Periodontal diseases are the most common human diseases globally, with gingivitis affecting up to 90% and periodontitis affecting 50% of adults. Tooth enamel is the only nonshedding tissue in the human body. In the absence of proper oral hygiene measures, microbial biofilm (dental plaque) develops on the teeth to include more than 700 different bacterial species, along with viruses, fungi, archea, and parasites. With time, ecological imbalances promote the growth of selected commensal species that induce host inflammatory pathways resulting in tissue destruction, including ulceration of the periodontal epithelium.
Key points
- •
Infection of the periodontal tissues causes inflammatory responses, both locally and systemically.
- •
Routine activities, such as chewing hard food items or tooth brushing or flossing, and periodontal treatment cause bacteremia in persons with periodontal infection.
- •
Periodontal treatment can lower levels of oral bacteria, several systemic disease endpoints, and markers of inflammation, and hence does influence systemic diseases.
- •
There is insufficient scientific evidence to claim that periodontal treatment should be performed solely to prevent or treat systemic diseases.
Introduction
Much attention is drawn to the notion that oral health is an important and indispensable element of general health. The lay press has widely touted the links between oral and systemic health, but not always with attention to the quality of the source information and with close scrutiny of the underlying scientific evidence. Indeed, an entire industry based on the oral-systemic relationship has developed and gained foothold in the conscience of professional colleagues and the public alike. However, claims are often loosely based on scientific evidence and are the result of overinterpretation of the available data (recall “Floss or Die”). With patients accumulating widely available (mis)information, it is increasingly important for the dental practitioner to be knowledgeable about the actual current scientific evidence regarding the effects of periodontal disease and its treatment on systemic health.
From public health and economic viewpoints, this is an important issue. Given that the estimated diabetes-related costs in the United States amount to $245 billion, with $176 billion in direct medical and $69 billion indirect (disability, work loss, premature death), the possibility that dental professionals could improve the quality of life of patients and diminish the economic burden for individuals affected and for society is an intriguing notion.
The overarching goal of this article is to summarize the existing evidence for the effects of periodontal treatment on general health. The specific goals are to provide an update on the state of the science regarding mechanisms that may explain the connections between periodontal disease and systemic diseases and effects of periodontal treatment on general health.
This goal will enable the reader to
- •
Understand that the underlying mechanisms are similar for most all of the diseases described
- •
Understand the systemic effects of periodontal treatment
- •
Be able to critically interpret future scientific reports
- •
Explain to their patients what is known and what is not known
- •
Avoid the pitfalls of the current trends to overinterpret the evidence
- •
Practice evidence-based dentistry.
Scannapieco and colleagues described in a comprehensive review published in January 2010 the then existing evidence for the effects of periodontal treatment on various general diseases. Therefore, this review focuses on the evidence published since 2010 and preferentially cites systematic reviews and meta-analyses in the relevant areas because they pool several studies to gain more weight and statistical power for their conclusions.
Periodontitis—What Is It?
Everyone knows what periodontal disease is—or do they? Dental clinicians know the condition when they see it, but their diagnosis and successful treatment will depend on their knowledge of each individual patient. Research teams have historically created their own periodontitis case definitions, based on a multitude of periodontal parameters. This lack of a global, generally accepted and applied case definition for periodontitis is often overlooked, but is one of the most important factors in periodontal research that prevents direct comparison of results generated by different study teams. For example, Manau and collaborators examined 1296 individuals while using more than 50 different measures of periodontitis used in 23 different published studies. Then they applied 14 periodontitis published case definitions to their data and found the prevalence of periodontitis in their study group ranged from 2.2% to 70.8%, with a mean of 35.9% and a median of 29.7%, depending on how periodontitis was defined. So whether an association is statistically significant can depend on which measures were made and on the definition of the disease. This disparity in definitions is one of the main reasons it is so difficult to compare the results of different studies—and so challenging to come to firm conclusions about the strength of the association between periodontal disease and treatment and any systemic disease or condition.
Introduction
Much attention is drawn to the notion that oral health is an important and indispensable element of general health. The lay press has widely touted the links between oral and systemic health, but not always with attention to the quality of the source information and with close scrutiny of the underlying scientific evidence. Indeed, an entire industry based on the oral-systemic relationship has developed and gained foothold in the conscience of professional colleagues and the public alike. However, claims are often loosely based on scientific evidence and are the result of overinterpretation of the available data (recall “Floss or Die”). With patients accumulating widely available (mis)information, it is increasingly important for the dental practitioner to be knowledgeable about the actual current scientific evidence regarding the effects of periodontal disease and its treatment on systemic health.
From public health and economic viewpoints, this is an important issue. Given that the estimated diabetes-related costs in the United States amount to $245 billion, with $176 billion in direct medical and $69 billion indirect (disability, work loss, premature death), the possibility that dental professionals could improve the quality of life of patients and diminish the economic burden for individuals affected and for society is an intriguing notion.
The overarching goal of this article is to summarize the existing evidence for the effects of periodontal treatment on general health. The specific goals are to provide an update on the state of the science regarding mechanisms that may explain the connections between periodontal disease and systemic diseases and effects of periodontal treatment on general health.
This goal will enable the reader to
- •
Understand that the underlying mechanisms are similar for most all of the diseases described
- •
Understand the systemic effects of periodontal treatment
- •
Be able to critically interpret future scientific reports
- •
Explain to their patients what is known and what is not known
- •
Avoid the pitfalls of the current trends to overinterpret the evidence
- •
Practice evidence-based dentistry.
Scannapieco and colleagues described in a comprehensive review published in January 2010 the then existing evidence for the effects of periodontal treatment on various general diseases. Therefore, this review focuses on the evidence published since 2010 and preferentially cites systematic reviews and meta-analyses in the relevant areas because they pool several studies to gain more weight and statistical power for their conclusions.
Periodontitis—What Is It?
Everyone knows what periodontal disease is—or do they? Dental clinicians know the condition when they see it, but their diagnosis and successful treatment will depend on their knowledge of each individual patient. Research teams have historically created their own periodontitis case definitions, based on a multitude of periodontal parameters. This lack of a global, generally accepted and applied case definition for periodontitis is often overlooked, but is one of the most important factors in periodontal research that prevents direct comparison of results generated by different study teams. For example, Manau and collaborators examined 1296 individuals while using more than 50 different measures of periodontitis used in 23 different published studies. Then they applied 14 periodontitis published case definitions to their data and found the prevalence of periodontitis in their study group ranged from 2.2% to 70.8%, with a mean of 35.9% and a median of 29.7%, depending on how periodontitis was defined. So whether an association is statistically significant can depend on which measures were made and on the definition of the disease. This disparity in definitions is one of the main reasons it is so difficult to compare the results of different studies—and so challenging to come to firm conclusions about the strength of the association between periodontal disease and treatment and any systemic disease or condition.
Systemic effects of periodontal infection
How Does Periodontal Infection Affect Systemic Health?
Fig. 1 depicts the 3 major mechanisms by which periodontal infection is thought to affect the rest of the body. Because these 3 mechanistic pathways also present the options for periodontal treatment to interrupt the deleterious effects, the evidence will be presented based on these pathways, with an emphasis on inflammation as the predominant mechanism.
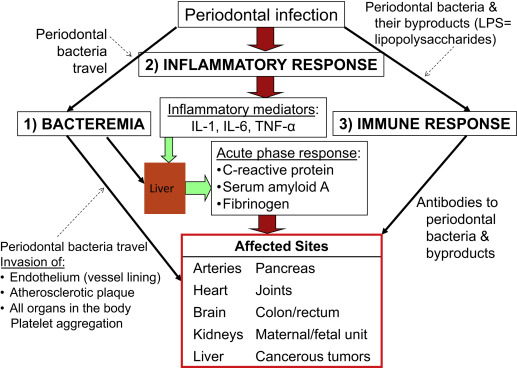
Bacteremia
The bacteria in deep periodontal pockets can easily penetrate through ulcerations of the inflamed epithelium into the bloodstream. Once in the blood, the bacteria can travel everywhere in the body, and many of the species have developed sophisticated ways to survive the host defense system. Not only can they survive; when they reach an inviting location, they make a landing and multiply in that remote location, for instance, an atherosclerotic plaque. The frequent, but consistent, dissemination of oral microbes via the bloodstream causes a chronic inflammatory response and can occur on daily activities, as reviewed by Tomas and colleagues : chewing of hard food items or chewing gum, tooth brushing, and flossing.
Invasive dental procedures also can cause bacteremia, for instance: tooth extraction, endodontic procedures, oral surgical treatment, nonsurgical soft tissue treatment, nonsurgical periodontal therapy, and periodontal probing. Even though bacteremia can be identified within 1 to 5 minutes after a procedure or activity, it is typically transient, with the bacteria usually undectable in the blood within 15 to 30 minutes, although bacteremia on chewing, tooth brushing, and scaling can last longer in persons with periodontitis.
The oral bacterial burden
The tooth is the only tissue that does not shed its surface cells and constantly undergoes renewal; this is the reason that bacterial biofilm can be established on teeth and over time grow and organize itself. This biofilm consists of a complicated system of bacteria and “glue” (extracellular polymers) that binds the members of this community together and makes it difficult for antibiotics to penetrate and reach bacteria that otherwise would be sensitive to their effects.
Novel laboratory techniques and development of powerful computers and software have enabled an exponentially growing understanding of the composition and function of oral microbiomes (the collective pool of microbes consisting of bacteria, virus, fungi, and archea) in various locations in the oral cavity.
It is important for periodontal treatment to target this pathway of bacteremia with the goal of reducing the quantity and changing the composition of the dental plaque in order to reduce the presence of particularly virulent bacteria that would cause bacteremia.
Inflammation
Only recently has the pivotal role of inflammation as the mechanistic basis for many chronic diseases come to the attention of health care professionals and medical researchers. For instance, in 1997, Ridker and colleagues suggested that inflammation, manifested by the general acute phase inflammatory biomarker C-reactive protein (CRP), is an important risk factor for a first thrombotic event (myocardial infarction or ischemic stroke). Ten years later, Ridker and Silvertown argued that CRP, which has been shown to be elevated in patients with periodontitis, might be more important than low-density cholesterol levels as a cause of atherosclerosis. It was therefore suggested that periodontal disease may indirectly influence”risk, manifestation and progression of vascular events.”
What is inflammation?
Inflammation is a biological process that describes the host’s response against any attack on its integrity, including exogenous infections; bacterial overgrowth of commensal periodontal bacteria that flourish due to environmental changes; injuries, with or without breakage of the skin or any other visible surface; sunburn; chronic autoimmune disease; physical, chemical, thermal, or hormone-induced irritation; and exercise. Inflammation is a response for the host’s protection and consists of
- •
Vascular reactions: vasodilation, increased permeability
- •
Plasma cascade systems producing molecular mediators: coagulation system (thrombin); fibrinolysis system (plasmin)
- •
Immune cell-derived mediators: enzymes; vasoactive amine (histamine); cytokines (tumor necrosis factor-α [TNF-α], interleukin-1 [IL-1], interferon-γ [IFN-γ]); chemokines (IL-8); soluble gas (nitric oxide)
- •
Leukocyte extravasation (the process of movement of the leukocytes from the blood through the vessel wall to the site of injury)
- •
Phagocytosis (engulfing and ingestion of bacteria, cell debris, and so on by phagocytes [eg, macrophages, neutrophils, and monocytes]).
The purposes of inflammation are to eliminate the initial cause of cell injury; remove necrotic cells and tissues damaged both from the original insult and from the subsequent inflammatory process; and to initiate tissue repair. The classic cardinal signs of inflammation are heat, pain, redness, swelling, and loss of function. Inflammation can be local or systemic; acute or chronic; and of various morphologic patterns (granulomatous, fibrinous, purulent, serous, or ulcerative). Resolution of inflammation occurs via several mechanisms, for example:
- •
Production and release of anti-inflammatory substances
- •
Apoptosis (normal, programmed cell death) of pro-inflammatory cells
- •
Production of resolvins that promote healing, especially in the presence of aspirin.
If the inflammation does not resolve by complete healing, an abscess can form. Otherwise, the inflammation becomes chronic. Tissue affected by chronic inflammation will be dominated by macrophages whose toxins eventually will cause destruction of both foreign and host soft and hard tissue, as seen in chronic periodontitis that, if left untreated, may result in tooth loss.
Inflammation originating in the periodontal microbiome
It is now gaining acceptance that a microbial imbalance (dysbiosis) in the oral microbiome mediates inflammation not only locally in the periodontal tissues but also systemically, via the host response to the bacteremia and via the inflammatory biomarkers from the periodontium spreading in the body. Inflammation is thought to be the common mechanism underlying the links between periodontitis and most of the systemic diseases that periodontitis affects.
Oral biofilm on the teeth and in the periodontal pocket induces inflammation that will continue as long as biofilm is present. Periodontitis is the polymicrobial inflammatory disease that emerges because of disruption of the ecologic equilibrium (homeostasis) between the periodontal microbiome and its host that then can enter a vicious cycle of increasing imbalance and loss of alveolar bone. The inflammatory mediators that are produced in the periodontium will cause local inflammatory effects, but also spill into the bloodstream and travel to all parts of the body. However, the intensity of the inflammatory response will depend more on the individual host’s immune system than simply the amount and microbial composition of the dental plaque.
Therefore, the inflammatory pathway is the most important target for periodontal therapy.
Immune response
The immune system will be stimulated to form antibodies to the bacteria and their toxins that are found both in the periodontium and in distant sites due to bacteremia. This pathway seems to be protective and to not have deleterious effects on the development of diseases, although cross-reactive antibodies to heat shock proteins from bacteria in the oral biofilm may be formed and could contribute to atherogenesis.
Systemic effects of periodontal treatment
The often overlooked, overarching goal for periodontal therapy is to improve or maintain the patient’s quality of life, via the preservation of functional teeth surrounded by a healthy periodontium. A multitude of studies have demonstrated that nonsurgical periodontal treatment is effective in improving the periodontal health status, also in persons who suffer from various other medical conditions, such as diabetes, kidney disease, and pregnancy. Moreover, successful periodontal intervention may not only reduce periodontal disease and extend tooth survival but also prevent the initiation or progression of, as well as ameliorate, several chronic systemic diseases.
Effects on Bacterial Load, Inflammation, and Immune Response
It has been demonstrated that periodontal treatment can reduce the
- •
Bacterial load on the teeth
- •
General inflammatory response to the microbes usually associated with periodontal breakdown and their toxins
- •
Specific immune response to these microbes and toxins.
Therefore, it intuitively follows that such reduction of bacteria and inflammatory biomarkers that cause or aggravate the systemic consequences of their circulation by periodontal treatment should have significant and clinically relevant effects on the systemic diseases that are wholly or partially caused by these bacteria and substances.
However, it is not straightforward to measure the end effects of reduction of oral bacteria directly on disease outcomes for a variety of reasons, including
- •
Chronic, multicausal, and complex nature of the diseases
- •
Changing composition of the microbiomes in the oral cavity
- •
Variability in methods used to detect the microbes
- •
Inability to identify many of the microbes involved
- •
Variability of case definitions for periodontitis and many chronic diseases.
Effects of Periodontal Treatment on Local and Systemic Inflammation
The 3 main systemic disease pathways for periodontal infection to influence the body and that periodontal treatment may therefore target are depicted in Fig. 1 , namely, (1) bacteremia, (2) inflammatory response, and (3) immune response.
Bacteremia
- •
Sudden, transient increase in blood levels of periodontal bacteria immediately following tooth brushing or periodontal treatment
- •
Decreased blood levels of periodontal bacteria on resolution of the immediate bacteremia in saliva, supragingival and subgingival plaque
- ○
Total periodontal bacterial load
- ○
Aggregatibacter actinomycetemcomitans
- ○
Fusobacterium nucleatum
- ○
Porphyromonas gingivalis
- ○
Prevotella intermedia
- ○
Several other periodontal bacteria
- ○
Inflammatory response
- •
Sudden, transient increase in blood levels of inflammatory markers immediately on periodontal treatment
Followed after 1 week by
- •
Decreased levels of pro-inflammatory mediators
- ○
Cytokines (cell signaling proteins)
- ▪
Interleukins (IL-1, IL-6, IL-8, IL-18)
- ▪
Tumor necrosis factors (TNF-α, TNF-β)
- ▪
IFN-γ
- ▪
- ○
- •
Decreased levels of other acute phase reactants
- ○
CRP with a significant reduction of 0.50 mg/mL
- ○
Serum amyloid A
- ○
Fibrinogen
- ○
Plasminogen-activator inhibitor 1
- ○
- •
Decreased levels of blood glucose
- ○
Fasting
- ○
Random
- ○
Glycated hemoglobin (Hb A1c , A 1c )
- ○
Immune response
- •
Only a modest or temporary decrease in levels of antibodies to periodontal bacteria and their toxins
- ○
Immunoglobulins (IgG, IgA)
- ○
A Special Note on Antibodies (Immune Response) to Periodontal Bacteria
Antibodies to periodontal bacteria persist in the human body long after periodontal treatment, for instance, after full-mouth extraction. Lakio and colleagues showed that serum and salivary immunoglobulin to P gingivalis and A actinomycetemcomitans remained remarkably stable over a period of 15 years in all individuals who at the end were periodontally examined and found to have slight to moderate periodontitis.
Despite successful periodontal therapy, antibodies to 16 of 19 periodontal bacteria declined only modestly after 30 months and remained much higher than in the control group with periodontal health. Therefore, Papapanou and colleagues suggest that such antibodies may reflect a history of periodontal infection, not only current periodontal status.
Serum antibody titers against bacteria associated with periodontitis are linked to several systemic diseases, such as diabetes and metabolic syndrome. Although no treatment studies have been published, several interesting observations supporting links to other diseases also are reported, such as impaired cognitive function:
- •
High-serum IgG levels to an oral bacterium, Actinomyces naeslundii, doubles the hazard risk for development of new Alzheimer disease over a period of 5 years, whereas high anti- Eubacterium nodatum IgG was associated with half the risk of developing Alzheimer disease over a period of 5 years.
- •
In an analysis of the US population-based NHANES III data, high levels of antibodies to P gingivalis were associated with poorer performance in three cognitive tests (cognitive impairment).
Gingivitis Treatment Alone Also Has a Positive Effect on Bacterial Load and Inflammation
Even in otherwise healthy persons with gingivitis only (no periodontitis), ultrasonic debridement supplemented with rinsing with mouthwash containing essential oils is shown to lead to a decrease in levels of overall bacterial load, Tannerella forsythia, A actinomycetemcomitans, gingival crevicular fluid (GCF) volume, and IL-1β. Rinsing with essential oil mouthwash alone also reduced the levels of selected bacteria individually and the total bacterial load by half, on average, with 21% to 52% in saliva, on average 53% in supragingival plaque, and by 21% to 38% in subgingival plaque. However, these reductions by oral rinse were less pronounced than after scaling and root planing in the same study (52%–63% in saliva, 68%–81% in supragingival plaque, and 68%–93% in subgingival plaque).
Effects on Systemic Diseases
Diabetes
Diabetes is a growing public health problem in the United States and in many other countries. Almost half of the roughly 240 million adults living in the United States have hyperglycemia ( Box 1 ). Prediabetes is defined in Table 1 .
-
It is estimated that among the US adults, there are:
-
21.0 million diagnosed with manifest diabetes
-
8.1 million with undiagnosed manifest diabetes
-
28.9 million with manifest diabetes
-
86.0 million with prediabetes (up to 90% being unaware)
-
114.9 million adults with diabetes or prediabetes (see Table 1 )
-
| Panel A | Panel B | ||||
|---|---|---|---|---|---|
| HbA 1c (%) | Average Plasma Glucose (mg/dL) (CI) | HbA 1c (%) | Fasting Plasma Glucose (mg/dL) | 2-h OGTT (75 g Glucose) (mg/dL) | Diabetes Status |
| 4 | ∼ 68 | ≤5.6 | <100 | — | No diabetes |
| 5 | 97 (76–120) | n/a | |||
| 5.6 | ∼ 114 | — | |||
| 5.7 | ∼ 117 | 5.7–6.4 | 100–125 | — | Prediabetes |
| 6 | 126 (100–152) | n/a | |||
| 6.4 | ∼ 137 | — | |||
| 6.5 | ∼140 | ≥6.5 | ≥126 | ≥200 | Diabetes |
| 7 | 154 (123–185) | ||||
| 8 | 183 (147–217) | ||||
| 9 | 212 (170–249) | ||||
| 10 | 240 (193–282) | ||||
| 11 | 269 (217–314) | ||||
| 12 | 298 (240–347) | ||||
Table 1 illustrates how to translate Hb A1c into mean plasma glucose levels (Panel A) and to categorize diabetes status by HbA 1c or fasting or 1-hour postoral glucose tolerance test (OGTT) plasma glucose levels (Panel B).
The epidemic of obesity parallels the increasing prevalence of type 2 diabetes ( Fig. 2 ).
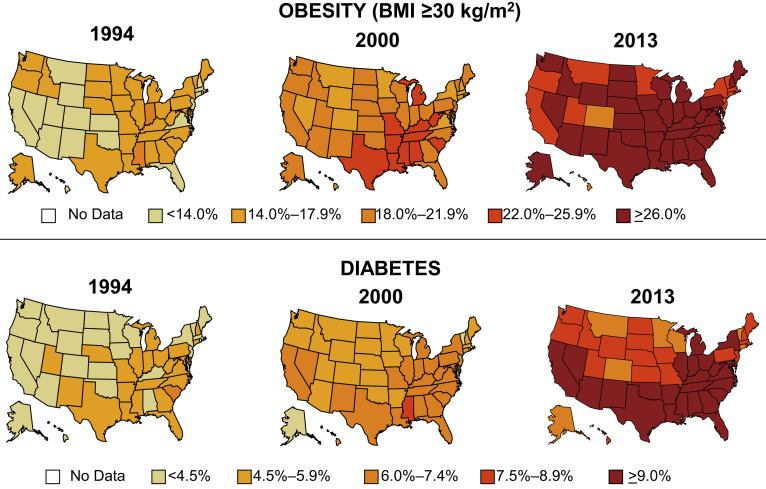
Periodontitis: associations with diabetes and prediabetes
There is a widespread belief that all persons with diabetes are at much higher risk for developing or having periodontitis and that the extent and severity of periodontitis are greatly increased. However, this is true mostly when the long-term blood sugar known as glycated hemoglobin (HbA 1c or A 1c ) exceeds about 7%, the threshold above which diabetes complications mostly occur and below which glucose levels generally are considered to be under control. Hyperglycemia negatively impacts the structure and function of many tissues. A large German population study illustrates this: the association between clinical attachment loss was stronger in poorly controlled diabetes than in well-controlled diabetes, and absent in prediabetes.
Nevertheless, a large study of mostly Hispanics in Northern Manhattan reported that periodontitis severity and extent was associated with both newly diagnosed prediabetes and diabetes. The periodontal breakdown in prediabetes ranged between that in persons with normal blood sugar levels and those with overt diabetes, suggesting hyperglycemia as potentially causing the periodontal damage even before the diagnosis of prediabetes. Persons with prediabetes had worse clinical periodontal health, radiographic bone loss, and self-reported oral health (gingival bleeding, chewing pain, dry mouth, and burning mouth) than individuals with normal glycemic levels.
Interestingly, even among persons with prediabetes, periodontal health assessed clinically, radiographically, and subjectively was better in those with better controlled prediabetes. Furthermore, hyperglycemia in smokers actually seemed to have a stronger effect on poor periodontal health than smoking. A systematic review and meta-analysis estimate that type 2 diabetes is 5 to 6 times more likely to develop in a person with prediabetes than in one with normal glucose levels.
Periodontal infection: effects on blood glucose levels
The first systematic review of the effect of periodontal infection on blood glucose levels included studies in which directionality could be determined, that is, periodontitis occurred before the outcome (increase in blood sugar level and its consequences), suggesting a potentially causal relationship. It was concluded that there is evidence to suggest that periodontal infection, often in a dose-response manner, contributes to
- •
Hyperglycemia in previously healthy individuals and in smokers
- •
Impaired glucose tolerance
- •
Development of prediabetes
- •
Development of manifest type 2 diabetes (40% higher odds in gingivitis and 50% in periodontitis; 3.5 times relative risk over a period of 5 years for those with 6+ mm pockets)
- •
Poorer glycemic control in existing type 2 diabetes
- •
Development of gestational diabetes
- •
Development of one or more components of metabolic syndrome
- •
Complications of diabetes
- ○
Retinopathy
- ○
Neuropathic foot ulceration
- ○
Kidney disease, including end-stage renal disease (also in type 1 diabetes)
- ○
Subclinical heart disease: thickening of the intima media of the carotid artery and more advanced calcification of atherosclerotic plaque
- ○
Myocardial infarctions and stroke (also in type 1 diabetes)
- ▪
Mortality due to cerebrovascular disease
- ▪
- ○
Mortality due to kidney disease
- ○
The notion that periodontal infection affects blood glucose is expected because elevation of glucose levels in the blood occurs as part of any inflammatory response, regardless of the location of the initiating sources.
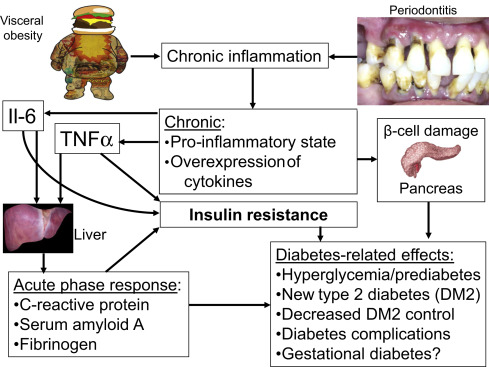
How Inflammation Leads to Hyperglycemia—What to Tell the Patient
Elevated blood sugar normally accompanies the inflammatory response, because energy is required for fending off intruders that are in places they should not be, such as bacteria in the bloodstream.
The main steps in the effect of periodontal infection on diabetes to be explained to patients are
- •
Bacteria from the periodontium cause general inflammation in the body
- •
Inflammation weakens the action of insulin (insulin resistance)
- •
Inflammation reduces the production of insulin in the pancreas
- •
Sugar builds up in the blood (hyperglycemia)
- •
Formation of long-term blood sugar (Hb A1c ) happens because of the high sugar concentration in the blood (hyperglycemia).
Fig. 3 illustrates conceptually the main mechanisms for how inflammation caused by periodontitis and obesity contributes to hyperglycemia and eventually to manifest type 2 diabetes.
Periodontal Treatment in Type 2 Diabetes: Effects on Glycemic Control
The enormity of the hyperglycemia problem in the United States is the reason that it is so important for the dental clinician to be aware and to contribute to amelioration of the issue as much as possible, even if modestly. The dental clinician will see increasing numbers of patients with diabetes, partly because of the increasing prevalence of obesity, partly because of the prevalence of type 2 diabetes increasing with age—a quarter of seniors 65 and older have overt diabetes—and people are keeping their teeth longer.
A multitude of clinical studies have been conducted in many different countries in study populations with a variety of ages, ethnicities, socioeconomic status, and so on. The vast majority of these studies show that it is possible for periodontal therapy to reduce the levels of HbA 1c in persons with type 2 diabetes and periodontitis. A 2014 study in India showed decreases in HbA 1c between 0.55 and 0.88 percentage points, illustrating that the greatest improvement was obtained in the group with poor (more than 8%) glycemic control, namely, from 9.79% to 8.91%.
The diabetes and periodontal therapy trial
The gold standard in interventional clinical research is the randomized controlled trial (RCT). Such a US federally funded (USD 15.4 million) study named the Diabetes and Periodontal Therapy Trial (DPTT) “was designed to determine whether nonsurgical periodontal therapy (scaling and root planing and supportive periodontal therapy), compared with no therapy, reduces HbA1c levels at 6 months in persons with type 2 diabetes and moderate to advanced chronic periodontitis.” Periodontal treatment was reported to have no effect on HbA 1c , even though the periodontal status improved significantly. This study was limited by the fact that the periodontal intervention was not definitive since, at 6 months after treatment, the participants had on average 20% sites with 4+ mm pockets and 10% (mean: 16 sites) at 5+ mm; more than 40% sites bleed on probing; and almost three-quarters (72%) of the sites had plaque. A detailed critique of this study and various comments were subsequently published in 2014.
Table 2 displays all published systematic reviews and meta-analyses conducted on studies regarding the effect of nonsurgical periodontal treatment on HbA 1c in type 2 diabetes. Such meta-analyses combine data from sufficiently similar studies to obtain more robust results.
| Author, Year | No. of Studies | No. of RCT | Pooled No. of Subjects | HbA 1c Change (Percentage Point) | 95% CI | P Value |
|---|---|---|---|---|---|---|
| Janket et al, 2005 | 4 | 1 | 268 | −0.66 f | −2.2; 0.9 | n/s |
| Darré et al, 2008 | 9 | 5 | 485 | −0.46 e | −0.82;−0.11 | .01 |
| Teeuw et al, 2010 | 5 | 3 | 180 | −0.40 f | −0.77; −0.04 | .03 |
| Simpson et al, 2010 (Cochrane Review) | 3 | 3 | 244 | −0.40 | −0.78; −0.01 | .04 |
| Sgolastra et al, 2013 | 5 | 5 | 315 | −0.65 f | −0.88; −0.43 | <.05 |
| Engebretson & Kocher, 2013 | 9 | 9 | 775 | −0.36 | −0.54; −0.19 | <.0001 |
| Liew et al, 2013 | 6 | 6 | 422 | −0.41 | −0.73; −0.09 | .013 |
| Wang et al, 2014 a | 3 | 3 | 143 | −0.24 e | −0.62; 0.14 | .217 |
| Sun et al, 2014 | 8 | 8 | 515 | 1.03%(3 mo) d | −0.31%;−1.70% d | .003 |
| Sun et al, 2014 (6 mos) | 3 | 3 | 150 | −1.18%(6 mo) d | −1.64% d ;−0.72 | <.001 |
| Additional reviews that each includes one study on type 1 diabetes | ||||||
| Corbella et al, 2013 (3 mo) b | 8 | 8 | 678 | −0.38 | −0.53; −0.23 | <.001 |
| Corbella et al, 2013 (6 mo) b | 3 | 3 | 235 | −0.31 | −0.74; 0.11 | .15 |
| Wang et al, 2014 c | 10 | 10 | 1135 | −0.36 | −0.52; −0.19 | <.0001 |
a Includes only studies using adjunct doxycycline.
b Includes one study with type 1 + type 2 diabetes subjects; 1 study with only type 1 diabetes subject.
c Includes one study with 39 type 1 + 66 type 2 diabetes subjects and 9 studies with type 2 diabetes subjects, including DPTT study.
d Percent of initial HbA 1c value; 3 mo/6 mo, 3/6 months after treatment.
Even though many individual nonsurgical periodontal treatment studies are conducted among small numbers of participants and use different exposure and outcome measures and case definitions of periodontitis, and even though the different meta-analyses do not include the same studies, the conclusions of the meta-analyses are remarkably similar:
- •
None show a posttreatment increase in HbA 1c
- •
All show a (short-term) posttreatment reduction in HbA 1c
- •
The mean HbA 1c reduction hovers around 0.4 percentage points.
Of special note is the Cochrane Collaboration systematic review from 2010 that, based on 3 studies, reported a decrease of 0.40 percentage points, but calls for “larger, carefully conducted and reported studies.” As well, it should be noted that the DPTT study is included in the 2014 meta-analysis by Wang and colleagues, but the HbA 1c decrease is still 0.36 (or 0.4) percentage points.
Is a reduction of 0.4 percentage points HbA 1c clinically meaningful?
When a person is diagnosed with type 2 diabetes, the first mode of action to bring the blood sugar under control is to try to improve eating and drinking habits and to do more physical exercise. If that is not sufficient, the first-line medication of choice is metformin taken by mouth, the actions of which are to suppress hepatic glucose production (gluconeogenesis), increase insulin sensitivity, and enhance peripheral glucose uptake while decreasing glucose absorption from the intestinal tract. The expected effect of metformin on HbA 1c is a reduction of about 1 percentage point, which is about twice that of which nonsurgical periodontal treatment is shown to lead to. Therefore, the effect of successful nonsurgical periodontal treatment may be clinically significant in diabetes management.
This improvement in glycemic control can help reduce the risk of potentially serious and even fatal complications of poorly controlled diabetes
- •
Retinopathy (blindness)
- •
Nephropathy (chronic kidney disease/renal failure/end-stage renal disease)
- •
Neuropathy (numbness or pain in feet or fingers, burning mouth syndrome, dry mouth/hyposalivation)
- •
Hypertension
- •
Heightened susceptibility to infections, delayed wound healing
- •
Amputation
- •
Cardiovascular disease (CVD)/myocardial infarction (premature death)
- •
Stroke (premature death)
Periodontal Treatment in Prediabetes: Effects on Blood Glucose Levels
Recent attention to the importance of prediabetes has resulted in the emergence of intervention studies among that population group. For instance, in 35 to 65 years old individuals with prediabetes, the HbA 1c level decreased significantly 3 months after nonsurgical periodontal treatment, namely from 6.08(±0.23)% to 5.67(±0.33)%. Another small study of persons between 35 and 75 years old with prediabetes reported a clinically and statistically significant decrease in HbA 1c level 3 months after nonsurgical periodontal treatment, namely, from 6.08(±0.51)% to 5.89(±0.45)%. Finally, among 66 persons with prediabetes, the HbA 1c level improved from an average of 6.0% (5.7%–6.3%) to 5.1% (4.9%–5.3%) in the 33 who received nonsurgical periodontal therapy only and from 6.2% (5.7%–6.4%) to 5% (4.8%–5.3%) in participants who additionally received oral doxycycline. Notably, the antibiotic made no difference in the clinical or glycemic outcomes.
In all 3 studies, nonsurgical periodontal treatment was sufficient to reverse prediabetes to healthy in a large proportion of the participants, with an HbA 1c level of 5.7% being the lower cut point for the diagnosis of prediabetes (see Table 1 ).
Gingivitis Treatment Only: Effect on Inflammation in Diabetes
Even in persons with type 2 diabetes and only gingivitis (not periodontitis), ultrasonic debridement (prophylaxis) leads to decreased gingival inflammation. Such 3-month routine prophylaxes also prevent progression of chronic periodontitis in both poorly controlled and well-controlled patients with type 2 diabetes mellitus. After professional prophylaxis, daily use of essential oils mouth rinse for 90 days is shown to lead to a decrease in levels of overall bacterial load, T forsythia, A actinomycetemcomitans, GCF volume, and IL-1β.
In summary
It makes intuitive sense that treatment of periodontal infection would diminish the inflammatory response and elevated blood glucose levels. This is because periodontal treatment is shown to reduce the levels of inflammatory mediators caused by periodontal infections, such as IL-β and TNF-α, which in turn reduce the blood glucose level that was elevated due to the inflammatory response to periodontal infection. Taking this chain of events further back, it could be speculated that effective daily oral hygiene measures, possibly supplemented with professional intervention, would contribute to a decrease in the bacterial burden from the biofilm, thereby help prevent or reduce the subsequent inflammation, and consequently that cleaning the teeth would function as both a preventive and a therapeutic measure.
There is evidence that nonsurgical periodontal treatment in persons with type 2 diabetes can
- •
Reduce systemic inflammation
- •
Improve periodontal health both in gingivitis only and in periodontitis
- •
Improve periodontal health both in poorly and in well-controlled diabetes
- •
Decrease levels of HbA 1c and thereby improve glycemic control in the short term (3 months)
What can be said to patients
- •
Periodontal disease can increase the amount of sugar in the blood
- •
Treatment of periodontal disease may lower the amount of sugar in the blood
- •
Treatment of periodontal disease may help control type 2 diabetes
- •
It is important to keep the mouth as clean as possible in people with diabetes.
Metabolic Syndrome
Metabolic syndrome is a cluster of metabolic and physiologic abnormalities that are risk factors for both diabetes and atherosclerotic CVD. A person is diagnosed with metabolic syndrome if 3 or more of the following conditions are present:
- •
Hyperglycemia (fasting glucose ≥100 mg/dL or receiving drug therapy for reduction)
- •
Hypertension (blood pressure ≥130/85 mm Hg or receiving drug therapy for reduction)
- •
Hyperlipidemia (triglycerides ≥150 mg/dL or receiving drug therapy for reduction)
- •
Low HDL cholesterol level (HDL-C <40 mg/dL in men or <50 mg/dL in women or receiving drug therapy for increase)
- •
Large waistline (abdominal or central obesity: waist circumference ≥102 cm [40 in] in men or ≥88 cm [35 in] in women; if Asian American, ≥90 cm [35 in] in men or ≥80 cm [32 in] in women).
Nonalcoholic Fatty Liver Disease
Nonalcoholic fatty liver disease (NAFLD) is a hepatic manifestation of metabolic syndrome. The highly virulent type of the periodontal bacterium P gingivalis is shown to be present with high frequency in oral samples from NAFLD sufferers. Importantly, nonsurgical periodontal treatment over a period of 3 months in such patients improved their liver function parameters, as measured by serum levels of aspartate aminotransferase and alanine aminotransaminase. The effects of periodontal treatment are mentioned under diabetes and cerebrovascular diseases, respectively.
In summary
There is evidence that
- •
Periodontitis is associated with metabolic syndrome (also in hemodialysis patients), diabetes, and gestational diabetes
- •
Serum antibody against specific periodontal bacteria is associated with metabolic syndrome
- •
Periodontitis is significantly associated with development of one or more components of metabolic syndrome over a period of 4 years
- •
Tooth loss is associated with metabolic syndrome
- •
Periodontal therapy improves total leukocyte count and levels of CRP, triglycerides, and HDL
- •
Tooth brushing at least once a day leads to
- ○
Lower levels of triglycerides
- ○
Lower prevalence and incidence (new cases) of metabolic syndrome
- ○
What can be said to patients
- •
Maintaining good periodontal health may control fat or blood sugar levels and help prevent metabolic syndrome.
Cerebrocardiovascular Disease
Bacteremia
Several reports have found periodontal bacteria in atheromatous plaque. T forsythia has been identified in the same location as hemoglobin and is therefore regarded as a potential trigger for intraplaque hemorrhage, which may increase the risk for plaque rupture. The periodontal bacterium, A actinomycetemcomitans , has been identified in the same person’s subgingival plaque, blood, and blood vessels, demonstrating the spread via bacteremia. Dorn and colleagues showed for the first time in 1999 that periodontal bacteria are able to invade both human coronary artery endothelial cells and coronary artery smooth muscle cells, so persons with periodontal infection have greater risk of myocardial infarction. Even though periodontal intervention studies with clinically important outcomes such as myocardial infarction and ischemic stroke are lacking, evidence exists that
- •
Viable bacteria originating from the periodontium are found in atherosclerotic plaque
- •
Periodontal bacteria can contribute to atherogenesis
- •
Periodontal bacteria most likely can contribute to destabilization of atherosclerotic plaque
- •
P gingivalis infection is associated with aortic aneurysms and with proliferation of smooth muscle cell tissue in developing aneurysms.
Inflammation
Cardiovascular and cerebral diseases and events considered in this review are those caused by atherosclerosis, which is related to inflammation. Atherosclerosis is the thickening and subsequent calcification of the inner artery wall with deposition of a waxy substance (plaque) that consists of fat, cholesterol, calcium, and other substances from the blood. Atherosclerosis may affect arteries anywhere in the body.
Previous notions of the cause of atherosclerosis centered on the role of lipids, but recent research has indicated that inflammation plays a crucial role. For example, as recently as in 2013, Abdelbaky and colleagues demonstrated for the first time in humans that inflammation actually precedes calcification of the arterial wall in atherogenesis.
Periodontal disease that is inflammatory in nature is recognized as an independent risk factor for atherosclerosis, regardless of other risk factors. For example, periodontitis was shown to predict recurrent cardiovascular events in a study that followed 668 survivors of a myocardial infarction for 3 years. Never-smokers with periodontal disease were found to have a 43% higher risk of experiencing another fatal or nonfatal cardiovascular event than such individuals without periodontal disease. In Japanese and US participants with clinical echocardiograms, 77% had cardiac calcifications and 51% had moderate to severe periodontitis. Calcification of structures of the heart is viewed as a marker of subclinical atherosclerosis. Not only were periodontitis and cardiac calcification significantly associated up on adjustment for confounders, their relationship was also dose dependent regarding severity of both conditions.
Periodontal treatment: effect on cardiovascular diseases and events
A systematic Cochrane Review from 2014 concluded that there were no studies that assessed periodontal therapy for primary prevention of CVD in persons with periodontitis. Although another 2014 systematic review also agreed that no trials used hard clinical endpoints of CVD, it concluded that periodontal treatment significantly reduces several risk factors for atherosclerotic CVD.
