Ameloblastoma is the most common benign tumor of the jaws, comprising approximately 10% of odontogenic tumors and 1% of all cysts and tumors. Ameloblastoma was first described by Malassez in 1885. Due to the historically high recurrence rates and potential for local destruction and locally uncontrollable disease, a thorough understanding of the pathogenesis and clinical behavior of ameloblastoma is imperative for the oral and maxillofacial surgeon. The treatment is based largely on the tumor biology, location, and extent of disease. Surgical management should primarily be focused on a curative intent with the least degree of associated morbidity. Surgeons have frequently struggled with this issue, due to the lack of standardized treatment for ameloblastoma and the variety of histologic variants described in the literature. This chapter will review the pathogenesis, clinical presentation, histology, and treatment for the contemporary management of ameloblastoma.
Clinical Presentation/Pathogenesis
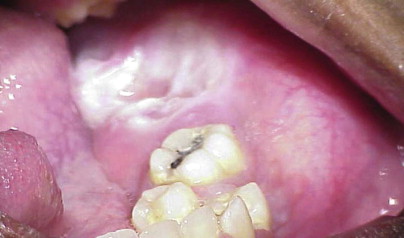
The ameloblastoma arises from odontogenic ectoderm. Malassez suggested that it arose from epithelial remnants of the developing root sheath. However, the causes of ameloblastoma are largely unknown. Ameloblastoma is classically described as a benign locally aggressive tumor, but it has the ability for metastasis in rare situations. Presumptive factors causing local inflammation, such as tooth extraction, caries, infection, calculus, developing teeth, trauma, nutritional deficiencies, and viruses, have been investigated; however, no direct causative correlation exists. Ameloblastoma is a slow-growing tumor that may take years before clinical presentation. The most common presenting symptom is painless swelling of the jaws ( Fig. 49-1 ). Other symptoms include pain, loosening of teeth, malocclusion, and altered sensation. Many tumors are associated with unerupted teeth, which are often displaced from the tumor. Root resorption of adjacent teeth is frequently seen. The majority of ameloblastomas, approximately 80%, arise in the mandible versus 20% in the maxilla. Most mandibular tumors arise in the third molar region. There are three classic varieties of ameloblastoma: solid/multicystic, unicystic, and peripheral. The solid/multicystic and unicystic types have several histologic variants that can have different biologic behavior. In a review of 3677 cases, the solid/multicystic type comprises 92% of ameloblastomas, the unicystic type 6%, and the peripheral type accounts for 2%.
Classification
Solid/Multicystic
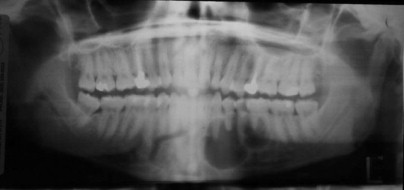
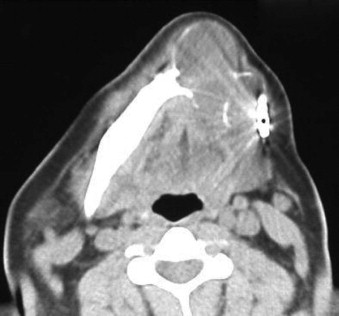
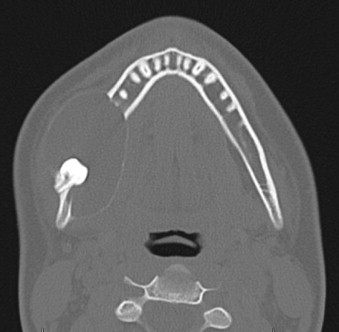
The solid/multicystic type is often termed the “conventional” ameloblastoma. It can be either a solid or multicystic lesion. It occurs equally in males and females. Commonly, it occurs in the third to seventh decade in life and is rarely seen in patients less than 20 years of age. The radiographic presentation is characterized as a multilocular radiolucency with a “soap bubble” or “honeycomb” appearance, although it may appear as a unilocular lesion ( Fig. 49-2 ). Cortical expansion, resorption of adjacent teeth roots, and association with an unerupted tooth are common ( Fig. 49-3 ). The radiographic margins appear irregular and scalloped, which may be seen with loss of the cortical bone margins. The histology of the solid/multicystic ameloblastoma is characterized by the presence of benign proliferation of odontogenic epithelium with stellate reticulum and varying composition of solid or cystic features. There are several variants of solid ameloblastoma from a histologic perspective that are important to recognize; however, these have limited impact on treatment decision making presently.
