Fig. 15.1
Structure of tetracyclines
Tetracycline antibiotics are protein synthesis inhibitors which inhibit the binding of aminoacyl-tRNA to the mRNA-ribosome complex. They do this mainly by binding to the 30S ribosomal subunit in the mRNA translation complex [13].
Properties
Tetracyclines, including tetracycline HCl, minocycline, demeclocycline, and doxycycline, are a group of broad-spectrum antibiotics that are effective against a wide range of microorganisms [13, 14]. Tetracyclines are bacteriostatic in nature [15]. This property may be advantageous because, in the absence of bacterial cell lysis, antigenic by-products such as endotoxin are not released [16]. Tetracyclines also have many unique properties other than their antimicrobial action, such as the inhibition of mammalian collagenases, which prevent tissue breakdown [17], and the inhibition of clastic cells [17, 18], which results in antiresorptive activity [18]. Inflammatory diseases such as periodontitis include an excess of tissue collagenases which may be blocked by tetracyclines, thus leading to enhanced formation of collagen and bone [16].
Applications in Endodontics
In endodontics, tetracyclines have been used as part of an irrigant to remove the smear layer from instrumented root canal walls [16, 19], for irrigation of retrograde cavities during periapical surgical procedures [20], and as an intracanal medicament [21].
Barkhordar et al. [16] showed that doxycycline HCl eliminated smear layer in a concentration-dependent manner with 100 mg/ml doxycycline being more effective than lower concentrations. In another investigation, Haznedaroğlu and Ersev [19] reported that tetracycline was as effective as citric acid in removing the smear layer. Barkhordar and Russell [20] evaluated the effect of doxycycline on the apical penetration of dye through the margins of retrograde fillings. The teeth with retrograde IRM or amalgam fillings placed following doxycycline irrigation had significantly less dye penetration than those that were not irrigated with doxycycline.
Pinheiro et al. [22] evaluated the antibiotic susceptibility of Enterococcus faecalis isolates from canals of root-filled teeth with periapical radiolucencies. The antibiotics were benzylpenicillin, amoxicillin, amoxicillin with clavulanic acid, erythromycin, azithromycin, vancomycin, chloramphenicol, tetracycline, doxycycline, ciprofloxacin, and moxifloxacin. The vast majority (85.7 %) of the isolates were susceptible to tetracycline and doxycycline.
Based on the hypotheses that microorganisms can reach the apical area of recently replanted teeth from the oral cavity (or from contaminated root surfaces during the extra-oral time) and that tetracyclines can potentially inhibit this route of bacterial contamination, Cvek et al. [23] developed a protocol for the topical treatment of exposed roots with doxycycline before replantation of avulsed teeth. Their aim was to eliminate the microorganisms from the root surface via direct local application of the antibiotic in order to decrease the frequency and severity of the inflammatory response. They showed that topical doxycycline significantly increased the chances of successful pulp revascularization and decreased the number of microorganisms that could be isolated from the root canals. They also reported a decreased frequency of ankylosis, external replacement resorption, and external inflammatory resorption. The beneficial effect of soaking a tooth in doxycycline has also been confirmed by Yanpiset and Trope [24].
Using laser Doppler flowmetry (LDF), radiography, and histology, a study investigated the effect of topical antibiotic treatment on pulp revascularization in replanted teeth in a dog model [25]. After extraction, the teeth were kept dry for 5 min and either covered with minocycline, soaked in doxycycline, or soaked in saline, and then they were replanted. Teeth in the positive control group were not extracted. Postoperative radiographs and LDF readings were obtained for 2 months after replantation. After sacrifice of the animals, the jaws were collected and processed for light microscopy. Pre- and post-replantation LDF readings and radiographs and the histological findings were analyzed to assess revascularization. Pulp revascularization occurred in 91 % of the teeth treated with minocycline, 73 % of those soaked in doxycycline, and only 33 % of the teeth soaked in saline [25].
Bryson et al. [26] evaluated the effect of minocycline on the healing of replanted dog teeth after extended dry times of 60 min. Their results indicated that the roots with and without minocycline treatment showed no significant differences in the remaining root mass or the percentage of favorably healed root surfaces. In addition, no benefit was found from the use of topically applied minocycline in the attenuation or prevention of external root resorption. The lack of significant differences is likely to have been a result of the extended dry period before replantation as most of the periodontal ligament cells would have died within this time period and therefore external replacement resorption is the typical result.
