Lipids
The Condensed Energy
• Describe how fatty acids affect the properties of fat.
• Explain the functions of fats in the body and how these affect oral health.
• Identify dietary sources for saturated, monounsaturated, polyunsaturated, omega-3 and trans fatty acids, and cholesterol.
• Calculate the recommended amount of dietary fat.
• Plan appropriate interventions when dietary modification of fat intake has been recommended to a patient.
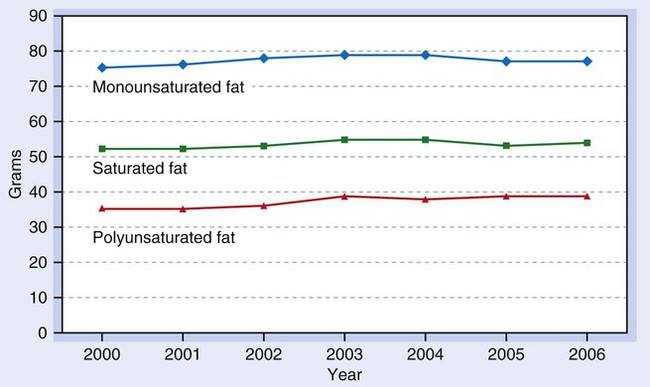
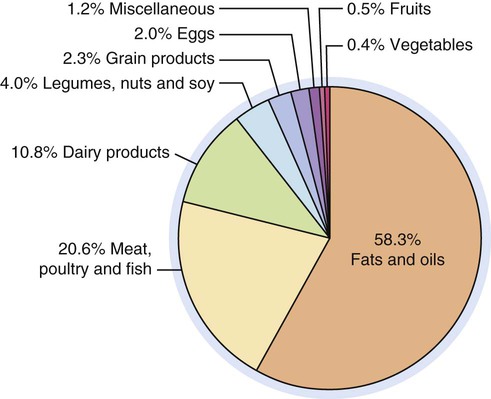
Added fats and oils provide more kilocalories in the average American diet than any other food group. Examination of U.S. food supply trends indicates total fat intake, which remains at a high level, increased slightly since 2000. The trend reflects a very slight increase in fats provided by vegetable oils (Fig. 6-1).1 Between 1977 and 2008, daily consumption of total fat declined from 39.7% to 33.4% of total caloric intake.2 Consumers have become more aware of healthy food choices, but changes in eating patterns are difficult. Food manufacturers, producers, and grocers have responded to concerns by (a) trimming fat from meats, (b) providing leaner cuts of beef and pork, (c) replacing tropical oils and trans fats in processed foods, and (d) manufacturing foods containing less fat. In addition, some consumers have increased their consumption of fish and poultry and substituted lower fat milk for whole milk. The fat content of very lean beef and pork cuts currently compares favorably with a skinless chicken breast. Added fats and oils provide most of the kilocalories Americans consume (Fig. 6-2).
Classification
Fats in the diet should actually be called lipids. Lipids contain the same three elements as carbohydrates: carbon, hydrogen, and oxygen. Lipids contain less oxygen in proportion to hydrogen and carbon than carbohydrates. The structure and function of lipids are covered in detail in Chapter 2 and on the Evolve website. Because of their structure, they provide more energy per gram than either carbohydrates or proteins.
Chemical Structure
Triglycerides are composed of fatty acids and glycerol, as shown:
< ?xml:namespace prefix = "mml" />
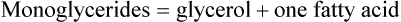
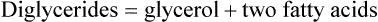
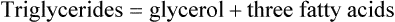
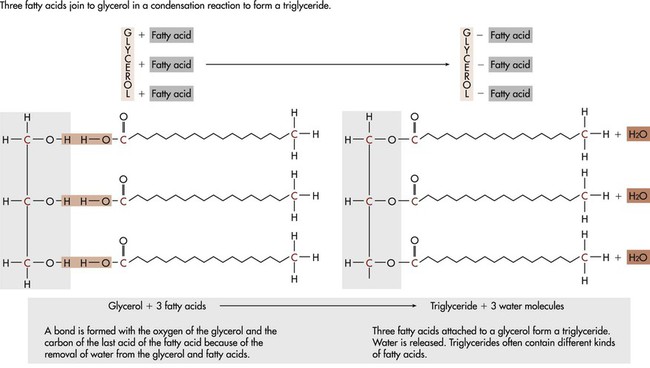
A fatty acid is a chain of carbon atoms attached to hydrogen atoms with an acid grouping on one end. Glycerol is the alcohol portion of a triglyceride to which the fatty acids attach. Triglycerides are the most common fat present in animal or protein foods (Fig. 6-3). Monoglycerides and diglycerides are found in the small intestine and result from the breakdown of triglycerides during digestion. Free fatty acids, monoglycerides, and glycerol can cross cell membranes.
Saturated Fatty Acids
As discussed in Chapter 2, fatty acids are classified according to their degree of saturation. Saturation of a fatty acid depends on the number of hydrogen atoms attached to the carbon chain. Saturated fatty acids (SFAs) contain only single bonds, with each carbon atom having two hydrogen atoms attached to it (see Chapter 2, Fig. 2-15). Palmitic and stearic acids (see Chapter 2, Table 2-4), the two most prevalent SFAs, are structural components of tooth enamel and dentin.
Monounsaturated Fatty Acids
When adjacent carbon atoms are joined by a double bond because two hydrogen atoms are lacking, there is a gap between the hydrogen atoms in the chain; it is called an unsaturated fatty acid. Monounsaturated fatty acids (MUFAs) contain only one double bond (see Chapter 2, Fig. 2-12). The most abundant MUFA is oleic acid. Oleic acid is also a structural component of the tooth.
Trans Fatty Acids
Hydrogenation is a commercial process in which vegetable oil is converted to a solid margarine or shortening by adding hydrogen to the oil. This process results in naturally unsaturated vegetable oils being changed to a SFA by changing unsaturated bonds to saturated bonds. Hydrogenation can be controlled, so “tub” or “soft” margarine is “partially hydrogenated,” or not completely saturated. The hydrogenation process not only increases the proportion of SFAs, but also changes the shape of the fatty acid. When the hydrogen atoms are rotated so that they are on opposite sides of the bond, in the “trans” position (see Chapter 2, Fig. 2-16), the fatty acid is called a trans fatty acid. Partial hydrogenation results in large numbers of fatty acids having this altered shape. Foods with trans fatty acids have a longer shelf life, and flavors are stable. The most common trans fatty acid is elaidic acid, found in partially hydrogenated vegetable oils, such as tub margarines and cooking oils. A naturally-occurring trans fatty acid, vaccenic acid, with double bonds on adjacent carbons, is present in small amounts in milk and meat of ruminants (cows, sheep, and deer). Limited research suggests that these trans fats may possibly have health-enhancing potential.
Polyunsaturated Fatty Acids
When carbons in a fatty acid are connected by two or more double bonds, the fatty acid is polyunsaturated (see Chapter 2, Fig. 2-13). Linoleic acid and arachidonic acid are polyunsaturated fatty acids (PUFAs). These PUFAs are omega-6 fatty acids. Their first double bond is on the sixth carbon from the omega (terminal) end; they are also referred to as n-6 PUFAs.
Omega-3 fatty acids, or α-linolenic acids, make up another class of PUFAs. As shown in Chapter 2, Figure 2-13A, these fatty acids are unique in that the first double bond is located three carbon atoms from the omega end of the molecule; hence they are called omega-3s or n-3s. Omega-3 fatty acids include α-linolenic acid, which has 18 carbon atoms and two double bonds, and eicosapentaenoic acid (EPA), which has 20 carbon atoms and five double bonds.
Compound Lipids
Phospholipids
Lipoproteins
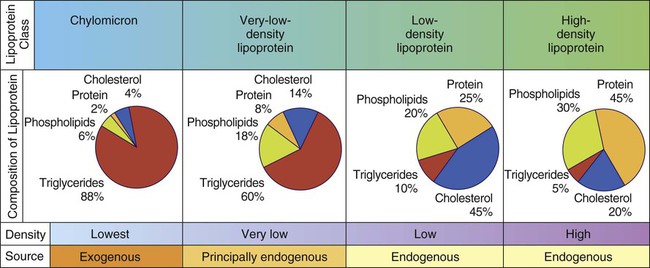
Lipoproteins are produced by the body to transport insoluble fats in the blood. Lipoproteins are compound lipids composed of triglycerides, phospholipids, and cholesterol combined with protein (see Chapter 2, Fig. 2-18). The liver and intestinal mucosa produce lipoproteins. Four different types of lipoproteins are present in the blood: high-density lipoproteins (HDLs), low-density lipoproteins (LDLs), very-low-density lipoproteins (VLDLs), and chylomicrons.
The ratio of lipid to protein in lipoproteins varies widely; these variations affect their density. Density increases as lipids decrease and protein increases. Lipoproteins can be classified according to their density and composition, as shown in Figure 6-4. Phospholipids in lipoproteins are present in approximately the same proportions in all individuals.
HDLs, which have been thought to protect against development of CHD, contain greater amounts of protein and less lipid. LDL cholesterol typically constitutes 60% to 70% of the total blood cholesterol. It is considered the main agent in elevated serum cholesterol levels, or the “bad” cholesterol. Serum HDL, LDL, and VLDL are important predictors of heart disease, as discussed in Health Application 6.
Cholesterol
Cholesterol is a fatlike, waxy substance classified as a sterol derivative with a complex ring structure (see Chapter 2, Fig. 2-17). More details on the structure and function of cholesterol are available online in the Evolve website. Because the body frequently produces more cholesterol than it absorbs, cholesterol intake is not essential. Cholesterol has important functions as a constituent of the brain, nervous tissue, and bile salts; a precursor of vitamin D and steroid hormones; and a structural component of cell membranes and teeth. Lipoproteins transport cholesterol in the blood.
Physiological Roles
Energy
Palatability
Fats contribute to the palatability and flavor of foods. In cooking, they improve texture. A receptor on the tongue and a potential pathway for detection of a “fatty taste” has been identified, which may affect food preferences.3,4 Preference for high-fat foods develops at an early age and persists through adulthood.
Complementary Relationships
Linoleic acid, an omega-6 fatty acid with 18 carbon atoms and two double bonds (see Chapter 2, Fig. 2-12), cannot be synthesized by the body and must be supplied from dietary sources. If linoleic acid is not furnished in the diet, signs of deficiency, including growth retardation, skin lesions, and reproductive failure, result. For this reason, linoleic acid is an essential fatty acid (EFA).
Arachidonic acid (18-carbon chain with four double bonds) and linolenic acid (18-carbon chain with three double bonds) are also considered EFAs, but healthy individuals can produce them from sufficient quantities of linoleic acid (see Chapter 2, Fig. 2-13). Linolenic acid can be converted rapidly into omega-3 fatty acids in the body. The conversion of linolenic acid to EPA and the conversion of linoleic acid to arachidonic acid are competitive because the processes use the same enzyme. Studies suggest that less than 10% of linolenic acid is converted to EPA. When intake of linoleic acid is substantially higher than intake of linolenic acid, less EPA is available. Linolenic acid may be a protective factor against CHD (Table 6-1).5
Table 6-1
Common food sources and physiological actions of fatty acids
| Fatty Acid Classification | Common Food Sources | Physiological Action |
| Saturated Fatty Acid (SFA) | ||
| Coconut oil, butter fat, most fats and oils, cocoa butter, fully hydrogenated vegetable oils | Raises total, LDL and HDL cholesterol (except stearic acid) | |
| Monounsaturated Fatty Acid (MUFA) | ||
| Cis configuration | Some fish oils, beef fat, most fats and oils, nuts, seeds, avocados | Decreases total and LDL cholesterol when substituted for SFAs and decreases total cholesterol compared with dietary carbohydrate |
| Trans configuration | Partially hydrogenated vegetable oils | Raises total and LDL cholesterol similar to SFAs, decreases HDL more than saturated fatty acids, and raises total-to-HDL ratio more than SFAs |
| Trans configuration | Dairy fat, meat from ruminating animals (beef, lamb) | May have beneficial health effects, especially vaccenic acid; more research needed |
| Polyunsaturated Fatty Acid (PUFA) | ||
| n-6 Fatty Acids | ||
| Linoleic acid | Liquid vegetable oils, nuts, seeds | Decreases total and LDL cholesterol |
| Arachidonic acid | Meat, poultry, fish, eggs | Precursor for important biologically active substances; substrate for synthesis of a variety of proinflammatory compounds |
| n-3 Fatty Acids | ||
| α-Linolenic acid | Flaxseed, canola oil, soybean oil, walnuts | Decreases cardiovascular risk |
| Eicosapentaenoic acid (EPA) Docosahexaenoic acid (DHA) |
Fish oil, algae | Decreases risk of sudden death from cardiovascular conditions and has beneficial effects on nervous system development and health |
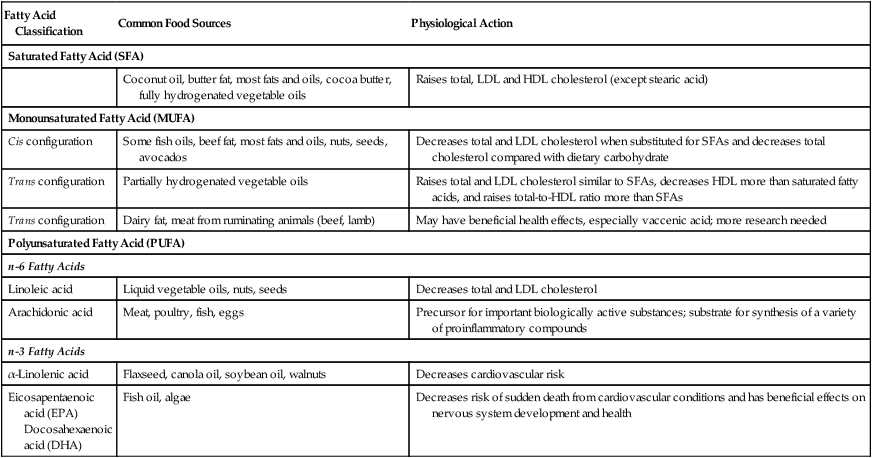
Adapted from Kris-Etherton P, Innis S: Position of the American Dietetic Association and Dietitians of Canada: dietary fatty acids. J Am Diet Assoc 2007; 107(9):1599-1611.
