Ze’ev Seltzer
Jeffrey S. Mogil
As shown throughout this book, chronic pain syndromes affecting the craniofacial region constitute an unsolved clinical problem because they are common, cause incalculable suffering and incapacitation, and are difficult to treat by existing therapies. Available painkillers currently provide only partial pain relief that is compromised by side effects. A clinically driven subfield in pain genetics is the focus of this chapter since it seeks genetic factors that explain interindividual differences in the susceptibility to develop chronic orofacial pain in order to account for the variance and also to identify new drug targets. On this basis, this chapter introduces the reader to pain genetics, describes the rationale and some methodological considerations that underlie studies in pain genetics, and explains the background for the forecast that genetics may discover new treatment targets. For a broader perspective with greater detail, see Mogil.1
Why Is the Variance in Chronic Orofacial Pain Important?
Any number of outcomes may emerge when an anatomically complex region responds to injury, disease, or exposure to toxins. Therefore, it should come as no surprise that there are large inter-individual differences in chronic orofacial pain. Each patient presents with a unique combination of spontaneous pain, stimulus-evoked pain, and pain aggravated by movement. Individuals with the same pathology may present highly variable pain intensity, the location where pain is felt, duration and frequency of pain episodes, and affective-emotional impact of chronic orofacial pain on quality of life and daily activities; even the description of what the pain feels like (eg, burning, crushing) may be different, as well as the analgesic efficacy derived from painkillers (for examples, see Aubrun et al2).
Figure 9-1 provides an example of the variability in chronic pain intensity presented by 228 traumatic leg amputees. After controlling for confounding variables such as height of the amputation, years since amputation, and ethnicity, enormous differences in phantom leg pain levels are apparent. While about 25% of the amputees never suffered from pain in the missing leg, approximately 75% reported having pain episodes occurring at relatively regular times, with the same episode duration and typical intensity within individuals. A few amputees only had very faint pain, a few others suffered from maximal pain, and the rest presented with pain ranging between these extremes. Note that in this example, the majority of amputees reported phantom pain. This is an exception; in most cases the percentage of individuals developing chronic pain after some insult is lower, as Fig 9-2 suggests.
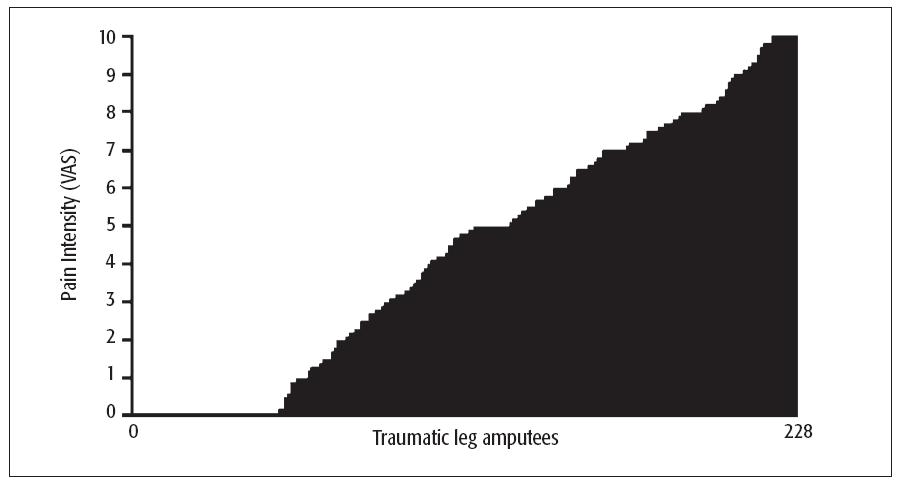
Fig 9-1 The typical intensity of phantom leg pain as reported by 228 leg amputees using a visual analog scale (VAS), where 0 represents “No pain” and 10 represents “The most intense pain imaginable.” (Seltzer, unpublished data).