Further details and applications of tetracyclines in endodontics and dental traumatology are outlined below in the sections regarding Ledermix paste and triple antibiotic pastes.
Substantivity of Tetracyclines
Tetracyclines readily attach to dentin and are subsequently released without losing their antibacterial activity [15]. This property creates a reservoir of active antibacterial agent, which is then released from the dentin surface in a slow and sustained manner. In an in vivo periodontal study, Stabholz et al. [27] compared the antibacterial substantivity of two concentrations of tetracycline HCl (50 mg/ml, 10 mg/ml) and 0.12 % chlorhexidine. Their findings showed that both concentrations of tetracycline demonstrated residual antibacterial activity and the antibacterial substantivity of the three solutions in descending order was 50 mg/ml tetracycline >10 mg/ml tetracycline >0.12 % CHX.
Abbott et al. [28] demonstrated that tetracyclines form a strong reversible bond with the dental hard tissues and that they exhibit slow release and diffusion through dentin over an extended period of time up to at least 12 weeks. [89] compared the antibacterial substantivity of 2 % CHX, 100 mg/ml doxycycline HCl, and 2.6 % NaOCl in bovine root dentin (Figure 15.2) over five experimental periods of 0, 7, 14, 21, and 28 days in vitro. Their findings indicated that after 7 days, the NaOCl and doxycycline groups showed the lowest and the highest number of colony-forming units (CFUs), respectively. However, after the longer time periods, the CHX group showed the lowest number of CFUs.
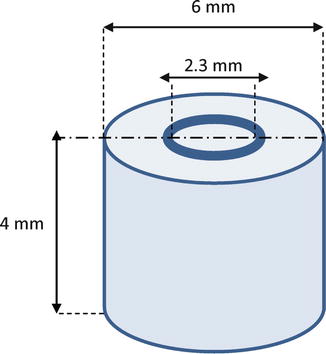

Fig. 15.2
Schematic view of used dentin tubes (Adopted from Mohammadi and Shahriari [40])
Mohammadi et al. [29] evaluated the antibacterial substantivity of three concentrations of doxycycline HCl (100, 50, and 10 mg/ml) in bovine root dentin over five experimental periods of 0, 7, 14, 21, and 28 days. At 7 days, the 100 mg/ml group and the 10 mg/ml group showed the lowest and highest numbers of CFUs, respectively. In each group, the numbers of CFUs increased significantly over time (Table 15.1).
Table 15.1
Means of the CFU and the standard deviation of E. faecalis in experimental groups (three concentrations of doxycycline) [29]
|
Day 0
|
Day 7
|
Day 14
|
Day 21
|
Day 28
|
|
|---|---|---|---|---|---|
|
100 mg/ml
|
0.40 ± 0.69
|
4.66 ± 2.34
|
9.70 ± 2.75
|
20.20 ± 3.22
|
44.44 ± 5.52
|
|
50 mg/ml
|
0.50 ± 3.97
|
9.00 ± 3.74
|
15.40 ± 4.55
|
37.00 ± 5.33
|
59.66 ± 5.36
|
|
10 mg/ml
|
4.70 ± 3.68
|
16.11 ± 8.05
|
37.40 ± 8.99
|
61.80 ± 11.11
|
88.55 ± 5.50
|
MTAD
BioPure (Dentsply, Tulsa Dental, Tulsa, OK, USA), otherwise known as MTAD, was introduced by Torabinejad et al. [15]. It is composed of 3 % doxycycline, 4.25 % citric acid, and a detergent (0.5 % polysorbate 80) [15].
Antimicrobial Activity
Several studies have evaluated the effectiveness of MTAD for disinfection of root canals. Torabinejad et al. [15] showed that MTAD was able to remove the smear layer and was effective against E. faecalis [30–32]. Using a human tooth model, Shabahang et al. [32] showed that the use of MTAD was more effective than 5.25 % NaOCl for disinfecting root canals. Torabinejad et al. [30] also demonstrated that MTAD was significantly more effective than the combination of NaOCl and EDTA against E. faecalis. Kho and Baumgartner [33] showed consistent disinfection of infected root canals when a combination of 5.25 % NaOCl/15 % EDTA was used. However, the combination of 1.3 % NaOCl/BioPure MTAD left nearly 50 % of the canals contaminated with E. faecalis. Krause et al. [34] compared the antimicrobial effect against E. faecalis of MTAD, two of its components (doxycycline and citric acid), and sodium hypochlorite in two in vitro models using two different methods. In the tooth model, NaOCl and doxycycline were more effective than the control in killing E. faecalis at shallow bur depths into dentin, but at deeper bur depths, the NaOCl was superior. In the agar diffusion model, NaOCl produced less inhibition of bacteria than MTAD or doxycycline. Ghoddusi et al. [35] indicated that removing the smear layer using MTAD as a final irrigant delayed bacterial penetration of filled root canals. Using the agar diffusion method, Davis et al. [36] determined that MTAD was significantly more effective than 5.25 % NaOCl, 2 % CHX, and Dermacyn against E. faecalis. Newberry et al. [37] showed that MTAD inhibited most strains of E. faecalis growth when diluted 1:8,192 times and it killed most strains of E. faecalis when diluted 1:512 times. Shabahang et al. [38] showed that the addition or substitution of chlorhexidine for doxycycline did not negatively impact the efficacy of MTAD. However, the substitution of this antimicrobial agent for doxycycline significantly reduced the efficacy of the solution. Furthermore, the contents of the root canal system may inhibit or decrease the antibacterial activity of MTAD. Portenier et al. [39] investigated the inhibitory effects of dentin and bovine serum albumin (BSA) on the antibacterial activity of MTAD and found that the presence of dentin or BSA caused a marked delay in the killing of bacteria.
Substantivity of MTAD
Tetracyclines (including doxycycline) readily attach to dentin and are subsequently released without losing their antibacterial activity [15]. The presence of doxycycline in MTAD suggests that MTAD may have some substantive antimicrobial action [15]. In an in vitro study using a human tooth model, Mohammadi and Shahriari [40] showed that, during a 4-week period, the substantivity of MTAD was significantly greater than CHX and NaOCl (Table 15.2). In another study, the substantivity of 100 % MTAD was significantly greater than the two other concentrations of MTAD [41]. Tay et al. [42] found that when MTAD was applied to 1.3 % NaOCl-irrigated dentin, its antimicrobial substantivity was reduced. They attributed this phenomenon to the oxidation of MTAD by NaOCl in a manner similar to the peroxidation of tetracycline by reactive oxygen species.
Table 15.2
Means of the CFU and the standard deviation of E. faecalis in the experimental groups [40]
|
Day 0
|
Day 7
|
Day 14
|
Day 21
|
Day 28
|
|
|---|---|---|---|---|---|
|
NaOCl
|
0.31 ± 0.58
|
17.16 ± 7.05
|
34.40 ± 8.79
|
66.78 ± 10.11
|
95.25 ± 5.61
|
|
CHX
|
3.56 ± 3.72
|
10.35 ± 3.77
|
14.49 ± 4.67
|
34.35 ± 4.22
|
51.53 ± 5.35
|
|
MTAD
|
0.70 ± 3.85
|
4.46 ± 2.24
|
8.68 ± 2.71
|
19.25 ± 3.49
|
40.44 ± 5.42
|
MTAD and Biofilms
Clegg et al. [43] reported that 6 % NaOCl was the only irrigant capable of both rendering bacteria nonviable and physically removing the biofilm. Dunavant et al. [44] showed that MTAD killed 16.08 % of the bacterial cells in E. faecalis biofilms, while Giardino et al. [45] showed that MTA was not able to disintegrate and remove bacterial biofilms.
In summary, based on the available literature, MTAD does not appear to be effective against bacterial biofilms.
Smear Layer Removal and Effect on Dentin
Torabinejad et al. [15] showed that MTAD was an effective solution for the removal of the smear layer and that it did not significantly change the structure of the dentinal tubules when root canals were irrigated with NaOCl, followed by a final rinse of MTAD. In another study [30], they showed that although MTAD removed most of the smear layer when used as an intracanal irrigant, some remnants of the organic component of the smear layer remained scattered on the surface of the root canal walls. The effectiveness of MTAD in completely removing the smear layer was enhanced when low concentrations of NaOCl were used as intracanal irrigants before using MTAD as a final rinse. Lotfi et al. [46] showed that MTAD could not remove the smear layer and their regimen did not significantly change the structure of the dentinal tubules [30]. On the other hand, Tay et al. [47] found that both irrigants created a zone of demineralized collagen matrices in eroded dentin and around the dentinal tubules, with the mildly acidic BioPure MTAD being more aggressive than EDTA. These demineralized dentin zones create the opportunity for dentin hybridization by infiltration of hydrophilic adhesives/sealers. However, the potential consequences of compaction of hydrophobic sealers against air-dried, collapsed collagen matrices, and hydrolytic degradation of incompletely infiltrated matrices remain unresolved. In an ultrastructural study, Tay et al. [47] showed that MTAD created a thicker demineralized dentin matrix (5–6 μm) than EDTA (1–2 μm). De-Deus et al. [48] found that the demineralization kinetics prompted by MTAD were significantly faster than those prompted by a 17 % EDTA solution.
