the implant sulcus has been challenged in the literature because sound scientific criteria for the rationale are lacking. The location of the probe tip subgingivally for a tooth depends on the pressure used, the presence of inflammation, and the angle at which the probe is introduced in the sulcus depth between the junctional epithelium and the root surface. The correct pressure recommended for probing is 20 g, yet conventional probing often exerts a force more than five times this level and greatly varies. The potential for damage to the fragile hemidesmosome attachment to the implant or marring of the implant surface exists during probing. In addition, reports in the literature suggest that the reproducibility of attachment level measurements may be questionable independently from the instrument used to perform the measurements.47,48 Many of these variables are similar for a dental implant. Unlike natural teeth, fixed implant prostheses with subgingival margins of crowns often have wide emergence profiles, making probe positioning difficult around most implant bodies.
The implant sulcus depth may be a reflection of the original soft tissue thickness of the area before implant placement. The posterior maxillary tissue can be thicker than 4 mm after tooth extraction and subsequent bone volume loss before implant placement. As a result, the tissue above the bone before implant insertion may be 4 mm thick or more. As a result of greater tissue thickness before surgery and a greater probing depth compared with teeth, the probing depth next to a healthy implant may be greater than that of a healthy natural tooth.
When the tissues are thick, gingivoplasty to reduce the flap thickness and pocket depth can be performed at the initial surgery. The advantage of the reduction in tissue thickness at this time is the tissue heals and matures as the bone-implant interface develops. However, thinning the flap at the initial surgery may cause greater loading of the implant body during healing from an overlying soft tissue–borne temporary prosthesis. After initial bone healing, the stage II uncovery surgery also may correct tissue thickness.
Lekholm et al found that the presence of deep pockets was not accompanied by accelerated marginal bone loss.49 Stable, rigid, fixed implants were reported with pocket depths ranging from 2 to 6 mm. Healthy, partially edentulous implant patients consistently exhibit greater probing depths around implants than around teeth. An increasing probing depth next to an implant is a more significant sign than a probing depth unrelated to a time interval because it usually signifies bone loss except in cases of gingival hyperplasia or hypertrophy. Probing using fixed reference points on the abutment or crown margin allows evaluation of crestal bone loss vs. tissue hypertrophy.
Despite the limitations, charting the attachment level in implant permucosal areas does aid the clinician in monitoring these regions. As the sulcus depth increases, the oxygen tension decreases. The bacteria in an implant sulcus are similar to those of a natural tooth. A toothbrush and daily hygiene procedures has been shown to be unable to clean a sulcus greater than 2 mm.50 Sulcus depths greater than 5 to 6 mm have a greater incidence of anaerobic bacteria (Table 18.5). As a consequence, this sulcus depth often requires gingivectomy or bone revision surgery. As a general rule, to enable the patient to perform effective daily hygiene, the ideal implant sulcus should be maintained at less than 5 mm.
TABLE 18.5
Subgingival Microflora Associated With Human Dental Implants
| Pocket Depth (mm) | ||
| Microflora | <5 | >6 |
| Spirochetes (%) | 2 | 32 |
| Motile rods (%) | 16 | 18 |
| Coccoids (%) | 64 | 30 |
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses