Introduction
Childhood oral breathing can alter muscular balance and lead to facial deformities. No articles in the literature have reported on the alteration of facial growth patterns in patients who have received tracheotomies. The purpose of this study was to evaluate craniofacial developmental consequences originating from variations in breathing mechanisms in children who are nasal breathers or oral breathers, and those who have been tracheotomized.
Methods
The sample was divided into 3 groups of 10 each. The nasal group had a mean age of 13.9 years, the oral group had a mean age of 12.7 years, and the tracheotomy group had a mean age of 12.8 years. The masseter and suprahyoid muscles were evaluated with electromyography. The following measurements were made: facial, maxillary, and mandibular widths; nasion-sella-gnathion angle; and facial index.
Results
The tracheotomized group was similar to the nasal group for greater activity of the masseter muscles than of the suprahyoid muscles during mastication, as well as in the measurements of facial, maxillary, and mandibular widths. The oral group showed reductions in each category. The tracheotomized group was similar to the oral group during maximum dental occlusion for significantly higher activity of the suprahyoid muscles compared with the masseter muscles, with reductions in vertical values.
Conclusions
A childhood tracheotomy might affect facial development in a way comparable with that of oral breathers, including abnormal facial growth variations.
Irregular or inadequate breathing patterns can occur during childhood, and variable facial growth consequences can result. The effect of the breathing pattern on craniofacial growth has been widely debated and controversial for several decades. The concept of the functional matrix presented a logical reason for the findings in patients with nasal obstruction by ensuring that bone remodeling occurs as a response to the functions of facial muscles and other soft tissues. This presumably explained the narrow palate and the long face observed in patients who are chronic oral breathers. Because of oral breathing, the tongue goes to a lower position in the oral cavity, and the force balance of the facial muscles and the tongue becomes changed compared with nasal-breathing children.
The standard breathing pattern is apparently nasal, and oral breathing is presumed to be an adaptive resource or a habit acquired after birth, adapting to changes in nasal pathways. Experiments with primates by Harvold et al and Tomer and Harvold had the objective of associating orofacial muscular activity with facial morphogenesis. The animals were induced toward oral breathing by total obstruction of their nostrils with silicone plugs; this resulted in an adaptive strategy for oral breathing. Responding to the new functional pattern, all animals developed some sort of malocclusion, with subjective variations among them. This was apparently an answer to nasal obstruction and resulted in different morphofunctional consequences. The authors concluded that the increase in chronic activity of some muscular groups and the postural change of the mandible might cause alterations in muscle development and bone remodeling.
Nevertheless, because of the complexity of the cause-and-effect association, authors such as McNamara, Trotman et al, and Vig pointed out that craniofacial growth modifications derived from oral breathing should be analyzed more critically. There is a need for determining more basic and objective criteria when defining oral breathing as an etiologic growth-altering factor because interactions between form and function should explain any associations between oral breathing and growth, which seems to be multifactorial.
Until now, studies were lacking in the literature concerning the morphologic and functional facial developmental patterns in patients who had a tracheotomy during the facial growth period. Experiments with animals based on eliminating nasal breathing with a tracheotomy attempted to determine what facial growth changes occur when the breathing stimulus from nasal respiratory pathways was lacking. When breathing only with the lower respiratory pathways through the trachea, the animals could remain with their lips shut and thus free from the muscular imbalances frequently associated with oral breathing. A shorter mandibular length, smaller nasal width, reduced basicranial length and facial height, smaller palatine depth, and an increased palatal plane angle were verified. Nevertheless, these experiments did not consider that the animals could develop new functional adaptations, caused by the tracheotomy, to maintain their vital functions. The morphologic variations that were observed in dogs during their growth might be related to the possibility that new functional growth adaptations occur because of the tracheotomy. A recent study warned that changes in the normal nasal-breathing pattern can profoundly affect the development of the craniofacial skeleton in both humans and experimental animals.
Tracheotomized children do not have the breathing stimulus from nasal pathways or the facial muscular imbalances frequently associated with oral breathing. It is generally accepted that there is a relationship between the form and function of the craniofacial skeleton. The behavior of the muscles of the face seems to suffer functional variations according to different breathing patterns; therefore, the responses of craniofacial growth might also be variable. Surface electromyography (EMG), first used by Moyers, is a reliable method generally used for the quantitative analysis of muscular function, and it is considered an excellent and appropriate test to determine the effects of variable muscular activity on the functional matrix that is directly affecting facial growth. The quantitative evaluation of the relationship between morphology and function is useful for adequate understanding of a growing face. Thus, it seems important to study the behavior of the facial muscles relating to developmental adaptations in different breathing patterns on facial development. We evaluated craniofacial consequences originating from variations in childhood breathing mechanisms of patients having predominantly nasal breathing or predominantly oral breathing, or those who had a tracheotomy.
Material and methods
The sample consisted of 30 patients divided into 3 groups. The project was approved by the ethics committee for research of the Federal University of São Paulo in Brazil. Group 1 had 10 patients (7 boys, 3 girls) with a mean age of 13.9 years (SD, 2.6 years) with clinically observed predominantly nasal breathing. Group 2 had 10 patients (3 boys, 7 girls) with a mean age of 12.7 years (SD, 1.7 years) having predominantly oral breathing. The oral-breathing pattern diagnoses were made by clinical and nasofibroscopic examinations by an otolaryngologist (N.G.D.B.), showing at least 1 of these affections: allergic rhinitis, obstructive hypertrophy of the palatine and/or pharyngeal tonsils, and nasal septum deviation. Group 3 had 10 patients (7 boys, 3 girls) with a tracheotomy performed at an average age of 8 years 9 months; their mean age was 12.8 years (SD, 2.5 years), and they had no neurologic or congenital alterations, genetic syndromes, or craniofacial malformations. No orthodontic or orthopedic procedures were performed before this study was completed.
The functional analysis was determined by EMG of the masseter and suprahyoid muscles. This was performed at the Larynx and Voice Service at the Federal University of São Paulo in Brazil, by using the EMG unit measuring system (model MEB 9200/9300, Nihon Kohden, Tokyo, Japan). Surface bipolar disposable electrodes were used, with a 20 × 25 mm button connection, silver/silver chloride with clear auto-adhesive hydrogel (Spes Medica, S.r.l., Battipaglia, Italy) with an interelectrode distance of 10 mm. The electromyographic activities were registered at 200 μV, with and 8-second recording for each test. The electromyographic wave signals were processed by EMG analysis software (version 1.01; EMG System, São José dos Campos, Brazil). The calculation of the root mean square of the electrophysiologic potentials was provided by EMG analysis, which also provided the graphs for the acquisition of the electromyographic signals generated by each muscle.
The subjects, sitting upright in a dental chair, with the head supported and the Frankfort plane parallel to the floor, had their electromyographic activity recorded. Electrodes were applied by the technician researcher (S.F.B.) on the skin above the bilateral bodies of the masseter and suprahyoid muscles. To determine the masseters’ electrode area, the patients were asked to achieve their maximum intercuspal force. The greatest muscular area was palpated and selected, taking care that the contralateral electrodes were symmetrically positioned. To determine the suprahyoid electrode area, the patients were asked to make movements with the tongue against the hard palate, and the greatest muscular area was palpated and selected. Care was taken to use the same positioning of the contralateral electrodes. EMG activities at different functions of the stomatognathic system were performed: habitual mastication, in which the patients chewed 3 raisins as usual on the side of their spontaneous choice; and maximum dental occlusion, the maximum voluntary clenching in the intercuspal position.
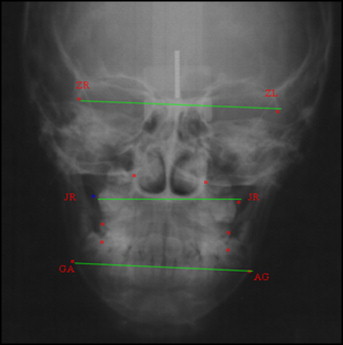
The morphologic analysis was determined with cephalometry in the frontal view, and linear transverse measurements were made for facial width (ZL-ZR), maxillary width (JL-JR), and mandibular width (A-G/G-A), as shown in the Figure . In the lateral view, the measurements were N-S-Gn (angle between sella, nasion, and gnathion), S-N/Go-Me (angle between the anterior cranial base and the mandibular plane), N-Me (linear measurement from nasion to menton), S-Go (linear measurement from sella to cephalometric gonion), and S-Go/N-Me (facial index).
Statistical analysis
The Kolmogorov-Smirnov test was applied to verify the symmetry of the sample, which did not follow the Gauss curve. The nonparametric Kruskal-Wallis test was used for independent data. When differences were found, identification was made by applying the Tukey multiple comparisons test. Comparisons between the masseter and suprahyoid groups during mastication were performed with Wilcoxon tests. The percentages of the relationship between the groups were calculated. The significance level for these tests was set at 5%.
Results
The Kruskal-Wallis test showed no significant differences in the EMG values for the right and left masseter muscles, mastication, or maximum dental occlusion for the 3 groups ( Table I ). Differences were identified for the maximum dental occlusion EMG values of the left and right suprahyoid groups. The Tukey multiple comparison test verified that the oral group was like the tracheotomized group, although the nasal group showed less electrical activity in the left and right suprahyoid muscles compared with the other groups during dental occlusion ( Table II ).
| Nasal (n = 10) | Oral (n = 10) | Tracheo (n = 10) | Kruskal-Wallis (P) | Results | |
|---|---|---|---|---|---|
| Masseter mastication | |||||
| Right | |||||
| Mean | 130.87 | 76.33 | 163.13 | 0.323 | Nasal = oral = tracheo |
| SD | 119.02 | 81.85 | 140.21 | ||
| Left | |||||
| Mean | 134.69 | 60.71 | 114.5 | 0.17 | Nasal = oral = tracheo |
| SD | 174.75 | 66.12 | 87.96 | ||
| Masseter occlusion | |||||
| Right | |||||
| Mean | 281.16 | 99.61 | 144.24 | 0.613 | Nasal = oral = tracheo |
| SD | 314.34 | 61.39 | 158.32 | ||
| Left | |||||
| Mean | 344.08 | 93.69 | 162.95 | 0.412 | Nasal = oral = tracheo |
| SD | 383.25 | 54.93 | 160.75 | ||
| Nasal (n = 10) | Oral (n = 10) | Tracheo (n = 10) | Kruskal-Wallis (P) | Results | |
|---|---|---|---|---|---|
| Suprahyoid habitual mastication | |||||
| Right | |||||
| Mean | 108.64 | 111.86 | 151.33 | 0.648 | Nasal = oral = tracheo |
| SD | 106.05 | 131.87 | 145.27 | ||
| Left | |||||
| Mean | 136.17 | 100.38 | 181.06 | 0.468 | Nasal = oral = tracheo |
| SD | 129.71 | 113.19 | 171.22 | ||
| Suprahyoid maximum occlusion | |||||
| Right | |||||
| Mean | 12.2 | 48.76 | 55.38 | 0.049 ∗ | Nasal < oral = tracheo (Tukey) |
| SD | 5.04 | 59.41 | 65.57 | ||
| Left | |||||
| Mean | 12.41 | 38.12 | 61.55 | 0.034 ∗ | Nasal < tracheo = oral (Tukey) |
| SD | 4.01 | 43.87 | 75.29 | ||
Comparisons were made between the masseter and suprahyoid groups during mastication by using Wilcoxon tests, and there was a significant difference in the oral group, with higher values for electrical activity in the suprahyoid group. Table III shows the means and standard deviations for the root mean square values of the masseter and suprahyoid groups on habitual mastication.
| Muscle group | Wilcoxon test (P) | ||
|---|---|---|---|
| Masseter | Suprahyoid | ||
| Nasal (n =10) | |||
| Mean | 132.78 | 122.40 | 0.575 |
| SD | 128.33 | 116.63 | |
| Oral (n =10) | |||
| Mean | 68.52 | 106.12 | 0.022 ∗ |
| SD | 73.00 | 119.88 | |
| Tracheo (n =10) | |||
| Mean | 138.81 | 166.20 | 0.575 |
| SD | 110.29 | 151.88 | |
The Kruskal-Wallis test verified significant differences in maxillary, mandibular, and facial widths ( Table IV ). The Tukey test verified that the nasal group was similar to the tracheotomized group, although the oral group showed a 5% mean reduction compared with the nasal group and a 6% reduction compared with the tracheotomized group for maxillary, mandibular, and facial widths.



