“The art of life is the art of avoiding pain; and he is the best pilot, who steers clearest of the rocks and shoals with which it is beset.” — Thomas Jefferson
This quote summarizes many patients’ attitudes when visiting the oral and maxillofacial surgeon. Unfortunately, many of the procedures commonly performed by oral surgeons result in moderate to severe pain. In fact, the third molar extraction pain model has become an accepted method of evaluating the management of acute postoperative pain. As clinicians we must be cognizant of our patients’ comfort during and after surgery. The importance of pain control following oral and maxillofacial surgery is evidenced by the growing body of literature aimed at evaluating postoperative quality of life outcomes. One study shows that the adverse impact on quality of life following third molar surgery is three times greater when patients are experiencing postoperative pain. Although this may seem obvious, its importance cannot be underestimated. Undergoing painless surgery and having limited postoperative pain is a major concern for our patients.
Pain is an unpleasant sensory experience perceived as arising from a specific location of the body and frequently associated with actual or potential tissue damage. In his review of acute and chronic craniofacial pain, Sessle describes pain as “a complex, multidimensional experience encompassing sensory-discriminatory, cognitive, affective, and motivational aspects.” In other words, an individual’s response to pain is dependent not only on the extent and site of tissue damage but also by its processing in the central nervous system. Pain should not be seen only as a burden. It has a useful function. It is a diagnostic tool for clinicians and acts as a protective mechanism for patients.
Pain management also plays an important role in developing the patient-physician relationship. Patient perception regarding pain, particularly in relation to oral and maxillofacial surgery, can be difficult for the surgeon to overcome. In fact, undue worry and an overestimation of the pain they will encounter can lead to the avoidance of treatment. Patient education is an essential part of any pain control strategy. By educating patients on pain expectations and the pain control regimen to be employed after surgery, we can better prepare them for their postoperative recovery and alleviate unnecessary anxiety.
The goal of this chapter is to give a comprehensive review of the management of acute postoperative pain in the oral and maxillofacial surgery patient. Although considerable attention has been given to nonpharmacologic methods of pain management (behavioral modification, music therapy, massage, acupuncture, transcutaneous nerve stimulation), this chapter will describe the most common pharmacotherapies used by oral and maxillofacial surgeons. In addition, the discussion will be geared toward acute pain management—pain that is of recent onset and limited duration (i.e., surgical pain). We start by examining the neurobiology and pathophysiology of pain. The major classes of analgesics are then described. Finally, specific strategies in the ambulatory and nonambulatory oral and maxillofacial surgery patient are given. Keeping in mind that evidence-based practice will continue to change the management of pain in our specialty, the basic principles will remain the same. It is the responsibility of the surgeon to: (1) develop an effective pain management strategy that is patient specific and takes into account both physiologic and psychosocial factors, (2) communicate that plan with the patient before surgery, and (3) adjust that strategy as necessary depending on patient feedback and clinical information. This ultimately leads to greater patient satisfaction and the building of a successful practice.
▪
NEUROANATOMY AND PATHOPHYSIOLOGY OF PAIN
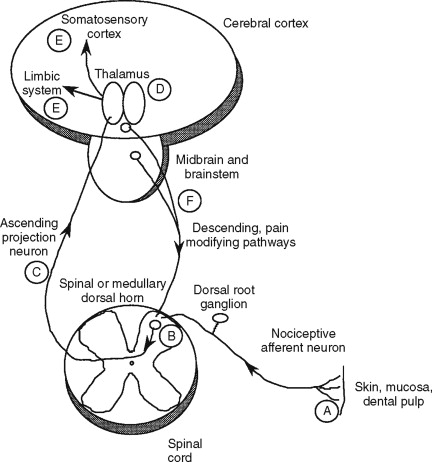
Tissue is inherently damaged during oral and maxillofacial surgery. Nociception is the electrical transmission of a noxious stimulus from a site of injury to higher brain centers. Following tissue damage outside of the craniofacial region, free nerve endings present in peripheral tissue are stimulated by mechanical, thermal, and chemical means. These free nerve endings act as afferent pain receptors, or nociceptors. After being stimulated, an electrical event is initiated and is propagated via primary afferent nerve fibers to the dorsal horn of the spinal cord via the dorsal root ganglia. The main two types of afferent nerve fibers associated with nociception are A-delta and C-polymodal fibers. A-delta fibers are lightly myelinated, have a larger diameter, and are fast conducting. They are primarily activated by mechanical stimuli and are characterized by sharp, stabbing, and shooting sensations. C fibers are termed polymodal because they are activated by mechanical, chemical, and thermal stimuli. They have a smaller diameter, are unmyelinated, transmit impulses at a slower rate, and are associated with dull, achy pains. After synapsing with second order neurons in the dorsal horn, the impulse then crosses to the contralateral ascending spinothalamic tract where it travels to the thalamus. From the thalamus, the impulse travels to higher brain centers, including the somatosensory cortex and limbic system. The transmission of this impulse may also be affected by descending pain-modifying pathways from the cerebral cortex, midbrain, and brainstem. These descending pathways generally synapse in the dorsal horn in the vicinity of second order ascending fibers and suppress excitatory transmission, thus inhibiting the transmission of pain sensation ( Figure 6-1 ).
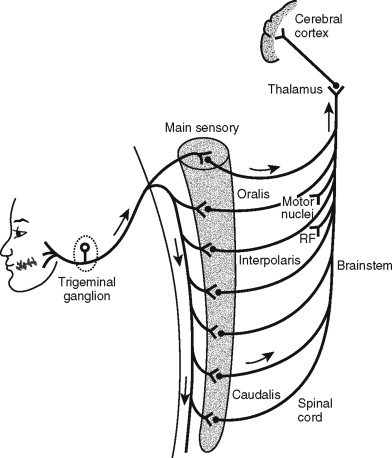
Pain sensation of oral and craniofacial origin follows a similar pathway to that outlined earlier. The main differences are in the specific nerves involved in transmission and the level of the central nervous system (CNS) at which they are transmitted to higher brain centers. The main sensory nerve of the orofacial region is the trigeminal nerve (cranial nerve V). There are also some tissues in the craniofacial region receiving afferent sensory innervation from other cranial nerves (cranial nerves VII, IX, X, and XII) and branches of the upper cervical spinal nerves (C1, C2, C3). After stimulating nociceptors in tissues innervated by the trigeminal nerve, an afferent impulse is directed toward the CNS via the trigeminal (Gasserian) ganglion. Whereas in the rest of the body the impulse travels to the dorsal root of the spinal cord, in the craniofacial region, sensory information is passed through a special area of the brainstem known as the trigeminal brainstem sensory nuclear complex (VBSNC). This is a bilateral, multinucleated structure in the dorsolateral brainstem extending from the pons to the upper cervical spinal cord. The VBSNC is divided into two major nuclei: the main sensory nucleus and the spinal tract nucleus. The spinal tract nucleus is further divided into the subnucleus oralis, the subnucleus interpolaris, and the subnucleus caudalis. After synapsing in the VBSNC, the impulse travels to the thalamus where it can be relayed to higher brain centers, including the somatosensory cortex and the limbic system. The impulse can also be directed to a variety of other locations in the brainstem and the spinal cord. Some of these connections are used in reflex pathways. Finally, descending modifying pathways are also present that can affect the transmission and sensation of pain ( Figure 6-2 ).
Inflammation plays an important role in the development of postoperative pain. Surgical manipulation of oral and facial tissues causes damage resulting in acute inflammation. As cells are disrupted, inflammatory mediators and metabolites are released into surrounding tissue beds. These factors include cytokines, substance P, prostaglandins, leukotrienes, kinins, histamine, and serotonin. Some of these factors can directly stimulate a nerve impulse in afferent pain fibers. Others, such as prostaglandins, serve to sensitize nociceptors by lowering their activation thresholds. This leads to the hyperalgesia found in surgically altered tissue.
As outlined previously, the sensation of pain is propagated by peripherally located receptors after tissue injury. Local mediators released into tissue beds contribute to the initiation of an electrical impulse directed toward the spinal cord and brainstem. From here, the impulse travels to higher brain centers where cognitive and emotional input is integrated and the quality of pain is perceived. The management of pain is directed at altering the peripheral and central mechanisms responsible for the propagation of pain impulses. These mechanisms should be kept in mind when designing a postoperative pain control regimen.