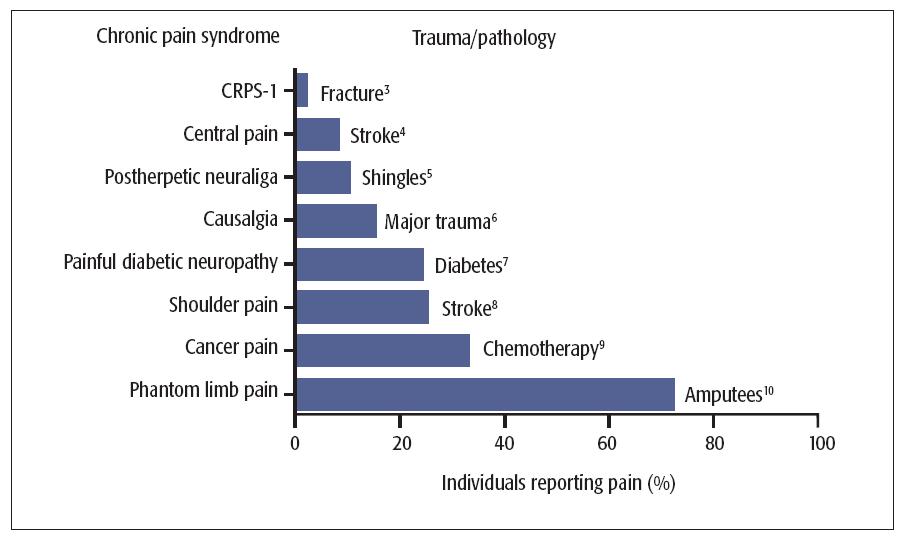
Fig 9-2 Percentage of individuals experiencing the traumas or pathologies listed on the right who developed the chronic pain syndromes on the left. CRPS-I = complex regional pain syndrome, type I.
Is Chronic Orofacial Pain a Heritable Trait?
The enormous variance in attributes describing chronic pain and the excess of females presenting with chronic orofacial pain (see chapters 1 and 13) suggest that the susceptibility to develop such pain may be heritable, that is, passed on from one generation to the next via genetic mechanisms. The following equation shows that, like any biologic trait, the phenotypic variance of chronic orofacial pain (VARP) is produced by genetic (VARG) and environmental or nongenetic sources (VARE), and their interaction (VARI):
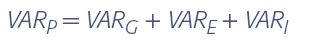
A phenotype is any measurable or categorizable trait that describes some feature of an organism (eg, height), whereas a genotype refers to the genetic constitution of an individual within its species.
Studies on the incidence of trigeminal neuralgia and familial migraine in twin pairs and pedigrees demonstrate the involvement of genetic factors.11 A few reports have already identified chromosomal regions, and even a small number of specific genes and single mutations affecting migraines, headaches, and chronic orofacial pain,12,13 as well as chronic pain in body parts other than the head.14–16 Studies in animal models of painful neuropathies have established that levels of chronic pain are considerably heritable.17–20 Furthermore, manipulations of individual genes by overexpression, deletion (in transgenic “knock-out” experiments), or inhibition of expression using small interfering ribonucleic acid (siRNA) or antisense oligodeoxynucleotide injections have identified more than 240 rodent genes that have some demonstrated role in pain.21–23
What Is the Size of the Heritable Component in Chronic Orofacial Pain?
Estimating heritability in chronic orofacial pain is not simple because the only measurable variance is that of the pain (VARP). However, the use of sophisticated statistics to analyze data from the incidence of chronic pain in closely related individuals has enabled researchers to measure the genetic and environmental variance in several chronic pain syndromes, thereby estimating the broad-sense heritability (H2), a value that reflects all genetic contributions to a population’s phenotypic variance. This is depicted in the following equation:

The importance of estimating heritability in chronic orofacial pain is in the ability to forecast the extent to which pain could be treated if pharmacologically addressing the genetic component. Figure 9-3 shows data from studies that estimated the size of H2 in various chronic pain syndromes. On average, genetic factors account for approximately 40% of the variance in chronic pain levels, similar to the average value in animal models of painful neuropathies.24 This calculation carries an optimistic message for pain patients, because it suggests that drugs developed on the basis of genetic knowledge could provide meaningful pain relief.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses



