Antimicrobial photodynamic therapy (PDT) has attracted much attention for the treatment of pathogenic biofilm associated with peridontitis and peri-implantitis. However, data from randomized controlled clinical studies (RCTs) are limited and, to some extent, controversial, making it difficult to provide appropriate recommendations. Therefore, the aims of the present study were (a) to provide an overview on the current evidence from RCTs evaluating the potential clinical benefit for the additional use of PDT to subgingival mechanical debridement (ie, scaling and root planing) alone in nonsurgical periodontal therapy; and (b) to provide clinical recommendations for the use of PDT in periodontal practice.
Key points
- •
Antimicrobial photodynamic therapy (PDT) has lately attracted much attention among clinicians for the treatment of pathogenic biofilm associated with peridontitis and peri-implantitis.
- •
At present, the data from randomized controlled clinical studies (RCTs) are still limited and, to some extent, controversial, which makes it difficult to provide appropriate recommendations for the clinician.
- •
The aims of the present study were: (a) to provide an overview on the current evidence from RCTs evaluating the potential clinical benefit for the additional use of PDT to subgingival mechanical debridement (ie, scaling and root planing [SRP]) alone in nonsurgical periodontal therapy; and (b) to provide clinical recommendations for the use of PDT in periodontal practice.
- •
In patients with chronic periodontitis (ChP), the combination of SRP and PDT may result in substantially higher short-term clinical improvements evidenced by probing depth or bleeding on probing reductions compared with SRP alone.
- •
In patients with aggressive periodontitis, the use of PDT cannot replace the systemic administration of amoxicillin and metronidazole. Because of the lack of data, no conclusions can be made to what extent PDT may replace the use of systemic antibiotics in patients with ChP.
- •
Limited evidence from one study indicates that PDT may represent a possible alternative to local antibiotics in patients with incipient peri-implantitis.
Biological rationale
Periodontitis is a multifactorial disease that is associated with loss of the supporting tissues (ie, periodontal ligament and alveolar bone) around the tooth. A major objective of periodontal therapy is to remove soft and hard, supragingival and subgingival deposits from the root surface in order to stop disease progression. Numerous studies have reported significant improvements of clinical and microbial parameters following nonsurgical periodontal therapy.
Despite that nonsurgical periodontal treatment may result in significant clinical improvements in the great majority of cases, evidence indicates that none of the currently available instrumentation techniques are effective in completely eliminating subgingival bacterial biofilm. These limitations may be attributed to several factors, such as the complex anatomy of teeth (ie, furcation involvements, root invaginations); the presence of intrabony defects, and others; mechanical limitations related to the size of instruments, or invasion of periodontal pathogens into the surrounding soft tissues, or possible recolonization of periodontal pockets from other diseased sites or intraoral niches. Power-driven instruments (ie, sonic and ultrasonic scalers) have been introduced to further enhance the effectiveness of scaling and root planing (SRP). However, findings from clinical studies have also shown comparative outcomes following power-driven and manual instrumentation. Thus, the current evidence indicates that nonsurgical periodontal treatment may result in substantial clinical improvements in most cases, but none of the currently available instrumentation techniques are able to completely eliminate subgingival bacteria and calculus.
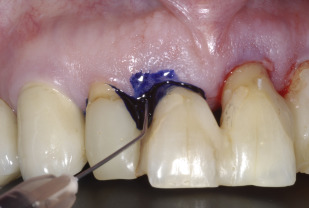
Photodynamic therapy (PDT), also called photoradiation therapy, phototherapy, photochemotherapy, photo-activated disinfection (PAD), or light-activated disinfection (LAD), was introduced in medical therapy in 1904 as the light-induced inactivation of cells, microorganisms, or molecules and involves the combination of visible light, usually through the use of a diode laser and a photosensitizer. The photosensitizer ( Fig. 1 ) is a substance that is capable of absorbing light of a specific wavelength and transforming it into useful energy. Each factor is harmless by itself, but when combined, can produce lethal cytotoxic agents that can selectively destroy cells. Thus, PDT has been proposed as a modality to reduce bacterial load or even to eliminate periodontal pathogens.