Introduction
This study was designed to test the hypothesis that thermoplastic retainers influence oral microbial flora during the retention period because they prevent the flushing effect of saliva on dental and mucous tissues.
Methods
Twenty-four orthodontic patients finished the study. After debonding, the patients were given thermoplastic retainers (Essix ACE 0.040-in plastic, Dentsply International, York, Pa) for both jaws and instructed to wear them all day. Plaque samples from tooth surfaces and saliva samples were collected from each patient just after debonding (T0), and on day 15 (T1), day 30 (T2), and day 60 (T3) of retention. The jaws were divided into 6 regions, and the data for each region were evaluated separately. Total viable Lactobacillus and Streptococcus mutans colonies were counted, and the numbers of the viable microorganisms were calculated.
Results
The numbers of Lactobacillus colonies at T3 were higher than at T0, T1, and T2, and the difference between T0 and T3 was statistically significant ( P <0.05). The numbers of S mutans colonies at T3 were higher than at T0, T1, and T2, and the differences between T0 and T1, and T1and T2 were statistically significant ( P <0.05).
Conclusions
Retention with thermoplastic retainers might create oral conditions conducive to S mutans and Lactobacillus colonization on dental surfaces.
Retention is an important phase of orthodontic treatment during which tooth movements after active treatment are stabilized. There are numerous types of retention methods for clinical use, and thermoplastic retainers are widely preferred for their advantages: removable, more esthetic, cost effective, and less time-consuming in the laboratory.
Identification of the changes in the oral cavity of patients who have orthodontic therapy, including the retention period, is important. Previous data about oral hygiene of orthodontic patients held removable and fixed orthodontic appliances responsible for worsening the oral hygiene and promoting the differentiation of microbial flora of the oral cavity that might result in caries, white spot lesions, and gingival inflammation. In studies concerning oral hygiene during or after orthodontic treatment, special attention is attributed to cariogenic populations such as Streptococcus mutans and Lactobacillus .
Many studies in the literature have evaluated the changes in S mutans and Lactobacillus levels during orthodontic therapy with either acrylic removable appliances or orthodontic bands and brackets. Besides, most of these studies concentrated on the active orthodontic treatment phase with fixed or removable appliances. However, microbiologic data during retention period are limited. Yet, as far as we know, there is no information about the effects of thermoplastic retainers on oral microbiota, although they are frequently preferred. In addition, there are no data evaluating the oral cavity by separating it into specific zones during retention with thermoplastic retainers. Therefore, the purpose of this study was to test the hypothesis whether thermoplastic retainers influence oral microbial flora during the retention period because they prevent the flushing effect of saliva on dental and mucous tissues.
Material and methods
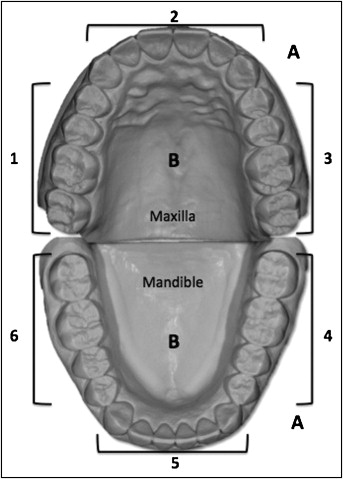
The initial study population comprised 40 subjects with an age range of 14 to 20 years, who had fixed orthodontic treatment at the Department of Orthodontics of Gazi University in Ankara, Turkey, and had started to wear thermoplastic retainers in the retention period. Exclusion criteria included caries-active patients, the use of oral antimicrobial agents or antibiotics within the past 3 months, smoking habit, periodontal or systemic disease, and prosthodontic appliances. Unfortunately, 16 subjects did not attend their periodic visits, and the study population was narrowed to 24 subjects. All patients included in the study signed informed consent forms. Thermoplastic retainers (Essix ACE 0.040-in plastic; Dentsply International, York, Pa) covering all tooth surfaces up to 2 mm above the gingival margin and the occlusal surfaces were prepared for both jaws. The patients were instructed to wear their retainers all day and brush them after toothbrushing. The subjects had refrained from eating or drinking beverages for at least 2 hours before sample collection and had been told not to brush their teeth before the examinations. Plaque samples from tooth surfaces and saliva samples were collected from each patient just after debonding (T0), and at day 15 (T1), day 30 (T2), and day 60 (T3) of retention with the thermoplastic retainers. First, unstimulated saliva samples were collected from the patients. They rinsed their mouth out well with drinking water and were advised to sit relaxed for 5 minutes. After this procedure, they were asked to spit about 5 mL of saliva into 50-mL sterile tubes. The jaws were divided into 6 regions and named as shown in the Figure . Plaque samples were collected with sterile swabs from the gingival margin and enamel surface of each tooth at the vestibule and the palatal-lingual sides, and the data for each region were evaluated separately. The entire surfaces of all teeth and 1 to 2 mm of gingiva in each region were swabbed. After scraping the dental plaque, the samples were put into presterilized test tubes containing 1 ml of trypticase soy broth (Merck, Darmstadt, Germany) separately and vortexed for 60 seconds. Then, the suspensions were diluted to 10 −1 , 10 −2 , and 10 −3 serially, and 100-μL amounts of each primer suspension and the diluted suspensions were put onto de Man, Rogosa and Sharpe (MRS) agar for Lactobacillus selective agar (Merck) and tryptone-yeast extract-cysteine-sucrose-bacitracin agar for the isolation of S mutans. The plates were incubated in an incubator at 37°C for 3 to 4 days in microaerophilic conditions (air + 5% carbon dioxide). After collecting the unstimulated saliva, 1 mL of saliva was put in another sterilized tube and diluted from 10 −1 to 10 −3 serially with sterilized distilled water. After vortexing the tubes for 15 seconds, 100 μL of each diluted saliva was put onto the agar media separately and incubated as mentioned before. After these processes, total viable S mutans and Lactobacillus colonies were counted, and the numbers of the viable microorganisms were calculated by means of colony-forming units per milliliter of volume (cfu/ml). The data and the statistics were done according to the 1 mL amount of volume as colony-forming units per milliliter. Homogeneities of the group variances were analyzed with the Levene test; because they were normally distributed, significance was determined with 1-way analysis of variance (ANOVA).