▪
OPIOID ANALGESICS
The term opioid refers to those compounds that have been synthetically manufactured to resemble the chemical structure of the naturally occurring alkaloids of the opium poppy. The natural by-products of opium, including morphine, codeine, and their semisynthetic derivatives, are known as opiates. The opiates can also be grouped under the single term opioid. The activity of all opioids is related to their binding to a special set of membrane-bound receptors. These receptors have been categorized into three groups based on their pharmacologic selectivity: mu, kappa, and delta. One method of classifying opioids is by their ability to bind opioid receptors. This classification groups opioids into agonists, antagonists, and agonist-antagonists. Agonists, such as morphine and codeine, elicit their analgesic effects by binding mu and kappa receptors. In contrast, antagonists bind opioid receptors, but do not stimulate them. Naloxone, a common narcotic reversal agent, is an opioid antagonist. Its ability to reverse the effects of opioid agonists is based on its higher binding affinity for opioid receptors. Finally, agonist-antagonist drugs act as agonists at one type of receptor and antagonists at others. For example, pentazocine acts as an agonist at kappa receptors and an antagonist at mu receptors.
The physiologic effect of opioids is dependent on the type and location of the receptors that they bind. Their analgesic effect is generated mainly by the inhibition of nociceptive impulses in the CNS. They have been found at various levels of the ascending pain pathway, including the dorsal horn of the spinal cord, the brainstem, the thalamus, and the somatosensory cortex. They have also been found in the midbrain and medulla, where they activate descending inhibitory pathways. Recent literature also points to a peripheral effect of opioid analgesia. At the cellular level, binding at opioid receptors may lead to one of several biochemical results: (1) decrease in calcium influx at afferent nerve terminals leading to decreased presynaptic neurotransmitter release, (2) increased potassium efflux leading to hyperpolarization of postsynaptic neurons and inhibition of impulse propagation, and (3) inhibition of GABAergic transmission in the brainstem leading to the excitation of descending modulating circuits.
In addition to analgesia, activation of opioid receptors can lead to several undesirable effects ( Box 6-1 ). The activation of mu receptors in respiratory centers of the brainstem leads to respiratory depression. This response is dose dependent and causes reductions in respiratory rate and minute volume. This is the most serious side effect of opioid administration, and overdose can lead to respiratory arrest and death. One of the more common unwanted effects of opioid use is nausea and vomiting. This response is produced by the activation of neurons in the chemoreceptor trigger zone of the medulla and is worse in ambulatory patients. Other CNS effects include mental clouding, sedation, and euphoria. All patients taking these medications should be cautioned against operating motor vehicles and making important decisions. Outside of the CNS, opioids can also have unwanted effects. In the gastrointestinal (GI) system, opioids decrease gastric and intestinal motility leading to constipation. The effects in the cardiovascular system can be both detrimental and advantageous. Opioids cause arteriolar vasodilation and can result in orthostatic hypotension. However, the decrease in cardiac demand caused by opioids can also be a benefit in patients experiencing angina or myocardial infarction. Opioids also cause an increase in smooth muscle tone of the bladder and urinary sphincter leading to urinary retention. Finally, itching may occur during opioid use as a result of the release of histamine from mast cells and possibly by the disinhibition of itch-specific neurons.
Respiratory depression
Nausea and vomiting
Mental clouding
Sedation
Euphoria
Constipation
Hypotension
Urinary retention
Pruritus
The concepts of tolerance, dependence, and addiction deserve mention in any discussion of opioid analgesics. Tolerance is a state of adaptation in which exposure to a drug induces changes that result in a decrease of the drug’s effect over time. The continual use of an opioid can lead to physiologic and psychological alterations that can cause withdrawal symptoms if the drug is discontinued. This is known as dependence. Addiction is a form of psychological dependence associated with the behavior patterns of obtaining and consuming the drug. Even though the postoperative use of opioid analgesics is usually for a limited duration, it is important for the oral and maxillofacial surgeon to keep these concepts in mind when managing pain. It is not uncommon to encounter patients with chronic pain conditions who may be tolerant to and dependent on opioids. In these situations, the choice of a nonopioid analgesic or an increase in the dose of an opioid may be warranted in treating acute postoperative pain. The key to the use of opioids is maximizing the benefit of analgesia while limiting any undesirable effects.
