CHAPTER 8. Odontogenic tumours and tumour-like lesions of the jaws
Tumours or tumour-like swellings of the jaws can be odontogenic, derived from odontogenic tissues, or non-odontogenic (Box 8.1).
Box 8.1
Important causes of tumours (swellings) of the jaws
• Cysts, predominantly odontogenic cysts
• Odontogenic tumours
• Giant cell lesions
• Fibro-osseous lesions
• Primary (non-odontogenic) neoplasms of bone
• Metastatic neoplasms
Odontogenic tumours are the most common neoplasms of the jaws. There are many types and the majority are rare. Classifications are complex. A simplified version of the current World Health Organization classification is shown in Box 8.2 and the whole classification is given in Appendix 8.1 together with brief details of the rarer lesions. It should be noted that this is a classification of tumours, not only of neoplasms, and that it therefore includes lesions of differing types and behaviour.
Box 8.2
Important types of odontogenic tumours
Benign epithelial neoplasms
• Ameloblastoma and its variants
• Squamous odontogenic tumour
• Calcifying epithelial odontogenic tumour
• Adenomatoid odontogenic tumour
• Calcifying cystic odontogenic tumour (calcifying odontogenic cyst)
Benign mixed epithelial and connective tissue neoplasms
• Ameloblastic fibroma
Benign connective tissue neoplasms
• Odontogenic fibroma
• Odontogenic myxoma
• Cementoblastoma
Malignant epithelial neoplasms
• Odontogenic carcinomas
• Clear cell odontogenic carcinoma
Malignant connective tissue neoplasms
• Odontogenic sarcomas
Hamartomas
• Odontomas
Dysplasias
• Cemento-osseous dysplasia
AMELOBLASTOMAS → Summary p. 136
Several types of ameloblastoma are recognised. This section describes the commonest type, the solid or multicystic type. Unicystic ameloblastomas are dealt with in the next section and the very rare peripheral and desmoplastic ameloblastomas are included in Appendix 8.1
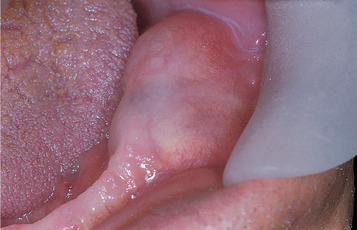
Ameloblastomas are the most common neoplasms of the jaws (Box 8.3). They are usually first recognised between the ages of 30 and 50 and are rare in children and old people. Eighty per cent form in the mandible; of these 70% develop in the posterior molar region and often involve the ramus. They are symptomless until the swelling becomes obtrusive (Fig. 8.1).
Box 8.3
Ameloblastoma: key features
• Neoplasm of odontogenic epithelium
• The most common odontogenic neoplasm
• Usually presents between ages 30 and 50
• Locally invasive but does not metastasise
• Typically asymptomatic and appears as a multilocular cyst radiographically
• Most commonly forms in posterior mandible
• Treated by excision with a margin of normal tissue
• Maxillary ameloblastomas can invade the cranial base and be lethal
 |
| Fig. 8.1
Ameloblastoma. Typical presentation. There is a rounded, bony swelling of the posterior alveolar bone, body and angle of the mandible. There is no ulceration, a feature only seen in very large tumours that have perforated the cortex.
|
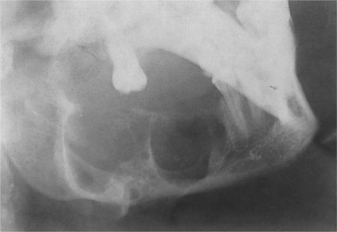
Radiographically, ameloblastomas typically form rounded, cyst-like, radiolucent areas with moderately well-defined margins and typically appear multilocular (Fig. 8.2) or with a honeycomb pattern. Lingual expansion may sometimes be seen, but is not pathognomonic of ameloblastoma. However, differentiation from non-neoplastic cysts and other tumours or tumour-like lesions of the jaws is not possible by radiography alone.
 |
| Fig. 8.2
Ameloblastoma. The typical radiological appearance of ameloblastoma is a multilocular cyst or multiple cyst cavities of different sizes, as shown here.
|
Genetic aspects
The causes of ameloblastomas are not known. However, there is evidence that various genes normally expressed during tooth formation are dysregulated in ameloblastoma. Deletion of an ameloblast adhesion molecule and enamel matrix protein called ameloblastin can cause odontogenic tumours similar to ameloblastoma in animals but is not yet linked to human ameloblastoma. Many other genes have been proposed to play a role in pathogenesis including heat shock proteins, inducible nitric oxide synthase and matrix metalloproteinases, but none is the likely primary cause.
Pathology
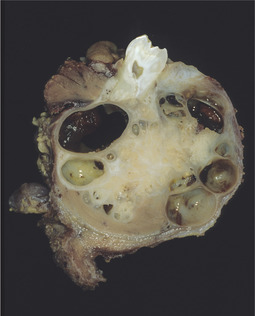
Conventional ameloblastomas are usually a mixture of solid neoplasm and cysts (Fig. 8.3), though either component may predominate. If only one cavity is present, the lesion is a unicystic amelobastoma (below).
 |
| Fig. 8.3
Ameloblastoma of the conventional solid/multicystic type in a resection specimen showing multiple cysts.
|
Solid and multicystic ameloblastomas may have a variety of histological patterns. All have the common feature of islands or strands of epithelium with a peripheral layer of pre-ameloblast-like cells that, at least focally, have nuclei at the opposite pole to the basement membrane (reversed polarity). The histological patterns have not been convincingly shown to affect the tumour’s behaviour and, apart from sometimes complicating microscopic diagnosis, they are of no significance.
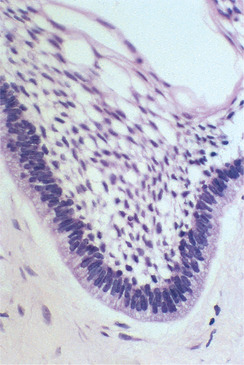
The follicular pattern is the most common and most readily recognisable type, with islands and trabeculae of epithelial cells in a connective tissue stroma (Fig. 8.4). The outer layer consists of a well-organised single layer of tall, columnar, ameloblast-like cells with reversed polarity, surrounding a core of loosely arranged polyhedral or angular cells, resembling stellate reticulum (Fig. 8.5). In the plexiform pattern the epithelium forms strands and trabeculae of small, darkly staining epithelial cells in a sparsely cellular connective tissue stroma (Fig. 8.6).
 |
| Fig. 8.4
Ameloblastoma. Islands of follicular ameloblastoma comprising ‘stellate reticulum’ and a peripheral layer of elongated ameloblast-like cells.
|
 |
| Fig. 8.5
Ameloblastoma. At high power in this follicular ameloblastoma the palisaded, elongate peripheral cells with reversed polarity are seen to be very similar in appearance to ameloblasts.
|
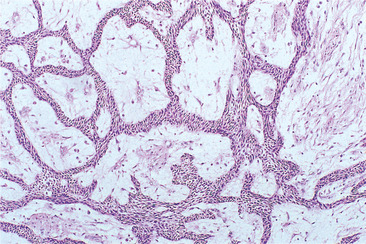 |
| Fig. 8.6
Ameloblastoma, plexiform type. There are thin, interlacing strands of epithelium but typical ameloblasts are not seen.
|
Cyst formation is common and there are usually several large cysts up to a few centimeters in diameter. Apparently solid ameloblastomas have numerous microscopic cysts. Cysts may develop either within the epithelial islands or result from cystic degeneration of the connective tissue stroma (Fig. 8.7).
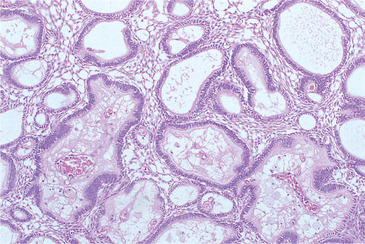 |
| Fig. 8.7
Ameloblastoma. Plexiform ameloblastoma composed of interconnecting strands of epithelium surrounding islands of connective tissue. Several of the stromal islands have degenerated to form small cystic cavities.
|
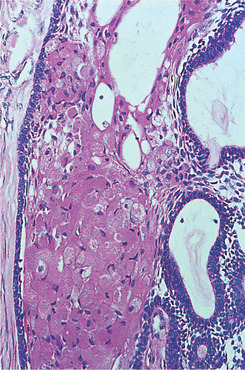
Other less common histological variants include the acanthomatous type with squamous metaplasia of the centre of epithelial islands (Fig. 8.8) producing prickle cells, and sometimes keratin, rather than a stellate reticulum-like tissue. The basal cell variant is rare and consists of more darkly staining cells predominantly in a trabecular pattern with little evidence of palisading at the periphery. They have been mistaken for basal cell carcinomas. The granular cell pattern is also rare. It resembles the follicular type, but the epithelium, particularly in the central areas of the tumour islands, forms sheets of large eosinophilic granular cells (Fig. 8.9).
 |
| Fig. 8.8
Squamous metaplasia in an ameloblastoma. Stellate reticulum-like cells have undergone squamous metaplasia to form keratin.
|
 |
| Fig. 8.9
Ameloblastoma. Granular cell change in an ameloblastoma. Ameloblastoma and stellate reticulum-like cells have undergone degenerative change to form large pink granular cells. In some tumours this change is extensive and the term ‘granular cell ameloblastoma’ is applied.
|
Although ameloblastoma is considered benign, a proportion have islands of tumour that extend into the surrounding medullary spaces. This growth pattern is reminiscent of invasion by a malignant tumour but extends only a few millimetres beyond the main bony cavity (Fig. 8.10).
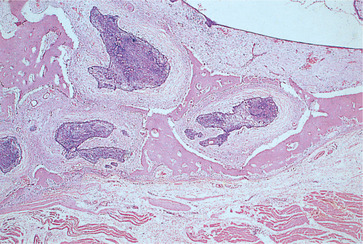 |
| Fig. 8.10
Ameloblastoma. Islands of ameloblastoma penetrating surrounding bone at the periphery of the lesion. Such bony infiltration demands that ameloblastoma is excised with a margin rather than curetted.
|
Additional types of ameloblastoma are listed in Appendix 8.1
Management
The diagnosis must be confirmed by biopsy. Treatment is by wide excision, preferably taking 1–2 cm of apparently normal bone around the margin. Complete excision of a large ameloblastoma may therefore require partial resection of the jaw, often with the condyle, and bone grafting. Smaller lesions may be excised leaving the lower border of the jaw intact and extending the resection subperiosteally. Bony repair then causes much of the jaw to re-form.
Excision with a bony margin ensures that any extension into surrounding bone is removed and guarantees a cure. Enucleation is usually followed by recurrence and should be reserved for unicystic ameloblastomas (below).
Regular radiographic follow-up is essential as any recurrence may not appear for several years. Limited re-operation can be performed if necessary. The patient must be warned of the necessity for regular follow-up and, possibly, of a further operation. Spread of ameloblastomas into the soft tissues is difficult to manage (Fig. 8.11).
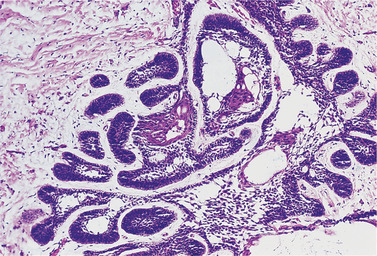 |
| Fig. 8.11
Ameloblastoma. Soft tissue spread after repeated inadequate excisions.
|
Maxillary ameloblastomas are particularly dangerous, partly because the bones are considerably thinner than those of the mandible and present weak barriers to spread. Maxillary ameloblastomas tend to form in the posterior segment and to grow upwards to invade the sinonasal passages, pterygomaxillary fossa, orbit and eventually the cranium and brain. They are thus occasionally lethal.
Key features of ameloblastomas are summarised in Box 8.3.
Unicystic ameloblastoma
The unicystic ameloblastoma is defined as an ameloblastoma that presents as a single cyst. The importance of differentiating this type is that enucleation carries a low risk of recurrence.
Unfortunately, making the diagnosis of unicystic ameloblastoma is difficult. An ameloblastoma with only one bony cavity radiographically can be a conventional solid or multicystic ameloblastoma. This will not become apparent until the lesion has been opened or examined histologically.
It is therefore important to bear in mind the various configurations of an ameloblastoma that might account for a unilocular presentation radiologically. These are shown in Figure 8.12.
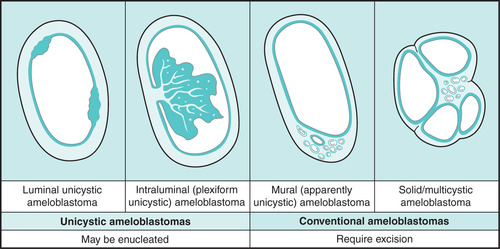 |
| Fig. 8.12
Explanations for a unicystic presentation of ameloblastoma radiologically. The two patterns on the left are true unicystic ameloblastomas while that on the right is a conventional ameloblastoma with one very large cyst. See the text for a further explanation of the significance.
|
The danger of making this diagnosis on radiological grounds alone is shown by the third diagram. Here a conventional ameloblastoma has developed one very large dominant cyst. However, there is a focus of solid/multicystic ameloblastoma in one area of the wall that might penetrate its full thickness or into surrounding bone. This is the mural type of (apparently unicystic) ameloblastoma. This is not a true unicystic ameloblastoma. It is a conventional ameloblastoma that could easily be misdiagnosed as one.
Unicystic ameloblastoma is diagnosed predominantly between the ages of 10 and 20, earlier than conventional ameloblastoma, and almost all arise in the posterior mandible. The single well-defined cavity resembles a dentigerous cyst in over 80% of cases (Fig. 8.13), but may also present as a radicular, residual or other types of cyst.
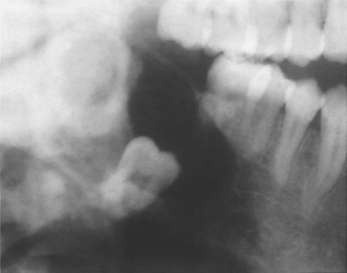 |
| Fig. 8.13
Unicystic ameloblastoma. This ameloblastoma forms a monolocular radiolucency enveloping the crown of an unerupted tooth. The radiological appearance mimics a dentigerous cyst, reinforcing the maxim that ameloblastoma should be considered in the differential diagnosis of every radiolucency at the angle of the mandible.
|
The histological appearances of all three true unicystic ameloblastomas are similar and are shown in Figure 8.14. The tumour cells forming the cyst wall are flattened and easily mistaken for those of a non-neoplastic cyst.
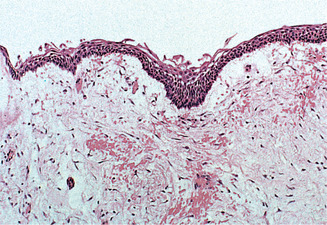 |
| Fig. 8.14
Unicystic ameloblastoma. Part of the lining of a large unicystic ameloblastoma. The epithelium is often stretched and loses its typical features, with only a few ameloblast-like basal cells.
|
Unicystic ameloblastomas may be enucleated because the epithelium is enclosed by the fibrous cyst wall. Nevertheless, a 10% recurrence rate after 10 years can be expected. In the mural type of unicystic ameloblastoma, there is a risk that ameloblastoma extends through the cyst wall. This would predispose to recurrence and re-excision of the bony cavity would have to be considered if histological examination showed penetration of the full thickness of the cyst wall.
The diagnosis of unicystic ameloblastoma can only be made confidently after removal because it requires detailed histological examination of the wall. The whole wall needs to be evaluated to define the extent and a single biopsy of a stretched cyst lining is insufficient for diagnosis. The treatment of apparently unilocular ameloblastomas by enucleation therefore has to be peformed before definitive diagnosis and it carries an element of risk. It would be very unfortunate if a conventional ameloblastoma containing one large cyst were enucleated in error as this would risk recurrence, perhaps in soft tissue, and a protracted course. In view of the dangerous sequelae of ameloblastoma in the maxilla, enucleation at that site would always be unwise.
Metastasising ameloblastoma and ameloblastic carcinoma
Both of these exceptionally rare tumours are little more than pathological curiosities – though not to the patient of course.
Metastasising ameloblastoma is a histologically typical ameloblastoma which, though apparently benign, nevertheless gives rise to distant lesions. The metastases are usually in the lung and they have the typical benign microscopic appearances of the primary. Some cases appear to have resulted from aspiration implantation at surgery and others follow surgical disruption at the primary site or repeated incomplete removal. Neither the primary tumour nor its ‘metastases’significantly differ histologically from conventional, non-metastasising ameloblastomas. It is therefore not possible to predict on the basis of morphology alone, whether an ameloblastoma will metastasise. The diagnosis of metastasising ameloblastoma can only be made after a metastasis has developed. However, the chances of it so doing are remote. As the ‘metastases’ are really benign, local excision of the secondary deposit should be curative.
Ameloblastic carcinoma is a tumour which initially resembles an ameloblastoma histologically, but loses differentiation and develops into a carcinoma. It spreads to lymph nodes or beyond. Later, the microscopic appearances become very poorly differentiated or similar to squamous cell carcinoma. Rarely, the progressive loss of differentiation can be seen in successive specimens from the same patient and confirms the diagnosis (Fig. 8.15). The treatment is that of an intraosseous carcinoma but the prognosis is poor if metastases are present.
