Introduction
The aim of this study was to monitor the alveolar bone formation at the tension sites of teeth supporting the appliances for rapid maxillary expansion (RME) during the retention phase according to the local gingival crevicular fluid (GCF) alkaline phosphatase (ALP) activity.
Methods
This split-mouth prospective study included 23 prepubertal subjects (15 girls, 8 boys; mean age, 9.0 ± 1.4 years) who had a constricted maxillary arch and were undergoing RME. Periodontal parameters, including probing depth, were recorded at 3 and 6 months after RME. Furthermore, the GCF ALP activity was measured at the tension sites of the supporting test teeth (TT) and at the antagonist control teeth (CT) sites.
Results
Periodontal parameters were generally similar between the TT and CT sites during the study, with the exception that probing depth underwent a slight increase at the TT sites. At baseline, the GCF ALP activity was similar between the TT and CT sites; however, at both 3 and 6 months, significantly greater enzymatic activity was seen at the TT sites. The overall probing depth changes were not significantly correlated with the corresponding GCF ALP activity changes for either the TT or the CT sites.
Conclusions
Alveolar bone formation at the tension sites would last up to 6 months of retention after RME. These results warrant more comprehensive studies to assess whether the GCF ALP activity has potential as a diagnostic tool for bone formation during the retention phase of RME.
Highlights
- •
We examined bone remodeling at tension sites of first molars receiving rapid maxillary expansion.
- •
The local gingival crevicular fluid (GCF) alkaline phosphatase (ALP) activity was evaluated at baseline and up to 6 months of retention.
- •
Antagonist control teeth were also considered.
- •
The GCF ALP activity increased in the tension sites during the whole term.
- •
Retention after rapid maxillary expansion should not be less than 6 months.
When a skeletal constriction of the maxillary arch is diagnosed, orthopedic skeletal expansion involving separation of the midpalatal suture is the treatment of choice. The most common treatment is rapid maxillary expansion (RME). It has been reported that mainly skeletal effects of RME are seen if this treatment is performed before puberty, whereas at later development stages, more dental effects are to be expected, with possible tissue damage. However, even when treatment is performed during the optimal skeletal maturation phase—ie, prepubertal—some dentoalveolar effects appear to occur. Moreover, there is always some degree of skeletal or dentoalveolar relapse after RME, thus requiring hypercorrection followed by 4 to 5 months of retention. Although most previous studies on RME treatment focused on the effects on the midpalatal suture or other sagittal and vertical changes regarding dentoalveolar effects, a decrease in the buccal bone plate thickness of the supporting teeth has been reported at the end of active RME followed by a significant recovery of the original thickness after a retention period of 6 months.
Gingival crevicular fluid (GCF) is a transudate with constituents from a variety of sources, including microbial dental plaque, host tissues, and serum, with a high site specificity. A number of GCF constituents including host enzymes have been proposed as diagnostic indicators of periodontal status. Among these enzymes, one of the first to be identified was alkaline phosphatase (ALP). GCF ALP has a primary role in bone mineralization, and it has been shown to be sensitive to alveolar bone formation during orthodontic tooth movement. To date, only a few studies have investigated the levels of GCF constituents during maxillary expansion. In particular, the levels of the inflammatory mediators interleukin-1β and β-glucuronidase and prostaglandin E2 in GCF retrieved from the maxillary teeth of adolescents undergoing RME were increased. Interestingly, a few investigations on the GCF ALP activity changes during or after RME treatment have been reported, in spite of the primary role of this enzyme in alveolar bone formation. Moreover, none of these studies included prepubertal subjects.
Because of the small amount of data available on the metabolic changes at the palatal (tension) alveolar sites during the retention phase after RME treatment, the aim of this split-mouth prospective study in prepubertal subjects was to monitor alveolar bone formation at the tension sites of the first molars undergoing RME treatment. GCF ALP activity was used as a biomarker of tissue remodeling to determine the existence and duration of active alveolar bone formation during the retention phase.
Material and methods
The study sample consisted of 23 healthy white children (15 girls, 8 boys; mean age, 9.0 ± 1.4 years; range, 6.7-11.9 years) who sought orthodontic treatment at the unit of orthodontics of the School of Dentistry at the Second University of Naples in Italy. The experimental protocol was approved by the institutional ethical committees, and voluntary informed consent was obtained from the patients after they received detailed information about the clinical trial.
Subjects were included according to the following criteria: (1) constricted maxillary arch with a unilateral or bilateral posterior crossbite; (2) prepubertal stage assessed by the cervical vertebral maturation method (stage 1 or 2), (3) good general health, (4) variable degree of crowding, and (5) no use of anti-inflammatory drugs in the month preceding the beginning of the study. Further periodontal inclusion criteria were (1) probing depth (PD) values not exceeding 3 mm in the whole dentition, (2) no radiographic evidence of periodontal bone loss evaluated by panoramic x-ray examination, and (3) full-mouth plaque and full-mouth bleeding scores of 20% or less. The full-mouth plaque and full-mouth bleeding scores were recorded as the percentages of tooth surfaces with supragingival plaque or bleeding within 15 seconds after probing with a 20-g controlled force probe (Vivacare TPS Probe; Vivadent, Schaan, Lichtenstein). Exclusion criteria were (1) absence of maxillary first molars; (2) previous or current periodontal disease, or attachment loss in the posterior teeth; and (3) previous orthodontic treatment.
The study design is shown in Figure 1 . A preliminary visit took place 1 month before the baseline visit when the RME treatment began. At this preliminary visit, after the periodontal examination, full-mouth supragingival and subgingival scalings were carried out, and oral hygiene instructions were given to all patients. Moreover, in the month preceding the study and throughout it, all subjects received repeated oral hygiene instructions about the correct use of the toothbrush, dental floss, and interdental brush, and were not allowed to take any anti-inflammatory drugs that could have affected the results. Moreover, they rinsed twice with 0.012% chlorhexidine mouthwash during the 2 weeks before the baseline visit and before the times when the GCF was sampled.
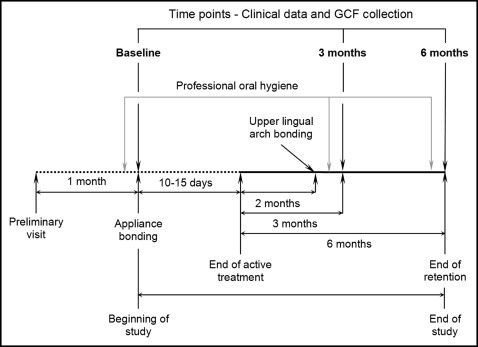
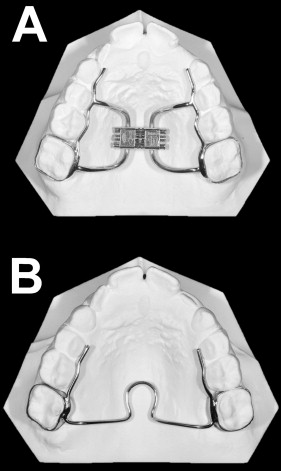
At the baseline visit, the periodontal parameters were recorded, and the GCF was sampled immediately before the beginning of treatment, which consisted of mounting a hyrax rapid maxillary expander ( Fig 2 , A ) cemented on the maxillary permanent first molars. The appliance was constructed with plain bands with a lowered vertical dimension such that they did not reach, or minimally entered, the crevicular sulcus. Laser welding was used, and the wires were modeled so that they were not in direct contact with the palatal gingival margins. An expansion screw with reduced dimension (model AO-0630-10; Leone, Sesto Fiorentino, Italy) was used and activated at 2 turns per day (0.20 mm per turn) for 16 to 23 days, thus reaching the total amount of expansion of 6.4 to 9.2 mm in all subjects (mean expansion, 7.5 mm). According to an in-vitro study, the force exerted by the screw used here, with arms orthogonal to the screw guide, is generally about 16 N per turn. Then the screw was tied off with a ligature wire, and the expander was kept on the teeth as a passive retainer for 2 months. Immediately after the removal of the maxillary expander, a passive palatal bar ( Fig 2 , B ), also constructed on plain bands with a reduced vertical dimension, was cemented on the maxillary permanent first molars. The palatal bar had short mesial arms on both sides and was kept for a further 4 months as a retainer.
The same clinical recordings and GCF samplings were repeated at 3 and 6 months after the RME active phase. The maxillary left and right permanent first molars were considered the test teeth (TT), and the mandibular left and right permanent first molars were considered the control teeth (CT).
For all TT and CT, the periodontal parameters included the presence of supragingival plaque (PL+), bleeding on probing (BOP+) 15 seconds after probing with a 20-g controlled force probe, and PD. Clinical data were always collected at 6 sites per tooth (mesiobuccal, midbuccal, and distobuccal; and mesiopalatal or mesiolingual, midpalatal or midlingual, and distopalatal or distolingual) by the same operator (F.D’A.).
Contamination of the GCF samples was minimized by recording the plaque presence before carefully cleaning the tooth with cotton pellets, collecting the GCF from the isolated area, and then recording the PL+ and BOP+ as previously described. GCF was collected using number 30 standardized sterile paper strips inserted 1 mm into the gingival crevice and left in situ for 30 seconds, after the sites had been isolated with cotton rolls and dried by a gentle airstream. The GCF was collected at the mesiopalatal and distopalatal sites of the TT and at the mesiolinual and distolingual sites of the CT. The GCF samples were transferred to plastic vials and stored at −80°C until they were analyzed.
For the enzymatic activity determination, the biochemical assays were performed by blinded operators (J.P., K.R.) as previously described. Briefly, the 4 samples from the 4 collection sites, for each resting and flow GCF sample, were resuspended in 250 μL of buffer containing 200 mmol/L of Tris and 20 mmol/L of magnesium chloride (pH, 9.8 ± 0.1) and 1 mg per milliliter of p-nitrophenol phosphate (N2770-5SET, Sigma Fast; Sigma-Aldrich, St Louis, Mo). The samples were then incubated at 37°C (± <0.1°C fluctuations) for 3 hours. During this incubation, the ALP in the samples hydrolyzed the p-nitrophenyl phosphate to p-nitrophenol and inorganic phosphate. The reactions were then stopped by the addition of 5 μL of 3-mol/L of sodium hydroxide, and the absorbance was read with a spectrophotometer at 405 nm. The relevant control for each analysis consisted of the reagent and the Tris buffer without the sample, and all samples were analyzed in 1 session. By using 18.45 as the p-nitrophenol mmol/L absorptivity, the absorbance was converted into enzyme activity units (1 unit = 1 mmol of p-nitrophenol released per minute at 37°C) and expressed as total activity in milliunits per sample.
A sample size of 24 subjects was necessary to detect an effect size coefficient of 0.8 for the GCF ALP activity in the comparison between sites (or time points), with an alpha set at 0.05 and a power of 0.80. The effect size coefficient is the ratio of the difference between the recordings of the 2 sites, divided by the within-site standard deviation. Even though the effect size coefficient is not a measure of diagnostic accuracy, a greater intergroup difference compared with the corresponding variability means greater expected diagnostic accuracy of the GCF ALP activity in detecting bone formation. Therefore, an effect size of at least 0.8, regarded as a “large effect,” was chosen. During the study, 1 subject dropped out, leaving a sample size of 23 subjects.
On a separate group of 10 untreated matched subjects with the same features as the experimental group, GCF samples were collected twice 1 week apart with similar modalities on the maxillary and mandibular first molars. These data were used for the calculation of the method error in the GCF collection and ALP activity quantification through the method of moments variance estimator. The results were expressed as percentages of mean error and 95% confidence intervals (CIs). The method of moments variance estimator has the advantage of not being affected by any unknown bias: eg, systematic errors between pairs of measurements.
Statistical analysis
The Statistical Package for Social Sciences software (version 13.0; SPSS, Chicago, Ill) was used for data analysis, and the patient was the statistical unit. Within each subject and each group, PL+ and BOP+ were considered as the mean numbers of experimental sites positive for supragingival plaque and bleeding on probing; PD and GCF ALP activity were similarly considered as the mean values obtained by the TT or CT in the same palatal or lingual sites for each subject. These mean values were the statistical units of each data set considered in the analysis to reduce intersubject variability.
Each continuous data set was tested for normality of the data with the Shapiro-Wilk test and by Q-Q normality plots. Equality of variance was also tested with the Levene test and Q-Q normality plots of the residuals. Each data set was treated as ordinal data by nonparametric tests because of the failure to meet the required assumption for parametric analyses. Nevertheless, the means and standard deviations for the clinical parameters are reported for descriptive purposes.
For each periodontal parameter and GCF ALP activity, the Wilcoxon paired signed rank test was used to assess the significance of the differences between the TT and CT sites at each time point. The significance of the differences over time in the TT and CT sites was assessed using the Friedman test followed by a Bonferroni-corrected Wilcoxon paired signed rank test, where appropriate.
For either the TT or CT, the overall changes in PD and in GCF ALP activity (considering the subject as the statistical unit) were calculated as the difference between the scores recorded at 6 months and the corresponding baseline values. Finally, the Spearman rank correlation coefficient was used to evaluate the significance of the correlation between the overall changes in PD and GCF ALP activity in the TT and CT sites.
A P value less than 0.05 was used for rejection of the null hypothesis.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