REVIEW OF OPIOIDS
Table 6-1 lists the most common opioids used by oral and maxillofacial surgeons. Oral administration is the most common route of administration in our population of patients. It is important to keep in mind that opioids absorbed in the GI tract are subject to first-pass hepatic metabolism. This limits their efficacy and increases interpatient variability when administered orally. Opioids may also be administered parenterally, intramuscularly, transdermally, transmucosally, subcutaneously, and rectally.
| Analgesic | Dose (mg) | Dosing Interval (hr) | Comments |
|---|---|---|---|
| AGONIST | |||
| Morphine | 10-30 | 4 | Standard analgesic dose used for potency comparisons |
| Codeine | 60 | 3-4 | Can be administered as a single agent or in combination with acetaminophen or aspirin |
| Hydrocodone | 5-10 | 4-5 | Only available in a combination formulation with either acetaminophen, aspirin, or ibuprofen |
| Dihydrocodeine | 16-32 | 4-5 | Only available in a combination formulation with either acetaminophen or aspirin |
| Oxycodone | 5-10 | 4-5 | Only available in a combination formulation with either acetaminophen or aspirin |
| Propoxyphene | 65-130 | 4-6 | Weak analgesic properties |
| MIXED AGONIST-ANTAGONIST | |||
| Pentazocine | 50-100 | 4-6 | Will precipitate an abstinence syndrome in patients dependent on opioids |
| OTHER | |||
| Tramadol | 50-100 | 4-6 | Not available in a combination formulation; although currently unscheduled by the Drug Enforcement Administration, abuse potential exists |
Morphine is considered the prototype for all opioids. Accordingly, it is the standard by which all opioids are measured. Table 6-2 compares the potencies of the common opioids. All doses are based on the pain relief elicited from 10 mg of IV morphine. Administration of morphine is indicated for severe pain, and thus its use in oral and maxillofacial surgery is limited. When administering this drug intravenously (IV), it is always wise to do so in a monitored setting. Doses should be started low (i.e., 1 to 2 mg IV) and then titrated to effect. It is important to remember that opioids have no ceiling effect for analgesia. Their administration is limited by the undesirable effects that occur as doses are increased.
| Drug | IM/IV (mg) | PO (mg) |
|---|---|---|
| Morphine | 10 | 30-60 |
| Meperidine | 75 | 300 |
| Fentanyl | 0.1-0.2 | N/A |
| Codeine | 120 | 200 |
| Pentazocine | 30 | 150 |
| Oxycodone | N/A | 20-30 |
| Hydrocodone | N/A | 30 |
| Propoxyphene napsylate | N/A | 200 |
| Tramadol | N/A | 100-150 |
Fentanyl and meperidine are two opioids typically administered via the IV route. Fentanyl is a fast-acting opioid that is approximately 100 times more potent than morphine. It is indicated for severe pain and is a common agent used for conscious sedation in oral and maxillofacial surgical procedures. The respiratory depressive effects limit its use in the postoperative period. Like morphine, this drug should only be administered in a monitored setting. In contrast, meperidine is 10 times less potent than morphine, has a slower onset, and lasts about twice as long as morphine and fentanyl. Its use has generally fallen out of favor because of its undesirable effects. Normeperidine is a neurotoxic metabolite of meperidine. It has been shown to cause dysphoria, tremors, and generalized seizures. In addition, use of meperidine with monoamine oxidase inhibitors (MAOIs) can cause hypertensive crisis, hyperpyrexia, and cardiovascular collapse.
Codeine is another naturally occurring alkaloid that is structurally similar to morphine. It is typically given in the oral form. Codeine is a weak mu agonist. However a small percentage of codeine is converted to morphine after administration. In the oral form, codeine is about four times less potent than morphine. Doses greater than 60 mg are not recommended because the side effects above this dose outweigh the analgesic benefits. Hydrocodone and oxycodone are semisynthetic opioids that are similar to morphine and codeine. These drugs are available only in the oral formulation and are about 6.5 and 10 times more potent than codeine, respectively.
Propoxyphene is an opioid from the diphenylheptane chemical class of opioids. It is structurally similar to methadone, a compound typically prescribed to treat heroin dependence and addiction. Propoxyphene has about two-thirds of the potency of codeine. In addition to the common side effects of opioids, it also causes cardiotoxicity, pulmonary edema, and arrythmias. It is therefore not generally considered a first-line choice for opioid analgesia. Because propoxyphene is from a different chemical class than the more commonly prescribed opioids, it may be of some use in patients that exhibit sensitivities to those medications.
Pentazocine is an opioid agonist-antagonist. It exhibits mu antagonist activity and kappa agonist activity. It has less respiratory depressive effects than the pure opioid agonists. However, because it antagonizes mu receptors, it can precipitate opioid withdrawal symptoms in patients concurrently taking mu agonists. When crushed and given IV, pentazocine can cause marked euphoria. To prevent illicit use of this drug, it is commonly formulated to include the opioid antagonist naloxone. When given orally, naloxone has minimal activity. However, when taken IV, the naloxone will antagonize the euphoric effects caused by pentazocine.
Tramadol is an analgesic drug marketed for treatment of moderate to severe pain. It is a synthetic compound chemically unrelated to the other opioid classes. It appears to have several mechanisms of action: (1) it is a weak mu agonist; (2) it inhibits the serotonin and norepinephrine reuptake systems. Unlike pure opioids, respiratory effects and abuse potential are minimal. The efficacy of tramadol for pain relief following dental extractions appears to be comparable with codeine and its oral combinations. It is typically administered in doses of 50 to 100 mg and has been associated with side effects such as headache, nausea, vomiting, somnolence, and seizures.
Most oral opioids are offered in combination with nonsteroidal antiinflammatory medications. In the acute postoperative period, inflammation occurring after the surgical manipulation of tissue is the major cause of pain. Combining an opioid with a nonsteroidal antiinflammatory drug (NSAID) attacks multiple levels of the pain pathway and helps reduce the dose of opioid necessary to reach the desired amount of analgesia. It also limits unwanted side effects seen with higher opioid doses.
▪
OPIOID ANALGESICS
The term opioid refers to those compounds that have been synthetically manufactured to resemble the chemical structure of the naturally occurring alkaloids of the opium poppy. The natural by-products of opium, including morphine, codeine, and their semisynthetic derivatives, are known as opiates. The opiates can also be grouped under the single term opioid. The activity of all opioids is related to their binding to a special set of membrane-bound receptors. These receptors have been categorized into three groups based on their pharmacologic selectivity: mu, kappa, and delta. One method of classifying opioids is by their ability to bind opioid receptors. This classification groups opioids into agonists, antagonists, and agonist-antagonists. Agonists, such as morphine and codeine, elicit their analgesic effects by binding mu and kappa receptors. In contrast, antagonists bind opioid receptors, but do not stimulate them. Naloxone, a common narcotic reversal agent, is an opioid antagonist. Its ability to reverse the effects of opioid agonists is based on its higher binding affinity for opioid receptors. Finally, agonist-antagonist drugs act as agonists at one type of receptor and antagonists at others. For example, pentazocine acts as an agonist at kappa receptors and an antagonist at mu receptors.
The physiologic effect of opioids is dependent on the type and location of the receptors that they bind. Their analgesic effect is generated mainly by the inhibition of nociceptive impulses in the CNS. They have been found at various levels of the ascending pain pathway, including the dorsal horn of the spinal cord, the brainstem, the thalamus, and the somatosensory cortex. They have also been found in the midbrain and medulla, where they activate descending inhibitory pathways. Recent literature also points to a peripheral effect of opioid analgesia. At the cellular level, binding at opioid receptors may lead to one of several biochemical results: (1) decrease in calcium influx at afferent nerve terminals leading to decreased presynaptic neurotransmitter release, (2) increased potassium efflux leading to hyperpolarization of postsynaptic neurons and inhibition of impulse propagation, and (3) inhibition of GABAergic transmission in the brainstem leading to the excitation of descending modulating circuits.
