The primary goal of regenerative endodontics is to restore the vitality and functions of the dentin-pulp complex, as opposed to filing of the root canal with bioinert materials. A myriad of growth factors regulates multiple cellular functions including migration, proliferation, differentiation, and apoptosis of several cell types intimately involved in dentin-pulp regeneration. Recent work showing that growth factor delivery, without cell transplantation, can yield pulp-dentin–like tissues in vivo provides one of the tangible pathways for regenerative endodontics. This review synthesizes knowledge on many growth factors that are known or anticipated to be efficacious in dental pulp–dentin regeneration.
- •
The goal of regenerative endodontics is to regain the vitality and functions of dental pulp–dentin complex. Dental pulp is the only vascularized tissue in mature, functional teeth in humans, and maintains homeostasis of the dentin.
- •
Current root canal therapy ends up with a devitalized tooth, therefore predisposing endodontically treated teeth to reinfections and fractures. Recent work showing regeneration of dental pulp-dentin–like tissues by cell homing that is orchestrated by growth factor delivery, without cell transplantation, provides one of the tangible pathways toward clinical translation.
- •
Growth factors regulate either transplanted cells or endogenously homed cells in dental pulp–dentin regeneration. Further understanding of the actions of growth factors is pivotal for dental pulp–dentin regeneration.
Introduction
Regenerative endodontics aims to restore the vitality and functions of the pulp–dentin complex that has been lost to trauma or infections ( Table 1 ). Several recent reports have shown that dental pulp–like tissues can regenerate in vivo following the delivery of dental or nondental stem/progenitor cells. An alternative approach is to orchestrate dental pulp–dentin regeneration by the homing of host endogenous cells relies on growth factor delivery, instead of cell delivery. Regardless of cell transplantation or cell homing approaches in dental pulp regeneration, a multitude of growth factors has been shown to have effects on dental pulp cells.
| Signaling Molecules | Target Cells | Primary Effects | Interactions |
|---|---|---|---|
| Growth Factors | |||
| PDGF | Dental pulp cells | Cell proliferation | Combined with IGF-1 or dexamethasone, increased cell proliferation |
| Dentin matrix synthesis | Combined with PDGF, increased cell proliferation | ||
| Odontoblastic differentiation | |||
| Dentinogenesis | |||
| TGFβ1 | Dental pulp cells | Cell proliferation | |
| Extracellular matrix synthesis Odontoblastic differentiation |
|||
| Dentinogenesis | |||
| Dental pulp stem cells | Chemotaxis | Combined with FGF2, increased odontoblastic differentiation | |
| BMP2 | Dental pulp cells | Odontoblastic differentiation | |
| Dentinogenesis | |||
| BMP4 | Dental pulp cells | Odontoblastic differentiation | |
| Dentinogenesis | |||
| BMP7 (OP-1) | Dental pulp cells | Dentinogenesis | |
| BMP11 (GDF11) | Dental pulp stem cells | Odontoblastic differentiation | |
| Dentinogenesis | |||
| VEGF | Dental pulp stem cells | Odontoblastic differentiation | Under osteogenic conditions, increased osteogenic differentiation |
| Cell proliferation | |||
| FGF2 (bFGF) | Dental pulp stem cells | Chemotaxis | Combined with TGFβ1, increased odontoblastic differentiation |
| Cell proliferation | |||
| Dental pulp cells | Cell proliferation | ||
| Dentinogenesis | |||
| IGF | Dental pulp cells | Cell proliferation | Combined with PDGF, increased cell proliferation |
| Odontoblastic differentiation | |||
| NGF | Dental papilla cells | Odontoblastic differentiation | |
| Cytokine | |||
| SDF-1 | Dental pulp cells (CD31 – /CD146 – SP cells) | Chemotaxis | |
| Cell proliferation | |||
The dental pulp is a unique, specialized loose connective tissue that contains mainly interstitial fibroblasts in a cell-rich zone in the center of the pulp and odontoblasts that align dentin surface in the periphery (the odontoblast layer). Stem/progenitor cells reside among interstitial fibroblasts and, perhaps, adjacent to blood vessels. When dental pulp cells are isolated and studied in vitro, some of the mononucleated and adherent cells, but certainly not all and not even the majority, have stem/progenitor cell properties including clonogenicity, self-renewal, and multipotentiality. Clonogenicity refers to the ability of a single cell to yield a progeny. Self-renewal refers to the ability of cells to multiply themselves, with the offspring cells possessing the same properties as the parent cells. Multipotentiality is the capacity of a cell to differentiate into multiple, dissimilar cell lineages.
Fundamental to our understanding of regenerative endodontics is the knowledge of growth factors that affect a broad range of cellular activities including migration, proliferation, differentiation, and apoptosis of all dental pulp cells, including stem/progenitor cells ( Fig. 1 ). Growth factors and cytokines may act as signaling molecules that modulate cell behavior by mediating intracellular communication. Growth factors are polypeptides or proteins that bind to specific receptors on the surface of target cells. Growth factors can initiate a cascade of intracellular signaling, and act in either an autocrine or paracrine manner. Cytokines are typically referred to as immunomodulatory proteins or polypeptides. Cytokines are often used interchangeably with growth factors because many cytokines share similar actions with growth factors. As opposed to systemic effects by hormones on target cells, growth factors or cytokines typically act locally on target cells. This review first discusses the effects of various growth factors on dental pulp cells, and then explores how some of the growth factors may participate in dental pulp–dentin regeneration.
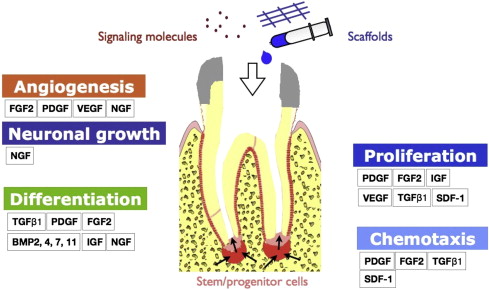
Platelet-derived growth factor
Platelet-derived growth factor (PDGF) is released by platelets, and has potency in promoting angiogenesis and cell proliferation. PDGF has 4 isoform homodimers AA, BB, CC, and DD, in addition to a heterodimer, PDGF-AB. PDGF dimers bind to 2 cell-surface receptors known as PDGFRα and PDGFRβ. The receptors form dimers before binding to different isoforms of PDGF. PDGF-AA, -BB, and -CC bind to PDGFR α/α, whereas PDGF-AB, -BB, -CC, and -DD bind to PDGFR α/β. PDGF-BB and -DD bind to PDGFR β/β. Therefore, the biological effect of PDGF depends on the expression level of PDGFR dimer on target cells.
The chemotaxis and proliferation of mesenchymal stem/progenitor cells can be induced by PDGF in the injury site. In trauma, hemorrhage is followed by blood-clot formation in dental pulp. Platelets in the blood clot release α-granules containing PDGFs and attract neutrophils and macrophages. These cells play key roles in early wound healing by producing other signaling molecules for the formation of granulation tissues. However, PDGFs appear to have little effect on the formation of the dentinlike nodule in dental pulp cells isolated from rat lower incisors, although PDGF-AB and -BB isoforms stimulate the expression of dentin sialoprotein (DSP). PDGFs stimulate cell proliferation and dentin matrix protein synthesis, but appear to inhibit alkaline phosphatase (ALP) activity in dental pulp cells in culture. DSP expression is inhibited by PDGF-AA but is enhanced by PDGF-AB and PDGF-BB, although the mineralized tissue formation is inhibited, suggesting diverging effects of PDGFs on odontoblastic differentiation depending on dimeric form. PDGFs enhance the proliferation of fibroblasts in human dental pulp. PDGF-BB may increase the expression of vascular endothelial growth factor (VEGF) in osteoblasts and promotes angiogenesis at the site of dental pulp injury. In vivo, PDGF promotes de novo formation of dental pulp–like tissues in endodontically treated human teeth that are implanted in rats.
Platelet-derived growth factor
Platelet-derived growth factor (PDGF) is released by platelets, and has potency in promoting angiogenesis and cell proliferation. PDGF has 4 isoform homodimers AA, BB, CC, and DD, in addition to a heterodimer, PDGF-AB. PDGF dimers bind to 2 cell-surface receptors known as PDGFRα and PDGFRβ. The receptors form dimers before binding to different isoforms of PDGF. PDGF-AA, -BB, and -CC bind to PDGFR α/α, whereas PDGF-AB, -BB, -CC, and -DD bind to PDGFR α/β. PDGF-BB and -DD bind to PDGFR β/β. Therefore, the biological effect of PDGF depends on the expression level of PDGFR dimer on target cells.
The chemotaxis and proliferation of mesenchymal stem/progenitor cells can be induced by PDGF in the injury site. In trauma, hemorrhage is followed by blood-clot formation in dental pulp. Platelets in the blood clot release α-granules containing PDGFs and attract neutrophils and macrophages. These cells play key roles in early wound healing by producing other signaling molecules for the formation of granulation tissues. However, PDGFs appear to have little effect on the formation of the dentinlike nodule in dental pulp cells isolated from rat lower incisors, although PDGF-AB and -BB isoforms stimulate the expression of dentin sialoprotein (DSP). PDGFs stimulate cell proliferation and dentin matrix protein synthesis, but appear to inhibit alkaline phosphatase (ALP) activity in dental pulp cells in culture. DSP expression is inhibited by PDGF-AA but is enhanced by PDGF-AB and PDGF-BB, although the mineralized tissue formation is inhibited, suggesting diverging effects of PDGFs on odontoblastic differentiation depending on dimeric form. PDGFs enhance the proliferation of fibroblasts in human dental pulp. PDGF-BB may increase the expression of vascular endothelial growth factor (VEGF) in osteoblasts and promotes angiogenesis at the site of dental pulp injury. In vivo, PDGF promotes de novo formation of dental pulp–like tissues in endodontically treated human teeth that are implanted in rats.
Transforming growth factor β
The transforming growth factor β (TGFβ) family comprises a group of diverse growth factors including TGFβ, bone morphogenetic proteins (BMPs), growth/differentiation factors (GDFs), anti-Mullerian hormone (AMH), activin, and nodal. TGFβ is composed of approximately 390 amino acids, which are released mainly from platelets, macrophages, and bone. This inactive polypeptide undergoes proteolytic cleavage to create the active C-terminal 112-amino-acid form. The active form of TGFβ dimerizes to form 25-kDa homodimers. The 3 isoforms present in mammals, TGFβ1, TGFβ2, and TGFβ3, are detected in human dentin. Inactive TGFβ exists as a large latent complex. After proteolytic cleavage, the active TGFβ binds to the type II receptor (TGFβRII) and recruits type I receptor (TGFβRI) to dimerize. TGFβRI, in turn, phosphorylates the intracellular proteins SMAD (homologues of Drosophila proteins including Caenorhabditis elegans protein [SMA] and mothers against decapentaplegic [MAD]), in particular, SMAD2 and SMAD3. The activated SMAD complex translocates to the nucleus and activates downstream TGFβ gene transcription.
The effect of TGFβ is highly variable and dependent on the type of cells and tissues. TGFβ1 regulates a wide range of cellular activities, such as cell migration, cell proliferation, cell differentiation, and extracellular matrix synthesis. TGFβ1has been shown to increase cell proliferation and production of the extracellular matrix in dental pulp tissue culture, and promotes odontoblastic differentiation of dental pulp cells. The effect of TGFβ1 can be synergistically upregulated by fibroblast growth factor 2 (FGF2), as evidenced by the increased ALP activity, the formation of mineralized nodule, and the expression of DSP and dentin matrix protein 1. The dentinogenic ability of dental pulp cells in the mechanically exposed dental pulp of dog teeth is shown to be induced by exogenous TGFβ1. TGFβ is chemotactic on dental pulp cells in vitro. TGFβ1 also plays an important role in the immune response during dental pulp injury.
Bone morphogenetic protein
BMPs comprise a subgroup of the TGFβ superfamily and are involved in many biological activities including cell proliferation, differentiation, and apoptosis. BMPs have strong osteoinductive and chondrogenic effects. BMP2 was discovered by Urist, who showed ectopic bone formation in connective tissues by transplanted dimineralized bone. Later some of BMPs were identified, purified and sequenced from proteins extracted from bone. To date, more than 20 BMPs have been identified and characterized, among which GDFs are included. Unlike TGFβ, BMPs are secreted as an active form of 30- to 38-kDa homodimers after proteolytic cleavage of a synthesized form composed of 400 to 525 amino acids. Two TGFβ receptors (type I and type II) are known to be involved in the BMP signaling pathway. The activity of BMPs is regulated by the antagonists of BMPs such as noggin and chordin. This modulation of BMP activity by the BMP antagonists may have a critical role in tooth development.
BMP2, BMP4, BMP7. and BMP11 are of clinical significance because of their role in inducing mineralization. Human recombinant BMP2 stimulates the differentiation of dental pulp cells into odontoblasts, inducing mRNA expression of dentin sialophosphoproteins (DSPPs) and higher ALP activity on BMP2 application, but has no effect on cell proliferation. DSPP expression and odontoblastic differentiation are regulated likely via BMP2-induced signaling by nuclear transcription factor Y. BMP2 also stimulates the differentiation of dental pulp stem/progenitor cells into odontoblasts in vivo and in vitro. Human recombinant BMP2 or BMP4 induces dentin formation when used in capping materials over amputated canine pulp. Osteodentin formation occurs in amputated canine pulps treated with BMPs in collagen matrix. Bovine dental pulp cells treated with BMP2 and BMP4 differentiate into preodontoblasts. BMP7, also known as osteogenic protein 1, promotes dentin formation when placed over amputated dental pulp in macaque teeth. The dentinogenic effect of BMP7 on amputated dental pulp has been shown in several animal models including rats, ferrets, and miniature swine. Dental pulp cells transfected with BMP11, also known as GDF11, yields mineralization. Dentin matrix protein 1, ALP, DSPP, enamelysin, and phosphate-regulating gene are highly expressed in BMP11-transfected cells. Transplantation of BMP11-transfected cell pellets induces formation of dentinlike tissue on amputated dental pulp in dogs. Ultrasound-mediated gene delivery of BMP11 stimulates odontoblastic differentiation of dental pulp stem/progenitor cells in vitro and reparative dentin formation in vivo.
Vascular endothelial growth factor
VEGF is a heparin-binding protein with specific affinity to endothelial cells, and plays a key role in angiogenesis. The functions of VEGF involve the proliferation of endothelial cells and their enhanced survival, stimulating neovascularization in the area of injury. The VEGF family includes VEGF-A, VEGF-B, VEGF-C, VEGF-D, and placenta growth factor. Among these isoforms, VEGF-A is the most versatile in function. VEGF-A, also known as vascular permeability factor, promotes cell migration, cell proliferation, vasodilatation, and vascular permeability by binding to 2 tyrosine kinases receptors, VEGFR1 and VEGFR2. VEGF increases microvessel density of the dental pulp when tooth slices containing severed dental pulp were treated with VEGF and implanted into subcutaneous tissues of severely combined immunodeficiency (SCID) mice.
VEGF appears to induce the differentiation of human dental pulp cells into endothelial cells. Dental pulp cells become positive for CD29, CD44, CD73, CD105, and CD166, but negative for CD14, CD34, and CD45 after VEGF treatment. VEGF increases the expression of VEGFR1 (fms-like tyrosine kinase, Flt-1) and VEGFR2 (kinase-insert domain containing receptor, KDR) and microvessel formation in a 3-dimensional fibrin mesh seeded with dental pulp cells. However, VEGF treatment does not appear to promote CD31, CD34, and CD144 positivity in dental pulp cells that are positive for CD29, CD90, CD105, CD166, CD146, and STRO-1. Of note, VEGF increases the proliferation and osteogenic differentiation of dental pulp cells under osteogenic conditions, suggesting a possible stimulatory role of VEGF in osteogenesis.
Fibroblast growth factor
Fibroblast growth factor (FGF) plays key roles in cell migration, proliferation, and differentiation during embryonic development and wound healing. To date 22 members have been identified in humans, of which FGF2 appears to be significant in regeneration of the pulp–dentin complex. Four FGF receptors, FGFR1 through FGFR4, are expressed in humans. Signal transduction is mediated by interaction between FGFs with the ability to bind to heparan sulfate and heparan sulfate proteoglycans on cell surface. FGF2 is a basic FGF, whereas FGF1 is acidic. FGF2 regulates tooth morphogenesis by controlling cell proliferation and differentiation. FGF2 is a potent angiogenic factor that stimulates formation of new blood vessels in the dental pulp along with PDGF and VEGF. Given its role in cell proliferation and angiogenesis, FGF2 acts as an early stimulating factor in formation of granulation tissue during wound healing.
FGF2 induces the migration of dental pulp cells. Using a transwell migration assay, significantly more dental pulp cells are recruited by basic FGF (FGF2) into a 3-dimensional collagen gel than compared with controls without cytokines and BMP7. FGF2 also stimulates the proliferation of dental pulp cells without differentiation, whereas FGF2 combined with TGFβ1 induces differentiation of dental pulp cells into odontoblast-like cells, and synergistically upregulates the effect of TGFβ1 on odontoblast differentiation. The FGF2 on exposed dental pulp in rat molars induces vascular invasion and cell proliferation early in wound healing. Also, FGF2 stimulates reparative dentin formation or dentin particles in the exposed pulp.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