Follicular
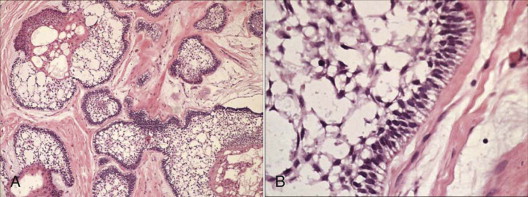
The follicular pattern is the most common subtype of the solid/multicystic ameloblastoma. The tumor comprises epithelial islands that resemble an enamel organ in a background of fibrous connective tissue stroma. The epithelial islands consist of a single outer layer of tall columnar or cuboidal cells with nuclei located on the opposite pole to the basement membrane (reverse polarization) and resemble ameloblasts, and there is a central zone of cells that resemble stellate reticulum. Areas of cyst formation are frequently observed; these areas vary from large macroscopic cystic spaces to microcysts that result from degeneration within epithelial islands. ( Fig. 49-4 ).
Plexiform
Plexiform ameloblastoma consists of larger sheets or long anastomosing cords of odontogenic epithelium. Similar to follicular ameloblastoma, the peripheral epithelial cells comprise ameloblast-like cells enclosing more loosely organized epithelial cells. Cyst formation is not common; however, if present, it is more likely due to degeneration of the connective tissue stroma ( Fig. 49-5 ).
Granular Cell
In some cases, the neoplastic epithelial cells may show transformation to granular cells with eosinophilic granules in the cytoplasm, which are consistent with lysosomes. The term granular cell ameloblastoma should be reserved for those cases that show extensive presence of granular cells. Although initially suggested as a degenerative change in long-standing ameloblastomas, granular cells have been reported in younger patients and in clinically aggressive tumors.
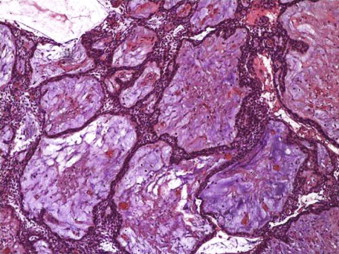
Basal Cell
The basal cell type contains nests of basaloid cells without central stellate reticulum-like cells. This is the least common type of solid/multicystic ameloblastoma ( Fig. 49-6 ).
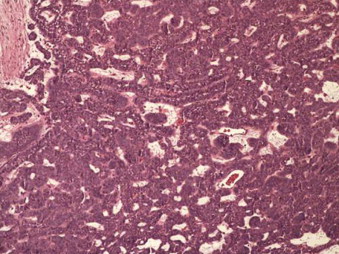
Acanthomatous
The term acanthomatous ameloblastoma is used for those cases that show extensive squamous metaplasia and keratin production occurring in the central epithelial islands of the follicular ameloblastoma. Caution must be used to not confuse this histologic appearance with squamous cell carcinoma or squamous odontogenic tumor ( Fig. 49-7 ).
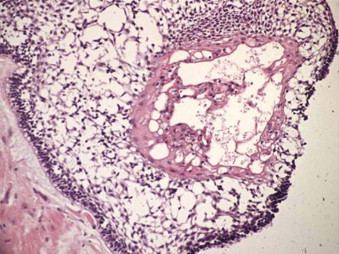
Desmoplastic
Desmoplastic ameloblastoma is characterized by extensive stromal collagenization (desmoplasia) with small islands and cords of tumor cells ( Fig. 49-8 ).
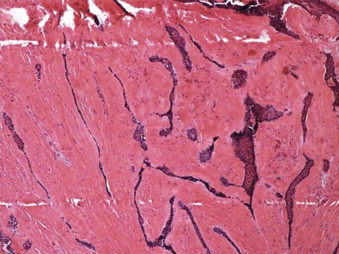
Although there are many histologic subtypes of ameloblastoma, there does not appear to be a significant difference in clinical behavior. However, Hong et al suggested that the follicular, granular cell, and acanthomatous subtypes showed a higher recurrence rate, whereas the plexiform and desmoplastic patterns showed a lower rate of recurrence. The desmoplastic type is the most recently identified form, first described by Eversole. The incidence ranges from 0.9% to 12.1% and has a predilection for the anterior maxilla relative to the other types of solid/multicystic ameloblastomas. Radiographic features are generally atypical of other ameloblastomas, being more characteristic of fibro-osseous lesions, including an indistinct radiographic border with radiolucent/radiopaque appearance and peripheral calcifications. The maxilla is often more frequently involved with greater than 50% of desmoplastic ameloblastomas. In a review of 80 cases of desmoplastic ameloblastoma by Beckley et al, the only reports of recurrence occurred when the tumor was treated with enucleation. Due to the atypical radiographic appearance of the desmoplastic ameloblastoma, many are treated with enucleation initially. Upon histologic diagnosis, it is imperative that the tumor is resected with clear margins, due to the high recurrence rate with enucleation. There is insufficient evidence in the literature for varying treatment algorithms based on the histologic subtype; therefore, all solid/multicystic ameloblastomas require similar treatment to limit recurrence.
Unicystic
The clinical presentation of unicystic ameloblastomas varies from the solid/multicystic types. Age at presentation is typically less than for other forms of ameloblastoma, typically in the second and third decade of life. The unicystic ameloblastoma appears radiographically, similar to the more common non-neoplastic dentigerous cyst ( Fig. 49-9 ). Findings such as association with a crown of an impacted tooth, resorption of tooth roots, and cortical expansion may be seen ( Fig. 49-10 ). In 1970, Vickers and Gorlin described three main histologic subtypes of unicystic ameloblastoma that have distinct clinical behavior: the luminal, intraluminal, and mural types. Any form of unicystic ameloblastoma may arise within the lining of a dentigerous cyst, therefore changing the diagnosis to a unicystic ameloblastoma. The lining of a unicystic ameloblastoma may not be uniform, with portions of merely a dentigerous cystic lining to extensions of ameloblastoma beyond the confines of the cyst wall. The unicystic variants can be difficult to histologically differentiate from one another, unless the entire lesion can be evaluated.
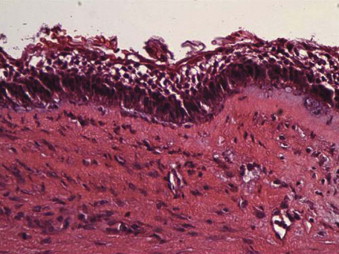
Luminal
With the luminal type of unicystic ameloblastoma, the tumor is restricted to the luminal surface of the cyst. It contains a dense fibrous cyst wall that encapsulates the entire cystic tumor component, which consists of a columnar or cuboidal basal layer resembling ameloblasts with overlying cells that resemble stellate reticulum ( Fig. 49-11 ).
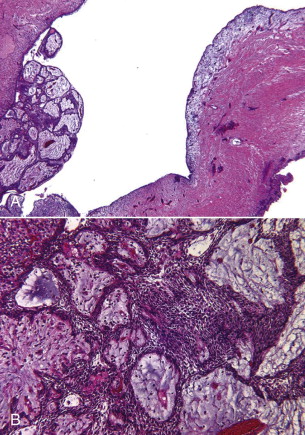
Intraluminal
The intraluminal type is histologically similar to the luminal type, except that one or more extensions or islands of ameloblastoma project into the lumen. These extensions range from small to large in filling the cystic space. In certain cases, the luminal tumor extensions resemble the conventional plexiform ameloblastoma. These are referred to as plexiform unicystic ameloblastoma ( Fig. 49-12 ).
Mural
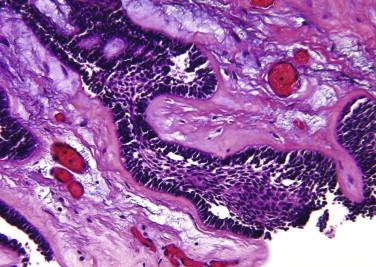
The mural type of unicystic ameloblastoma is characterized by infiltration of the tumor islands into the fibrous connective tissue wall of the cyst. The ameloblastic extension could resemble both follicular or plexiform types of solid/multicystic ameloblastomas. The extent of infiltration into the connective tissue varies significantly, and therefore it is necessary to obtain several sections through the specimen so that mural invasion will not be missed ( Fig. 49-13 ).
Peripheral
The peripheral ameloblastoma comprises 2% to 10% of ameloblastomas. The peripheral ameloblastoma is a tumor with the histologic characteristics of an intraosseous ameloblastoma, yet it occurs on the soft tissues covering the tooth-bearing aspect of the jaws. It arises from epithelial rests from the periodontal ligament. The peripheral ameloblastoma is characterized by a painless, firm, exophytic mass with a smooth surface without ulceration or induration. Its location is typically the attached gingiva or alveolar mucosa. The clinical presentation may be indistinguishable from pyogenic granuloma, peripheral ossifying fibroma, peripheral giant cell granuloma, or peripheral odontogenic fibroma. There is typically no radiographic evidence of bony involvement. Some lesions can cause superficial pressure erosion of alveolar bone without invasion, causing the “cupping” or “saucerization” defect of the bone. The mandible is the predominant site of occurrence, comprising approximately 70% of the lesions.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