In addition to analgesia, activation of opioid receptors can lead to several undesirable effects ( Box 6-1 ). The activation of mu receptors in respiratory centers of the brainstem leads to respiratory depression. This response is dose dependent and causes reductions in respiratory rate and minute volume. This is the most serious side effect of opioid administration, and overdose can lead to respiratory arrest and death. One of the more common unwanted effects of opioid use is nausea and vomiting. This response is produced by the activation of neurons in the chemoreceptor trigger zone of the medulla and is worse in ambulatory patients. Other CNS effects include mental clouding, sedation, and euphoria. All patients taking these medications should be cautioned against operating motor vehicles and making important decisions. Outside of the CNS, opioids can also have unwanted effects. In the gastrointestinal (GI) system, opioids decrease gastric and intestinal motility leading to constipation. The effects in the cardiovascular system can be both detrimental and advantageous. Opioids cause arteriolar vasodilation and can result in orthostatic hypotension. However, the decrease in cardiac demand caused by opioids can also be a benefit in patients experiencing angina or myocardial infarction. Opioids also cause an increase in smooth muscle tone of the bladder and urinary sphincter leading to urinary retention. Finally, itching may occur during opioid use as a result of the release of histamine from mast cells and possibly by the disinhibition of itch-specific neurons.
Respiratory depression
Nausea and vomiting
Mental clouding
Sedation
Euphoria
Constipation
Hypotension
Urinary retention
Pruritus
The concepts of tolerance, dependence, and addiction deserve mention in any discussion of opioid analgesics. Tolerance is a state of adaptation in which exposure to a drug induces changes that result in a decrease of the drug’s effect over time. The continual use of an opioid can lead to physiologic and psychological alterations that can cause withdrawal symptoms if the drug is discontinued. This is known as dependence. Addiction is a form of psychological dependence associated with the behavior patterns of obtaining and consuming the drug. Even though the postoperative use of opioid analgesics is usually for a limited duration, it is important for the oral and maxillofacial surgeon to keep these concepts in mind when managing pain. It is not uncommon to encounter patients with chronic pain conditions who may be tolerant to and dependent on opioids. In these situations, the choice of a nonopioid analgesic or an increase in the dose of an opioid may be warranted in treating acute postoperative pain. The key to the use of opioids is maximizing the benefit of analgesia while limiting any undesirable effects.
REVIEW OF OPIOIDS
Table 6-1 lists the most common opioids used by oral and maxillofacial surgeons. Oral administration is the most common route of administration in our population of patients. It is important to keep in mind that opioids absorbed in the GI tract are subject to first-pass hepatic metabolism. This limits their efficacy and increases interpatient variability when administered orally. Opioids may also be administered parenterally, intramuscularly, transdermally, transmucosally, subcutaneously, and rectally.
| Analgesic | Dose (mg) | Dosing Interval (hr) | Comments |
|---|---|---|---|
| AGONIST | |||
| Morphine | 10-30 | 4 | Standard analgesic dose used for potency comparisons |
| Codeine | 60 | 3-4 | Can be administered as a single agent or in combination with acetaminophen or aspirin |
| Hydrocodone | 5-10 | 4-5 | Only available in a combination formulation with either acetaminophen, aspirin, or ibuprofen |
| Dihydrocodeine | 16-32 | 4-5 | Only available in a combination formulation with either acetaminophen or aspirin |
| Oxycodone | 5-10 | 4-5 | Only available in a combination formulation with either acetaminophen or aspirin |
| Propoxyphene | 65-130 | 4-6 | Weak analgesic properties |
| MIXED AGONIST-ANTAGONIST | |||
| Pentazocine | 50-100 | 4-6 | Will precipitate an abstinence syndrome in patients dependent on opioids |
| OTHER | |||
| Tramadol | 50-100 | 4-6 | Not available in a combination formulation; although currently unscheduled by the Drug Enforcement Administration, abuse potential exists |
Morphine is considered the prototype for all opioids. Accordingly, it is the standard by which all opioids are measured. Table 6-2 compares the potencies of the common opioids. All doses are based on the pain relief elicited from 10 mg of IV morphine. Administration of morphine is indicated for severe pain, and thus its use in oral and maxillofacial surgery is limited. When administering this drug intravenously (IV), it is always wise to do so in a monitored setting. Doses should be started low (i.e., 1 to 2 mg IV) and then titrated to effect. It is important to remember that opioids have no ceiling effect for analgesia. Their administration is limited by the undesirable effects that occur as doses are increased.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