There is only one study on the effect of MTAD on dentin. Machnick et al. [49] evaluated the effect of MTAD on the flexural strength and modulus of elasticity of dentin. Their findings showed that there was no significant difference in flexural strength and modulus of elasticity between the dentin specimens exposed to saline or MTAD.
MTAD and Dentin Bonding (Anti-collagenolytic Activity)
Machnick et al. [50] compared the effect of MTAD and phosphoric acid on the bond strength to enamel and dentin using a conventional OptiBond Solo Plus dentin adhesive system. They reported that teeth endodontically treated with the MTAD protocol for clinical use (20 min 1.3 % NaOCl/5 min MTAD) might not need any additional dentin conditioning prior to the application of the dental adhesive. Garcia-Godoy et al. [51] evaluated the structure of the hybrid layer formed after the use of EDTA or MTAD solutions when used as a final rinse. Findings showed that the BioPure MTAD hybrid layer was thicker than the 17 % EDTA hybrid layer. Both the BioPure MTAD and EDTA caused collapse of the dentin matrix structure, which impeded sealer infiltration and the formation of high-quality hybrid layer bonding. The hybrid layers created in smear layer-covered dentin exhibited less potential for fluid penetration than the MTAD or EDTA hybrid layer. It was also shown that neither EDTA nor MTAD significantly improved Epiphany-dentin bond strengths when compared with NaOCl used alone [52]. Yurdaguven et al. [53] showed that the bonding of Clearfil SE Bond to coronal dentin was significantly reduced after using MTAD to irrigate the root canal system.
In summary, due to its broad-spectrum MMP-inhibitory effect, MTAD can significantly improve the stability of the resin-dentin bond.
Toxicity of MTAD
There are few studies regarding the toxicity of MTAD. Zhang et al. [54] examined the cytotoxicity of MTAD compared with that of commonly used irrigants and medicaments. L929 fibroblasts were grown on cell culture plates and placed in contact with various concentrations of test irrigants and medicaments. The cytotoxicity of these materials was evaluated 24 h after incubation using MTT assay. Results showed that MTAD was less cytotoxic than eugenol, 3 % H2O2, Ca(OH)2 paste, 5.25 % NaOCl, Peridex, and EDTA, while it was more cytotoxic than 2.63, 1.31, and 0.66 % NaOCl. Yasuda et al. [55] evaluated the cytotoxicity of MTAD on MC3T3-E1 and periodontal ligament cells at various concentrations. They reported that it was less cytotoxic and did not affect differentiation into osteoblasts compared with other irrigants such as H2O2, NaOCl, EDTA, and chlorhexidine.
Tetraclean
Tetraclean (Ogna Laboratori Farmaceutici, Muggiò (Mi), Italy), like MTAD, is a mixture of an antibiotic, an acid, and a detergent. However, the concentration of the antibiotic, doxycycline (50 mg/ml), and the type of detergent (polypropylene glycol) differ from those of MTAD [56]. Giardino et al. [57] compared the surface tension of 17 % EDTA, Cetrexidin, SmearClear, 5.25 % NaOCl, MTAD, and Tetraclean. The NaOCl and EDTA had the highest surface tensions, whereas Cetrexidin and Tetraclean had the lowest values.
Antibacterial Activity
There are only a few studies on the antibacterial activity of Tetraclean. Giardino et al. [45] compared the antimicrobial efficacy of 5.25 % NaOCl, MTAD, and Tetraclean against an E. faecalis biofilm generated on cellulose nitrate membrane filters. Only the NaOCl could disaggregate and remove the biofilm at every time interval tested although treatment with Tetraclean caused a high degree of biofilm disaggregation at each time interval when compared with MTAD [45]. Neglia et al. [58] showed that Tetraclean was very effective against E. faecalis in vitro.
Ardizzoni et al. [59] evaluated the effectiveness of Tetraclean against E. faecalis using an agar diffusion test and showed that it was 100 % effective against 54 clinical isolates at dilutions up to 1:256. Giardino et al. [60] showed that Tetraclean was more effective than CHX against common endodontic bacteria. Pappen et al. [61] demonstrated that Tetraclean was more effective than MTAD against E. faecalis in planktonic culture and in mixed species in an in vitro biofilm. Using the agar diffusion test, Poggio et al. [62] showed that the efficacy of Tetraclean against Enterococcus faecalis, Streptococcus mutans, and Staphylococcus aureus was significantly better than NaOCl, Chloreximid, and hydrogen peroxide. Mohammadi et al. [63] investigated the efficacy of sodium hypochlorite, chlorhexidine, Tetraclean, Hypoclean, and Chlor-XTRA against Enterococcus faecalis, Candida albicans, Actinomyces israelii, Pseudomonas aeruginosa, and Lactobacillus casei using the agar diffusion method. According to their findings, Hypoclean was the most effective irrigant against C. albicans, P. aeruginosa, and L. casei.
Substantivity of Tetraclean
Mohammadi et al. [64] demonstrated that the substantivity of Tetraclean was significantly higher than MTAD and it was retained in root canal dentin for at least 28 days (Table 15.3). In additional studies, Mohammadi et al. [65] showed that the substantivity of Tetraclean was significantly greater than Hypoclean and 5.25 % NaOCl, and there was a direct relationship between dentin treatment time with Tetraclean and its substantivity [66]. Pretreatment of dentin with NaOCl significantly decreased the substantivity of Tetraclean [67].
Table 15.3
Mean of the CFU and the standard deviations of E. faecalis in the experimental groups [64]
|
Day 0
|
Day 7
|
Day 14
|
Day 21
|
Day 28
|
|
|---|---|---|---|---|---|
|
Tetraclean
|
0.00 ± 0.00
|
0.00 ± 0.00
|
0.37 ± 0.65
|
6.68 ± 2.59
|
15.35 ± 3.21
|
|
MTAD
|
0.71 ± 3.79
|
4.41 ± 2.21
|
8.74 ± 2.75
|
19.20 ± 3.41
|
39.55 ± 5.43
|
|
NaOCl
|
0.29 ± 0.57
|
17.13 ± 7.02
|
33.42 ± 8.72
|
65.71 ± 10.14
|
93.22 ± 5.64
|
Smear Layer Removal Ability
Poggio et al. [68] compared the demineralizing capability on root canal dentin of Tetraclean, Largal Ultra, 17 % ethylenediaminetetraacetic acid and Tubuliclean in vitro. Results indicated that the higher release of Ca+2 observed in samples treated with Tetraclean demonstrated its significantly higher demineralizing capability compared to the other irrigants tested.
Ledermix Paste
Ledermix paste is a glucocorticosteroid-antibiotic compound which was developed and was released for sale in Europe by Lederle Pharmaceuticals in 1962 [69]. The sole reason for adding the antibiotic component to Ledermix paste was to compensate for what was perceived to be a possible corticoid-induced reduction in the host immune response. Schroeder and Triadan initially incorporated chloramphenicol in their first trials, but when Lederle Pharmaceuticals became the manufacturer, the antibiotic was changed to demeclocycline HCl. Today, Ledermix paste remains a combination of the same tetracycline antibiotic, demeclocycline HCl (at a concentration of 3.2 %), and a corticosteroid, triamcinolone acetonide (concentration 1 %), in a polyethylene glycol base [69].
The two therapeutic components of Ledermix paste (i.e., triamcinolone and demeclocycline) are capable of diffusing through dentinal tubules and cementum to reach the periodontal and periapical tissues [70]. Abbott et al. [28] showed that dentinal tubules were the major supply route of the active components to the periradicular tissues, while the apical foramen was not as significant as a supply route. Various factors can affect the supply of the active components to the periradicular tissues – these include the presence or absence of the smear layer [71], the presence or absence of cementum [71], and the presence of other materials within the canal, for example, calcium hydroxide [72, 73]. The concentration of demeclocycline within Ledermix paste itself (i.e., as it would be when placed within the root canal) is high enough to be effective against susceptible species of bacteria [74]. However, within the peripheral parts of the dentine and in the periradicular tissues, the concentration achieved through diffusion is insufficient to inactivate bacteria, especially over time [74
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