Periodontal treatment: effects on risk factors for cardiovascular events
Instead of exploring the direct effect of periodontal treatment on the actual end points, such as heart attacks, the effect on factors that are known to be risk factors for such events has been investigated. There is evidence that periodontal therapy improves
- •
Levels of several pro-inflammatory cytokines
- •
Concentrations of CRP in patients without and with coronary heart disease
- •
Levels of fibrinogen and white blood cells
- •
Levels of total cholesterol
- •
Levels of low-density (“bad”) cholesterol
- •
Levels of high-density (“good”) cholesterol
- •
Triglyceride levels
- •
Blood glucose levels (hyperglycemia is a risk factor for CVD)
- •
Endothelial function, especially in persons with diabetes and CVD
- •
Systolic and diastolic blood pressure
- •
Left ventricular mass (improvement is a reduction)
- •
Pulse-wave velocity (measure of arterial function).
Of particular interest is that the concentrations of the cytokines IL-18 and IFN-γ are shown to be decreased by 90% at 12 months postperiodontal therapy. This finding is of special interest because levels of IL-18 are reported to significantly predict acute myocardial infarction in people with coronary artery disease and major cardiovascular events 6 months after hospitalization for acute coronary syndrome. IL-18 independently predicts congestive heart failure, myocardial infarction, cardiovascular death, and all-cause non-CVD death.
Periodontal treatment: effect on intima media thickness and endothelial dysfunction
Increased intima media thickness (IMT) assessed by ultrasound is a marker for atherosclerosis and is demonstrated to be associated with future cardiovascular events. Endothelial dysfunction is an impairment of the arteries to dilate and contract properly to adjust blood pressure and is a sign of atherosclerotic changes in vessels. It is most often measured on the brachial artery on the inside of the upper arm or on the carotid artery on the side of the neck.
A 2014 meta-analysis concluded that the presence of periodontitis is associated with an increased IMT and a decreased flow-mediated dilation (a measure of endothelial dysfunction), with both differences being clinically significant. Importantly, the authors also concluded that periodontal treatment leads to improvement in endothelial function. Later, it was demonstrated that periodontal therapy can lead to decreased IMT of the carotid artery.
Rheumatoid Arthritis
Rheumatoid arthritis (RA) is an autoimmune, systemic inflammatory chronic disorder that leads to pain and deformity of the joints. Inflammatory mediators, such as TNF-α, and levels of P gingivalis are known to aggravate or partially cause the disease. The presence of periodontitis is also shown to hamper RA treatment with TNF-α blockers.
The periodontal bacterium P gingivalis is unique among the oral flora because it possesses an enzyme needed for protein citrullination, a major hallmark of RA. Therefore, it would follow logically that reduction of the P gingivalis level and inflammation levels via periodontal treatment should decrease the severity of RA.
The following is a brief summary of the most recent relevant evidence for periodontal therapy affecting RA:
- •
Based on 3 treatment studies, a 2013 systematic review concluded that evidence is emerging for periodontal treatment leading to improvement in biochemical markers in persons suffering from RA.
- •
A 2014 systematic review concluded, based on 5 eligible intervention studies, that nonsurgical periodontal treatment leads to significant improvement in both biomarkers and clinical arthritis manifestations , citing reductions in erythrocyte sedimentation rate (ESR) and a trend toward decreasing levels of TNF-α and the 28-joint count disease activity score (DAS28) assessed by CRP or ESR scores.
- •
More recent treatment studies confirm these findings, with a significant decrease in TNF-α levels.
- •
Improvement in signs and symptoms of RA occurred, regardless of whether the RA patients were under anti-TNF-α treatment.
- •
Anti-TNF-α RA treatment had no effect on the periodontal health without periodontal therapy.
Interestingly, a case report reported that periodontal treatment resulted in resolution of the RA disease, suggesting that in this case, RA was actually caused by the periodontal infection, specifically by P gingivalis.
Gingivitis treatment alone: effect on rheumatoid arthritis manifestation
Among persons suffering from RA, even plaque control consisting of supragingival scaling and oral hygiene instruction decreased the DAS28 as well as serum levels of IgG to P gingivalis hemin binding protein 35 and citrulline.
In summary
There is evidence to suggest
- •
A 2-way relationship exists between periodontitis and RA
- •
Periodontal treatment leads to improved biochemical markers of RA
- •
Periodontal treatment may ameliorate the clinical manifestations of RA.
What can be said to patients:
- •
Periodontal treatment and home oral hygiene can help lessen the swelling and pain of arthritic joints (symptoms of RA) and may help prevent RA.
Respiratory Tract Diseases
Aspiration pneumonia
Aspiration pneumonia is an infection of the lungs that is caused by aspiration of oral contents into the larynx and continuing via trachea and the primary bronchi to the lungs. Aspiration pneumonia is common among frail elderly and persons with swallowing disorders. Individuals who for any reason are not able to maintain good oral hygiene are also at risk.
The term nosocomial pneumonia describes a lung infection that is caused by microbes, typically in the hospital or other health care institution, at least 48 to 72 hours after being admitted and is therefore also known as hospital-acquired pneumonia (HAP). Bacteria from dental plaque are associated with pneumonia. Patients with periodontitis have more than a 3-fold higher risk of having lower respiratory tract infection than those without periodontitis among hospital patients. Among nursing home residents, those without oral health care have 3.6 times higher mortality from pneumonia than those who were assigned oral hygiene care by nursing assistants.
Systematic reviews published in 2003, 2006, 2008, and 2013 explored whether oral health care interventions in frail older persons in nursing homes or at hospitals could prevent aspiration pneumonia, and concluded that
- •
Mechanical oral hygiene and local chemical disinfection with antiseptics or antibiotics prevent aspiration pneumonia.
- •
About 40% of pneumonia cases are preventable by improved oral hygiene.
- •
Oral health care lowers not only the risk for both development and progression of aspiration pneumonia but also the risk of death from the disease.
- •
About 10% of deaths caused by pneumonia might be prevented by oral interventions.
Ventilator-assisted pneumonia
Mechanical ventilation contributes to the risk for HAP, but a 2013 Cochrane Systematic Review concluded that effective oral health care is important for adult patients in intensive care, and the use of either mouth rinse or gel that contains chlorhexidine is associated with a 40% decrease in the risk of developing ventilator-assisted pneumonia. However, there is no evidence for a decrease in risk for death from pneumonia, duration of mechanical ventilation, or duration of intensive care. Furthermore, the review found no difference regarding prevention of ventilator-assisted pneumonia in adults whether using (a) chlorhexidine together with tooth brushing versus chlorhexidine alone or (b) povidone iodine mouthwash versus saline, although weak evidence favors the former. Finally, the Cochrane Review did not find sufficient evidence to determine if other mouth rinses like saline, triclosan, or water can decrease the development of ventilator-related pneumonia.
Chronic obstructive pulmonary disease
Chronic obstructive pulmonary disease (COPD) is strongly associated with poor periodontal status, frequency of professional dental care, and knowledge about oral health. In COPD, periodontal treatment is reported to improve lung capacity function and lessen the frequency of exacerbations. However, a small study with 30 participants did not find any effect of periodontal therapy on quality of life 4 weeks after treatment, nor was there any effect on the COPD. The study did demonstrate that power brushes could be used without any adverse events.
