Introduction
Temporary paralysis of the masseter muscle caused by botulinum toxin is a common treatment for temporomandibular disorders, bruxism, and muscle hypertrophy. Loss of masseter force is associated with decreased mandibular mineral density. Our objectives were (1) to establish whether bone loss at the mandibular condyle is regionally specific and (2) to ascertain whether the treatment affects the condylar cartilage.
Methods
Young adult female rabbits received a unilateral masseter injection of botulinum neurotoxin serotype A (BoNT/A, n = 31), saline solution (n = 19), or no injection (n = 3) and were also injected with bromodeoxyuridine (BrdU), a replication marker. The rabbits were killed at 4 or 12 weeks after treatment. The condyles were processed for paraffin histology. Cortical thickness, cartilage thickness, and trabecular bone areal density were measured, and replicating cells were counted after BrdU reaction.
Results
The BoNT/A rabbits exhibited a high frequency of defects in the condylar bone surface, occurring equally on the injected and uninjected sides. Bone loss was seen only on the side of the BoNT/A injection. Cortical as well as trabecular bone was severely affected. The midcondylar region lost the most bone. Recovery at 12 weeks was insignificant. Condylar cartilage thickness showed no treatment effect but did increase with time. The numbers of proliferating cells were similar in the treatment groups, but the BoNT/A animals showed more side asymmetry associated with the condylar defects.
Conclusions
Bone loss may be a risk factor for the use of botulinum toxin in jaw muscles.
Highlights
- •
Botulinum toxin injection to the rabbit masseter caused extensive bone loss to the condyle.
- •
Condylar surface defects were seen on the treated and untreated sides.
- •
The condylar cartilage remained normal in thickness and proliferation rate.
Botulinum neurotoxin causes paralysis of neuromuscular junctions by blocking the release of acetylcholine. Of the 7 serotypes produced by Clostridium bacteria, botulinum neurotoxin type A (BoNT/A) is the most widely used by clinicians. Both striated muscle and smooth muscle respond to BoNT/A, and the approved applications range from erasing wrinkles to treating severe neurologic movement disorders. There are few parts of the body that have not become targets of BoNT/A.
Current off-label applications of BoNT/A include masseter muscle injection to reduce activity or to cause atrophy for cosmetic reasons. This therapy is often used for alleviating pain in patients with temporomandibular disorders and related conditions as well as for neuromuscular diseases. In addition, BoNT/A is a popular cosmetic treatment of benign masseter hypertrophy, despite the absence of controlled trials assessing the efficacy and safety of this procedure.
The paralysis of a major jaw adductor muscle may have unintended consequences for the temporomandibular joint (TMJ). Mechanical loading is important for bone maintenance; likewise, underloading is generally detrimental to bone health. BoNT/A treatment of the masseter reduces muscle force and has been used for that purpose after condylar fracture. However, loss of muscle force typically leads to rapid loss of bone mass in the mandible as well as in limb bones. Radiologically, the mandibular condyle is strongly affected by administration of BoNT/A to the masseter. Both a rabbit study and a pilot study on TMJ patients indicated severe loss of bone density compared with the controls. The rabbit study included microcomputed tomography scans of the condyles at 4 and 12 weeks after unilateral masseter injection. Central slices indicated that the injected-side condylar head had lost 40% of its bone area at 4 weeks relative to the uninjected side and the saline-solution controls; even at 12 weeks, it was 15% less than the control values. Our contribution is a histologic follow-up to that study and uses some of the same rabbit specimens to address questions that could not be answered from the microcomputed tomography scans.
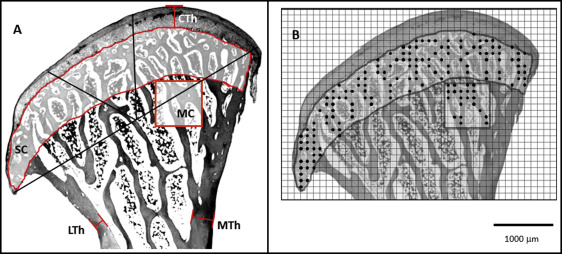
First, we sought to learn whether bone loss was specific to particular regions. Reasoning that the subchondral region receives loads directly from the overlying cartilage and transmits them to more distant condylar sites, we hypothesized that the subchondral trabeculae would show greater bone loss than more inferior trabeculae and the condylar cortex.
Second, we wanted to establish whether the condylar cartilage was affected as well as the bone. Cartilage is strongly affected by its mechanical environment; both understimulation and overstimulation can induce degenerative changes. Feeding rodents or rabbits a soft diet is claimed to thin the condylar cartilage, although the tissue remains healthy. One study with BoNT/A in the masseter muscles of young rats (apparently without incisor clipping) reported thinner cartilage in association with increased apoptosis. We expected that reduced loading of the TMJ caused by the BoNT/A masseter injection would be analogous to a soft diet, and thus there would be decreased proliferation and reduced thickness of the condylar cartilage after the BoNT/A injection of the masseter.
Material and methods
All procedures were approved by the Institutional Animal Care and Use Committee of the University of Washington. While under isoflurane anesthesia, 50 female New Zealand rabbits, 5 months old at acquisition, received unilateral injections of either 10 units of BoNT/A (n = 31; Botox; Allergan, Irvine, Calif) or an equivalent volume of 0.9% saline solution (n = 19) to the inferior portion of the superficial masseter muscle (split among 3 injection points ); the contralateral uninjected side served as an intra-animal control. Most of the animals were derived from the previous study. These animals were killed at 4 weeks (atrophy) or 12 weeks (recovery) after injection, therefore establishing 4 test groups: BoNT/A 4 week, BoNT/A 12 week, saline solution 4 week, and saline solution 12 week. Sample sizes for analysis ( Tables I-IV ) varied from 7 to 13 per group because of sectioning or staining errors. Three additional rabbits were untreated controls and were killed at 1 to 2 weeks after acquisition.
| Treatment/time | Affected ∗ rabbits/total rabbits | Injected side bony defect/total condyles | Uninjected side bony defect/total condyles | Sides combined bony defect/total condyles |
|---|---|---|---|---|
| BoNT/A, 4 wk | 6/13 (46%) | 5/13 (38%) | 5/13 (38%) | 10/26 (38%) |
| BoNT/A, 12 wk | 9/11 (82%) | 2/11 (18%) | 7/11 (64%) | 9/22 (41%) |
| Combined | 15/24 (63%) | 7/24 (29%) | 12/24 (50%) | 19/48 (40%) |
| Control, 0 wk | 2/3 (67%) | NA | NA | 3/6 (50%) |
| Saline, 4 wk | 1/7 (14%) | 0/7 (0%) | 1/7 (14%) | 1/14 (7%) |
| Saline, 12 wk | 2/9 (22%) | 1/9 (11%) | 1/9 (11%) | 2/18 (11%) |
| Combined | 5/19 (26%) | 1/16 (6%) | 2/16 (12%) | 6/38 (16%) |
| BoNT/A vs control + saline † | P = 0.03 | P = 0.11 | P = 0.02 | P = 0.02 |
∗ Affected condyles differed from normal in having a bony defect filled with cartilage and connective tissue and/or an irregular contour.
| Treatment/time | n | Injected side | Uninjected side | P , paired t test | n | Injected side | Uninjected side | P , paired t test |
|---|---|---|---|---|---|---|---|---|
| Subchondral trabecular bone areal density (%) | Midcondylar trabecular bone areal density (%) | |||||||
| BoNT/A, 4 wk | 10 | 52.1 (3.9) | 63.8 (5.7) | <0.01 | 10 | 26.0 (7.1) | 45.7 (14.2) | <0.01 |
| Saline, 4 wk | 7 | 64.4 (5.3) | 61.7 (5.8) | 0.44 | 7 | 41.2 (9.3) | 43.4 (12.4) | 0.59 |
| P , 2-sample t test | <0.001 | 0.58 | 0.01 | 0.62 | ||||
| BoNT/A, 12 wk | 10 | 55.5 (8.1) | 60.2 (9.9) | 0.11 | 10 | 28.5 (10.3) | 38.9 (15.1) | 0.01 |
| Saline, 12 wk | 8 | 61.7 (7.7) | 59.3 (8.7) | 0.44 | 8 | 35.6 (16.7) | 34.9 (16.9) | 0.85 |
| P , 2-sample t test | 0.07 | 0.86 | 0.22 | 0.68 | ||||
| Condylar neck cortical thickness (medial plus lateral, μm) | Condylar cartilage thickness (all layers combined, μm) | |||||||
| BoNT/A, 4 wk | 9 | 836.3 (132) | 1010.9 (216) | 0.06 | 10 | 234.1 (59) | 262.0 (70) | 0.37 |
| Saline, 4 wk | 6 | 1208.8 (248) | 1126.8 (132) | 0.46 | 5 | 219.6 (46) | 257.7 (30) | 0.32 |
| P , 2-sample t test | 0.03 | 0.20 | 0.69 | 0.75 | ||||
| BoNT/A, 12 wk | 9 | 926.1 (80) | 1099.2 (204) | 0.01 | 7 | 340.8 (88) | 315.4 (80) | 0.49 |
| Saline, 12 wk | 6 | 1010.3 (109) | 977.6 (200) | 0.78 | 7 | 331.7 (90) | 340.9 (82) | 0.87 |
| P , 2-sample t test | 0.09 | 0.26 | 0.81 | 0.55 | ||||
| Outcome variable | Mean difference | 95% Cl for difference | P value | Significance direction | |
|---|---|---|---|---|---|
| Lower | Upper | ||||
| Trabecular areal density: subchondral (%) | |||||
| Treatment | −9.6 | −14.0 | −5.3 | <0.001 | BoNT/A <saline |
| Time | −1.8 | −6.2 | 2.6 | 0.418 | – |
| Trabecular areal density: midcondylar (%) | |||||
| Treatment | −12.4 | −18.9 | −5.9 | <0.001 | BoNT/A <saline |
| Time | −2.5 | −9.1 | 4.1 | 0.451 | – |
| Cortical thickness (μm) | |||||
| Treatment | −226.7 | −353.4 | −100.1 | 0.001 | BoNT/A <saline |
| Time | 26.8 | 98.0 | 151.6 | 0.662 | – |
| Cartilage thickness (μm) | |||||
| Treatment | 14.4 | −44.7 | 73.5 | 0.620 | – |
| Time | −95.0 | −159.0 | −31.0 | 0.005 | 4 wk < 12 wk |
| Cell proliferation (number of BrdU + cells/condylar cartilage) | |||||
| Treatment | −27.2 | −71.2 | 16.8 | 0.218 | – |
| Time | 35.4 | −7.6 | 78.5 | 0.104 | – |
| Treatment/time | Injected side | Uninjected side | ||||
|---|---|---|---|---|---|---|
| n | Mean ± SD | Median (min-max) | n | Mean ± SD | Median (min-max) | |
| BoNT/A, 4 wk | 13 | 50 ± 39 | 34 (12-136) | 13 | 40 ± 47 | 18 (6-166) |
| Saline, 4 wk | 7 | 42 ± 20 | 40 (12-68) | 10 | 44 ± 27 | 30 (16-94) |
| BoNT/A, 12 wk | 11 | 58 ± 60 | 36 (11-200) | 11 | 100 ± 116 | 54 (4-419) |
| Saline/12 wk | 9 | 49 ± 69 | 33 (7-228) | 9 | 44 ± 44 | 37 (7-143) |
One week before they were killed, each animal was anesthetized and injected intravenously with bromodeoxyuridine (BrdU), a thymidine analog that labels cells in the S-phase of replication (40 mg/kg as a 10 mg/mL solution in phosphate-buffered saline solution). Anesthetized animals were killed by perfusion with 0.9% phosphate buffered saline solution followed by 4% paraformaldehyde. The mandibular condyles of each rabbit were removed from the mandibular ramus, decalcified in Immunocal (Decal Chemical, Tallman, NY), embedded in paraffin, and sectioned in the coronal plane at 7 to 10 μm. The observers (T.M. for condylar measurements; H.A.H.D. for the BrdU labeled cell counts and subjective obervations) were blinded to the treatment of the specimens.
For the condylar observations and morphometrics, 3 coronal sections through the widest portion of each condyle were stained with hematoxylin and eosin. Damaged sections were discarded, resulting in a few cases in which only 1 or 2 sections were used. Our original intent was to follow the standard staining and BrdU reaction with immunohistochemical stains for osteoblastic and osteoclastic markers. However, because of problems with decalcification and infiltration leading to the necessity of reembedding many specimens, the cellular contents of many marrow cavities (where osteoblastic and osteoclastic cells would have been found) were disconnected from bone surfaces; this caused distortion and in some cases missing marrow contents. Thus, we could not reliably identify either osteoblasts or osteoclasts in the marrow cavities. Instead, each section was qualitatively examined for signs of atypical morphology (unusual shape, surface defects, and so on) and resorption (multinucleated osteoclasts in Howship’s lacunae or scalloped surface). Then the section was aligned along the long axis of the condylar process and imaged at 10 times magnification (E400 microscope; Nikon, Tokyo, Japan) using MetaVue (Molecular Devices, Sunnyvale, Calif). Measurements were made with MetaMorph (Molecular Devices) and averaged for the 3 sections per condyle.
The measurements are illustrated in Figure 1 . The thickness of the condylar cartilage was measured at the most apical portion of the articular surface and included all zones (fibrous, proliferative, maturational, and hypertrophic), since separate measurements of each zone were not adequately repeatable. The percent of trabecular bone area (areal density) was assessed by superimposing a standardized grid over the chosen region and counting the numbers of intersections that fell on bone; this number was divided by the total number of grid intersections to yield a proportion. The 2 chosen regions were subchondral and midcondylar ( Fig 1 ). Cortical thickness was measured on the medial and lateral sides of the condylar neck perpendicular to the long axis of the condyle at a standard distance from the midcondylar region.
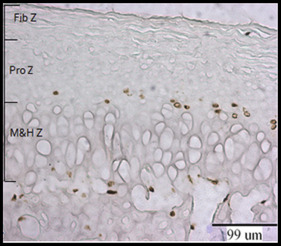
For cell replication, an additional 5 sections were chosen from the central region of each condyle; 1 slide was used as a negative control, and 4 were reacted for BrdU using a kit and following the manufacturer’s instructions (Becton Dickinson, Franklin Lakes, NJ). The sections were lightly counterstained with methyl green. A brown nucleus was the defining feature for a BrdU-positive (replicating) cell. The cartilage was divided into thirds (medial, central, lateral) as shown in Figure 1 . Three superficial-to-deep zones were identified for counting BrdU-labeled cells: dense fibrous connective tissue, designated the “fibrous zone”; small, flat cells underlying and parallel to the fibrous zone, designated “proliferative zone”; and an area grading from slightly larger ovoid cells to large spherical hypertrophic chondrocytes, designated “maturing and hypertrophic zone” ( Fig 2 ). However, the boundaries of these zones were not distinct. In addition to the condylar cartilage counts, positive cells in the periostea of the condylar poles and neck and in the lining of marrow cavities were noted. Counts from the 4 sections per condyle were averaged.

Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