Omega-3 fatty acids are used to produce compounds regulating blood pressure, clotting, immune responses, gastrointestinal secretions, and cardiovascular functions; they also prevent heart arrhythmias and decrease triglyceride levels. Omega-3 fatty acids are essential for the development of brain and retinal tissues in fetal and neonatal development, and ongoing cognitive development in childhood. The presence of omega-3 fatty acids in the diet has been linked to reduction or amelioration of several chronic diseases, including CHD, atherosclerosis and atherosclerotic plaque, and mortality risk from CHD (Fig. 6-5),6 rheumatoid arthritis, psoriasis, inflammatory and immune disorders, and serious eye problems such as macular degeneration (see Table 6-1). However, in numerous studies, supplementation has not been associated with either lower risk of all-cause mortality or major CHD outcomes.7 Studies are exploring the relationship of omega-3 fatty acids with mental aging and Alzheimer disease.
Dietary Fats and Dental Health
1. Some fatty acids, specifically oleic acid, are growth factors for lactic acid bacteria, whereas streptococcal organisms are inhibited by lauric acid (Lauricidin).
2. Long-chain fatty acids may reduce dissolution of hydroxyapatite by acids.
3. Oral food retention is reduced by fat intake.
4. Fats may lubricate the tooth surface and prevent penetration of acid to the enamel (i.e., the “greased” tooth is impervious to acid, protecting caries-susceptible areas).
5. Fats may produce a film on food particles and prevent partial digestion of food particles in the mouth.
6. Dietary fat delays gastric emptying, enhancing fluoride absorption, and increasing tissue fluoride concentration.
Bacterial inflammation and systemic immune response are believed to play a central role in the initiation and propagation of atherosclerosis. Periodontal diseases (including gingivitis and periodontitis) are oral conditions caused by bacteria (and poor oral hygiene) that are risk factors contributing to coronary artery disease. When bacteria are allowed to grow rampantly in the mouth, inflammation may occur throughout the body. Bacteria from dental plaque biofilm can cause blood clots when they escape into the bloodstream and could be involved in inflammation of the lining of blood vessels and atherosclerosis. This inflammation may serve as a base for development of arterial atherosclerotic plaques, but—contrary to some concerns—omega-6 fatty acids do not seem to promote inflammation.8 Research studies show that the inflammatory process can be attenuated by n-3 fatty acids.9 Consumption of greater amounts of docosahexaenoic acid (DHA) and—to a lesser degree—EPA were associated with lower prevalence of periodontitis.10
Dietary Requirements
A certain amount of fat is needed to provide adequate amounts of fat-soluble vitamins and EFAs. The acceptable macronutrient distribution range for fat is estimated to be 20% to 35% of energy intake for adults (Table 6-2). The lower limit for fat intake was established to minimize the increase in blood triglyceride levels and decrease in HDL cholesterol levels that occur with higher intakes of carbohydrates. The upper limit of 35% kcal from fat was based on information indicating higher fat intake is associated with a greater intake of energy and SFA, which may be detrimental to health. Box 6-1 shows a method for calculating the IOM recommendation for dietary fat.
Table 6-2
Fat recommendations for adults*
| Classification of Fat | Dietary Guidelines | IOM† Reference Dietary Allowance (RDA)/Adequate Intake‡ (AI) | IOM† Acceptable Macronutrient Distribution Range (AMDR) | American Heart Association§ | American Diabetes Association¶ | Canada’s Food Guide** |
| Total fat | 20% to 35% kcal | — | 20% to 35% kcal | 25% to 35% kcal | — | 20% to 35% kcal |
| Saturated fatty acids (SFA) | <10% kcal | Minimize | Minimize | <7% kcal (stay away from tropical oils such as coconut oil, palm oil and palm kernel oils that are high in SFA) | <7% kcal | Limit butter, hard margarines, lard, and shortening |
| Trans fatty acids (TFA) | <1% kcal | Minimize¶¶ | Minimize¶¶ | >1% kcal | Minimize¶¶ | Limit hard margarines, lard, and shortening |
| Omega-6 fatty acids (n-6 PUFA) | 14 g/day for males; 11 g/day for females | 5% to 10% kcal | Choose monounsaturated fatty acids (MUFA) or PUFA (vegetable oils, margarines with liquid vegetable oil as the first listed ingredient) | Use vegetable oils, such as canola, olive, and soybean | ||
| α-Linolenic acid | 1.6 g/day for males; 1.1 g/day for females | 0.6% to 1.2% kcal | No specific recommendation |
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses