Introduction
The release of metal ions from fixed orthodontic appliances is a source of concern. The aim of this study was to evaluate genotoxic damage in the oral mucosal cells of patients wearing fixed appliance, and the nickel and chromium ion contents in these cells.
Methods
Twenty patients undergoing orthodontic treatment formed the experimental group, and 20 untreated subjects comprised the control group. Oral mucosal smears were collected at 2 times: at debonding and 30 days after debonding. The smears were stained with Papanicolaou stain and studied under a light microscope to evaluate the presence of micronuclei. Inductively coupled plasma-mass spectrometry was used to quantify the presence of metal ions. The data were subjected to the Mann-Whitney U test and the Spearman rank correlation test.
Results
The mean micronuclei frequency was significantly higher in the treated group than in the control group at debonding; the difference was smaller and not statistically significant 30 days after debonding. The nickel and chromium ion contents in the experimental group were not significantly higher than in the control group. No correlation could be established between micronuclei frequency and metal ion content.
Conclusions
Nickel and chromium alloys of orthodontic appliances emit metal ions in sufficient quantities to induce localized genotoxic effects, but these changes revert on removal of the appliances.
Fixed orthodontic appliances consisting of bands, brackets, and wires are commonly made of austenitic stainless steel (SS) containing 18% chromium and 8% nickel. In the oral cavity, many factors work together to create an environment that makes aqueous corrosion in metals and alloys more favorable. The discharge of nickel ions, a strong immunologic sensitizer, can result in hypersensitivity, contact dermatitis, asthma, and cytotoxicity. In addition to the allergic issue, cellular and genetic toxicity has been assigned to nickel and, to a lesser extent, chromium.
Various studies have evaluated the discharge of metal ions from orthodontic appliances in biologic fluids, and most have concluded that they do not reach toxic concentrations. However, it cannot be excluded that even nontoxic concentrations might be sufficient to produce biologic changes in the oral mucosa. In addition to the cytotoxic effects of metal ions, the physical and mechanical effects of orthodontic appliances could also induce changes in the oral mucosa.
The oral mucosa is covered by a stratified epithelium composed of multiple layers of cells that show various patterns of differentiation (or maturation) between the deepest cell layer and the surface. The progenitor cells that cause cell division are situated in the basal layer. The turnover time of the epithelium is the time that it takes for a cell to divide and pass through the entire epithelium. Published data for turnover times for human buccal epithelium have a median value of 14 days. The epithelial stem cells play an important role in preserving the genetic information of the tissues, since the DNA is most vulnerable to damage during mitosis.
The genomic damage is produced by environmental exposure to genotoxins, medical procedures (eg, radiation and chemicals), micronutrient deficiency (eg, folate), lifestyle factors (eg, alcohol, smoking, drugs, and stress), and genetic factors such as inherited defects in DNA metabolism or repair. So, it is essential to have a reliable and minimally invasive biomarker to improve the implementation of biomonitoring, diagnostics, and treatment of diseases caused by, or associated with, genetic damage.
A genotoxicity risk evaluation in the buccal epithelial cells can be performed with some well-established endpoints such as the micronucleus (MN) assay. Micronuclei are extranuclear cytoplasmic bodies. The damaged chromosomes, in the form of acentric chromatids or chromosome fragments, lag behind in anaphase when centric elements move toward the spindle poles. After telophase, the undamaged chromosomes and the centric fragments give rise to regular daughter nuclei. The lagging elements are included in the daughter nuclei cells too, but a considerable portion is transformed into 1 nucleus or several secondary nuclei that appear in the cytoplasm of the daughter cells as a small nuclear particle, termed an MN.
Several authors in the last decade have focused on the appropriateness of using the MN assay in several clinical conditions as a noninvasive method for biomonitoring : eg, as a DNA source to study oxidative stress and the associations of micronuclei with cancer, and in Alzheimer’s disease. It has also been used to evaluate the effects of genotoxic agents such as tobacco smoke, alcohol, pesticides, and formaldehyde.
Genotoxic studies in response to fixed appliances are a cause for concern, and a few studies have evaluated changes in response to orthodontic appliances with the comet assay and the MN assay, and have found evidence of DNA damage in oral mucosa cells. Genotoxicity can be a mutagenic or a carcinogenic process. Yet, no study has reported an association of these conditions with fixed appliances. This could be because normal cells can repair these lesions. But a loss of repair capacity from a reduction in damage detection or an enzymatic deficiency in repair processes might be the initiating event of adverse biologic effects. However, to the best of our knowledge, no study has evaluated the reversal of these changes when orthodontic appliances are removed.
Thus, the aims and objectives of this study were to evaluate (1) the possible genotoxic damage to the oral mucosal cells in healthy patients undergoing orthodontic treatment with fixed appliance, compared with a control group, by using the MN assay; (2) the persistence of these genotoxic changes at 1 month after debonding the appliance; and (3) the metal (nickel and chromium) ion contents in the oral mucosal cells of a control group and an experimental group at two time intervals and to establish a correlation between this and the genotoxic changes.
Material and methods
Forty subjects between the ages of 14 and 24 years were enrolled in this study for 30 days after we obtained consent. They were divided into 2 groups of 20 each. Group 1 was the control group (subjects without fixed orthodontic appliances who had never been treated orthodontically), and group 2 was the experimental group (patients who had been treated with fixed appliances for a minimum of 18 months).
Inclusion criteria for the control and experimental subjects were (1) permanent dentition, (2) no amalgam fillings and metal restorations that could cause corrosion in the mouth, (3) no previous orthodontic treatment, and (4) no palatal or lingual appliances welded to the bands (ie, rapid maxillary expansion appliance) or extraoral auxiliary orthodontic appliances (ie, headgear).
Exclusion criteria for the control and experimental subjects were (1) syndromes; (2) mutagenic hazards unconnected with occupation (eg, smoking, drinking, drug consumption, and illness related to any genetic damage); (3) diabetes, anemia, or any debilitating disease and those under treatment with antibiotics or steroids during the study period; (4) use of alcohol-based mouthwashes; and (5) prostheses or tooth restorations with sharp edges or any lesions on the buccal mucosa.
Orthodontic appliances made of SS (0.07% carbon, 0.70% manganese, 1% silicon, 1%-17.5% chromium, 3.0%-5.0% nickel, 3.0%-5.0% copper, 0.04% phosphorus, 0.04% sulfur, and 0.15%-0.45% tantalum and niobium) consisting of 16 bonded brackets and 4 bands with buccal tubes were used in both arches. The SS materials were American Iron and Steel Institute type 304 for the bands and type 316 for the brackets (Ormco, Glendora, Calif). The archwires used in this study were nickel-titanium alloy (NiTi) and SS (both, 3M Unitek, Monrovia, Calif). The sequence of archwires was 0.016-in NiTi, 0.016 × 0.022-in NiTi, 0.016 × 0.022-in SS, 0.017 × 0.025-in SS, and 0.019 × 0.025-in SS.
After debonding, the patients in the experimental group were given retainers in both arches with fiber-reinforced composite during the study period.
We sampled the oral mucosal cells as follows. For the experimental group, samples were collected at 2 times: T0, at debonding of the fixed orthodontic appliance; and T1, 30 days after the debonding the fixed orthodontic appliance. For the control group, the samples were also collected similarly at 0 and 30 days.
The oral mucosal cells were collected from each subject by gentle scraping of the inside part of the lips and buccal mucosa with a metal spatula in a sweeping motion, after rinsing the mouth several times with tepid distilled water, to remove exfoliated dead cells.
The sample obtained was immediately smeared onto the center of a clean glass slide. Four smears were prepared for each subject for cytomorphometric analysis.
The smears were immediately fixed in absolute alcohol (isopropyl alcohol, 70%). Then the slides were hydrated with distilled water and stained with the Papanicolaou (PAP) method according to the standard protocol.
The oral smears were observed at 40-times magnification under a light microscope (CH20 i; Olympus, Ltd., New Delhi, India) by the same operator. One thousand cells from each subject were examined to determine the presence of micronuclei.
In the PAP method, the nuclear material appears blue, and the cytoplasm appears pink ( Figs 1 and 2 ).
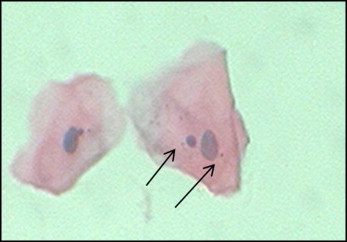
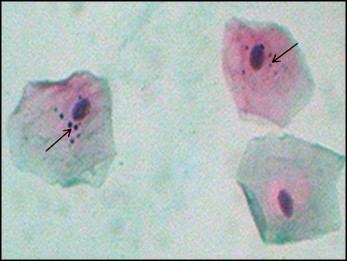
Cells that were not smeared, clumped, or overlapped, and those that contained intact nuclei were included. Cells undergoing degenerative processes such as karyorrhexis, karyolysis, fragmentation of the nucleus, broken egg, and pyknosis were excluded. The micronuclei were identified according to the standard protocol ( Figs 1 and 2 ).
The oral mucosal cells were sampled by using a cytologic brush (PAP smear test kit, Larsons, Delhi, India) and agitated (to loosen the cells that had adhered to the bristles) in 5 mL of distilled water in a 15-mL conical centrifuge tube.
For the digestion procedure for oral mucosal cells, 5 mL of the sample was treated with 1 drop of pure nitric acid (65% Supra pure nitric acid; Merck grade; Merck, Darmstadt, Germany) and heated in a water bath for about half an hour at 80°C. The sample was cooled at room temperature, and the final volume was measured for the estimation of nickel and chromium; 2 mL of the sample volume was required for the analysis.
The amounts of nickel and chromium in the cells were quantitatively assessed by using inductively coupled plasma-mass spectrometry (X series II, 187 LSD ES ICP-1, Thermoelectron X series 2 ICP-MS; Thermo Fisher Scientific, Waltham, Mass). The instrument was calibrated with standards in the range of 1 to 40 ppb. A 6-point calibration was used for the analysis. The standard solution was prepared with 2% nitric acid (National Institute of Standards and Technology, Gaithersburg, Md) traceable stock standard available in 100 ppm, and each calibration standard was made up to 100 mL. The calibration range was 1, 2, 5, 10, 20, and 40 ppb, and the samples were aspirated to the instrument.
Analyte masses 52 for chromium and 60 for nickel were selected. The calibration graph was verified, and the standard deviation and percentage of relative standard deviation were observed.
Statistical analysis
The mean and standard deviation were estimated in each study group (experimental and control). The normality of the data was tested in each group by using the Kolmogorov-Smirnov test.
The mean values were compared between the 2 groups by using the Mann-Whitney U test. The Wilcoxon signed rank test was used to compare the mean values between T0 and T1 in each group. The Spearman rank correlation coefficient was used to correlate the relationship between the 2 variables of both groups at T0 and T1. P <0.05 was considered to be the level of significance.
Results
At T0, the mean MN frequency in the experimental group was significantly higher than in the control group. At T1, the mean MN frequency in the experimental group was higher than in the control group, but this was not statistically significant ( P = 0.24) ( Table I ). The mean change in MN frequency between T0 and T1 was significant in the experimental group ( P <0.0001) but not in the control group ( P = 0.85).
| Time point | Experimental group (n = 20) | Control group (n = 20) | |
|---|---|---|---|
| Mean ± SD | Mean ± SD | P value | |
| T0 | 259 ± 233 | 53 ± 51 | <0.0001 (significant) |
| T1 | 48 ± 49 | 32 ± 35 | 0.24 (not significant) |
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