Chronic Kidney Disease
Individuals undergoing dialysis typically have very poor oral health, and for those with long-term hemodialysis, periodontitis is associated with an increased mortality. One study reported a higher periodontitis risk for older Japanese women with lower cystatin C-based estimated glomerular filtration rate (eGFR).
Periodontal treatment: effect on chronic kidney disease
Some evidence indicates that periodontal treatment can reduce chronic systemic inflammation (measured as CRP level) and improve nutritional status in patients who receive both hemodialysis and peritoneal dialysis.
Periodontal treatment reduces the concentration of IL-18, a pro-inflammatory cytokine that is shown to be significantly elevated in persons with type 2 diabetes in whom IL-18 also may be a predictor for both development and progression of diabetic nephropathy.
A systematic review concluded that “There is quite consistent evidence to support the positive association between periodontitis and CKD [chronic kidney disease], as well as the positive effect of PT [periodontal treatment] on eGFR.” However, a critical review of this paper concluded that “periodontitis may be associated with chronic kidney disease, but current evidence is insufficient.” Specifically, the reviewer considered the inclusion of only 3, nonrandomized treatment studies with ambiguous results inadequate to support the last part of the authors’ statement.
In summary
There is some evidence to suggest that periodontal treatment
- •
Can reduce systemic inflammation in persons receiving hemodialysis or peritoneal hemodialysis
- •
Can decrease systemic inflammation (importantly, IL-18 levels) that may predict incidence and progression of nephropathy in people with type 2 diabetes
- •
Can improve nutritional status in persons receiving hemodialysis or peritoneal hemodialysis
What can be said to patients:
- •
Periodontal disease treatment and home oral hygiene might help people with kidney disease, but there is not enough proof for that yet.
- •
It is important to keep the mouth as clean as possible also in persons with long-term kidney disease who typically have poorer oral health habits than persons without kidney disease.
Cancer
The notion that infection/inflammation plays a potentially pivotal role in development of cancer has recently gained support, as it is realized that cancer is not only due to genes and their mutations, obesity, or pollution. For instance, a 24-year follow-up study of 1390 Swedes showed that a history of chronic dental infections due to caries or periodontitis that had caused tooth loss was strongly linked to the development of cancer.
Specifically related to periodontitis is the role that lately has been demonstrated by F nucleatum , a commensal member of the periodontal microbiome that is predominant in chronic periodontitis and which can turn into a pathogen that travels to locations outside the oral cavity. F nucleatum is not only found to invade colorectal cancerous lesions, but a causal role is established. Other members of the periodontal microbiome also invade oral and pharyngeal cancers and may contribute to their development.
In summary
There is evidence for
- •
Various periodontal bacteria and virus as known risk factors for cancers of the digestive tract, including the oral cavity
- •
F. nucleatum contributing to the development of colorectal cancer
- •
Oral human papilloma virus infection or a history of periodontitis being associated with squamous cell carcinomas of the head and neck, with a stronger link in oropharyngeal cancer than in cancers of the mouth and larynx.
What can be said to patients:
- •
Normal bacteria in the dental plaque (biofilm) may contribute to the cause of cancer of the gut (colon/rectum), even more so when there also is periodontal disease.
- •
There is no proof that treatment of periodontal disease can prevent cancer from developing.
Other Diseases
Evidence is emerging that illuminates the understanding of the systemic effects of members of the “traveling oral microbiome.” There is also evidence for the association between periodontal disease and systemic diseases thought to be caused by the general inflammatory responses. There is emerging evidence for links between periodontal infection with its subsequent inflammation and systemic disease, such as
- •
Alzheimer disease/cognitive function decline
- •
Appendicitis
- •
Benign prostatic hyperplasia
- •
Erectile dysfunction even in large population-based studies
- •
Human T-lymphotropic virus type I associated myelopathy/tropical spastic paraparesis and adult T-cell leukemia
- •
Inflammatory bowel disease
- •
Lemierre syndrome
- •
Ruptured intracranial aneurysms, abdominal aortic aneurysms
- •
Spondyloarthritis.
However, no findings from intervention studies to support these reported associations are available at this time.
Obesity
Obesity has recently been declared a disease, but will not be described in any detail because studies have not attempted to show whether periodontal treatment has any direct effect on this condition. However, it should be borne in mind that the excess fat cells in all body organs and tissues cause a chronic, low-grade inflammation (see Fig. 1 ) that has been called metainflammation. It has the same negative consequences as any other inflammation and hence contributes to the total load of systemic inflammatory responses, which in turn affects all the inflammation-related diseases. Obesity, metabolic syndrome, type 2 diabetes, and CVD are part of a cardiovascular-metabolic dysfunction continuum that without sharp borders develop together. Finally, obesity is shown to be a predictor for poorer outcome of periodontal treatment.
Pregnancy
Even though pregnancy is not a disease, but a temporary condition, it should be mentioned that there is some evidence for associations between periodontal infection and adverse pregnancy outcomes, especially pre-eclampsia (hypertension and protein in the urine or organ damage), as demonstrated by 3 meta-analyses in 2013 and 2014. Although cautioning against the studies’ heterogeneity, they all conclude that mothers with periodontitis have about 2 to 4 times higher risk for pre-eclampsia than those with healthy periodontal tissues and suggest that periodontitis may be regarded as a possible, independent risk factor for pre-eclampsia. Moreover, identical bacteria have been identified in the mother’s subgingival plaque and in the stillborn fetus and were declared the cause of such negative outcome.
A multitude of smaller studies report that periodontal treatment leads to a lower incidence of various adverse pregnancy outcomes, most often preterm birth and babies born too small for their gestational age. However, there is no evidence from large, well-designed and well-executed RCTs that nonsurgical periodontal treatment can prevent adverse pregnancy outcomes. Importantly, all studies concur that periodontal treatment during pregnancy is safe for both mother and child.
Why It Is so Difficult to Determine Whether Periodontal Treatment Has Systemic Effects
Clinical research findings of high quality are trickling in, but here follow some reasons it is difficult to unambiguously determine whether periodontal therapy improves general health:
- •
The systemic diseases are typically chronic and require many years to develop.
- •
Periodontal disease and many chronic systemic diseases are multifactorial with a multitude of causes and modifying internal and external factors.
- •
Some individuals seem to cope well (exhibit resistance) with the frequent, recurrent, or constant microbial offense, whereas others are more susceptible to periodontal breakdown. That is, the amount of damage caused seems to depend more on the host than on the amount and composition of the offending plaque.
- •
There has never been one globally accepted case definition for periodontitis, which prevents direct comparison of results from different studies.
- •
A therapeutic effect could be caused by something other than the periodontal treatment provided—or be overshadowed by other factors.
- •
Most people with a given disease suffer from more than one disease or condition, complicating both study participant enrollment and statistical analyses of the data collected.
- •
Human studies are inherently costly to conduct.
- •
The current systems to determine whether a relationship between 2 factors is one of cause and effect (causal, so one leads to the other) versus association (“vary together” without knowing whether one factor causes the other) were developed for infectious diseases that had mostly one major cause.
- •
As yet unknown genetic and environmental factors cannot be controlled, but may impact the end results.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