Results
Means of the sums of all regions were evaluated. The mean amount of Lactobacillus at T3 (14.49 cfu/mL) was higher than at T0, T1, and T2, and the difference between T0 and T3 was statistically significant ( P <0.05) ( Table I ). The mean amount of S mutans at T1 (43.72 cfu/mL) was higher than at T0, T2, and T3, and the differences between T0 and T1, and T1 and T2 were statistically significant ( P <0.05) ( Table I ).
S mutans and Lactobacillus counts in the saliva showed no significant differences among T0, T1, T2, and T3 ( Table II ).
| Mean (cfu/mL) | SD | P | |
|---|---|---|---|
| Lactobacillus | |||
| T0 | 10.42 | 24.68 | 0.371 |
| T1 | 37.54 | 131.83 | |
| T2 | 4.57 | 8.7 | |
| T3 | 14.25 | 22.88 | |
| S mutans | |||
| T0 | 46.67 | 51.2 | 0.811 |
| T1 | 70.21 | 135.41 | |
| T2 | 53.21 | 74.14 | |
| T3 | 51.04 | 73.71 |
Overall, the mean amount of Lactobacillus in the mandibular right vestibular region was the highest at all times, whereas it was the lowest in the maxillary anterior region and the mandibular anterior region ( Table III ). On the other hand, the highest mean amount of S mutans was found in the maxillary left and mandibular palatal-lingual regions, but it was the lowest at the maxillary anterior labial region and the mandibular anterior region at all times ( Table IV ). Nevertheless, the differences in the numbers of both microbial agents were not statistically different among the regions where the samples were taken at T0, T1, T2, and T3 ( P >0.05) ( Tables III and IV ).
| 1A | 1B | 2A | 2B | 3A | 3B | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | |
| T0 | 4.96 | 6.94 | 4.17 | 7.86 | 2.71 | 5.55 | 2.00 | 5.02 | 15.29 | 60.90 | 2.75 | 4.87 |
| T1 | 3.58 | 6.39 | 8.83 | 21.89 | 1.21 | 3.18 | 1.54 | 2.73 | 8.42 | 16.70 | 7.54 | 12.03 |
| T2 | 12.17 | 27.87 | 11.17 | 24.79 | 7.58 | 18.47 | 5.04 | 11.59 | 12.25 | 20.66 | 10.33 | 19.5 |
| T3 | 20.63 | 47.12 | 19.48 | 46.8 | 7.75 | 15.91 | 8.33 | 18.42 | 13.29 | 27.77 | 12.21 | 29.31 |
| 4A | 4B | 5A | 5B | 6A | 6B | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | |
| T0 | 7.83 | 13.63 | 2.71 | 4.96 | 1.21 | 4.35 | 0.63 | 2.08 | 17.29 | 60.61 | 3.75 | 5.68 |
| T1 | 10.17 | 23.97 | 9.63 | 24.83 | 3.08 | 7.55 | 2.96 | 7.05 | 7.33 | 11.84 | 8.00 | 15.80 |
| T2 | 19.96 | 36.05 | 15.75 | 28.59 | 6.83 | 17.07 | 3.88 | 9.04 | 16.71 | 39.72 | 11.17 | 21.93 |
| T3 | 17.38 | 35.89 | 18.75 | 43.80 | 6.63 | 20.97 | 7.92 | 23.00 | 22.92 | 43.80 | 19.00 | 38.96 |
| 1A | 1B | 2A | 2B | 3A | 3B | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | |
| T0 | 57.21 | 202.27 | 28.67 | 35.65 | 19.54 | 27.25 | 25.21 | 26.16 | 25.04 | 36.40 | 33.25 | 37.89 |
| T1 | 34.79 | 64.63 | 42.83 | 82.10 | 28.96 | 67.34 | 60.17 | 155.99 | 45.92 | 83.31 | 53.67 | 96.42 |
| T2 | 29.92 | 44.06 | 32.58 | 42.05 | 26.04 | 35.54 | 28.29 | 35.37 | 29.63 | 35.19 | 41.17 | 43.76 |
| T3 | 43.04 | 68.85 | 51.08 | 88.26 | 26.00 | 39.84 | 41.96 | 91.31 | 43.71 | 57.51 | 55.33 | 93.18 |
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