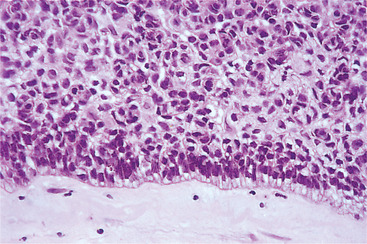 |
| Fig. 8.15
Ameloblastic carcinoma. Very rarely a malignant variant of the ameloblastoma is encountered. Histologically, they may be indistinguishable from other carcinomas but, in some cases, as here, a peripheral layer of palisaded ameloblast-like cells remains, indicating the tumour’s nature.
|
ADENOMATOID ODONTOGENIC TUMOUR → Summary p. 157
Adenomatoid odontogenic tumour is uncommon, completely benign and probably a hamartoma.
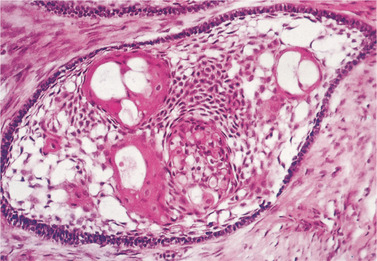
Clinically, adenomatoid odontogenic tumours are found either in late adolescence or young adulthood. Females are twice as frequently affected as males. The tumour is most frequently sited in the anterior maxilla and forms a very slow-growing swelling. Alternatively, it may be noticed by chance on a radiograph where it frequently presents as a dentigerous or radicular cyst (Fig. 8.16). Most specimens are only a few centimetres in diameter.
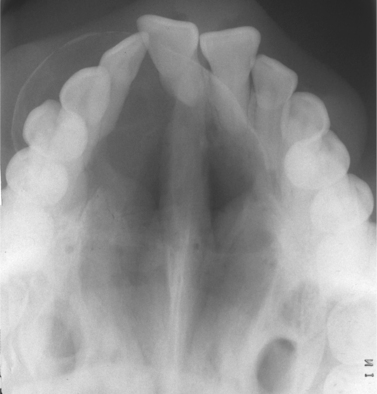 |
| Fig. 8.16
Adenomatoid odontogenic tumour, a typical cyst-like presentation in the anterior maxilla.
|
Pathology
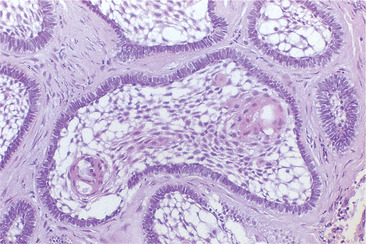
A well-defined capsule encloses whorls and strands of epithelium, among which are microcysts, resembling ducts cut in cross-section and lined by columnar cells similar to ameloblasts (Figs 8.17 and 8.18). These microcysts may contain homogeneous eosinophilic material. Microcysts have led to the tumours being called adenomatoid, but they are not ducts and are never seen cut longitudinally. Fragments of amorphous or crystalline calcification may also be seen among the sheets of epithelial cells.
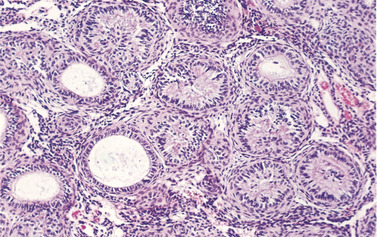 |
| Fig. 8.17
Adenomatoid odontogenic tumour. Low magnification shows duct-like microcysts and convoluted structures. The stroma is scanty.
|
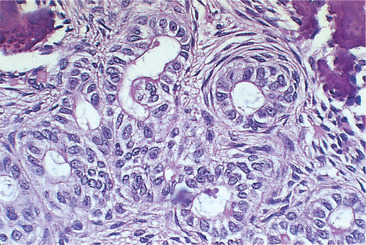 |
| Fig. 8.18
Adenomatoid odontogenic tumour. At higher power the duct-like spaces which give the tumour its name are seen.
|
These lesions shell out readily, enucleation is curative and recurrence is almost unknown.
Key features of adenomatoid odontogenic tumour are summarised in Box 8.4.
Box 8.4
Adenomatoid odontogenic tumour: key features
• Rare
• Hamartoma of odontogenic epithelium
• Usually presents between ages 15 and 20
• Most common in the anterior maxilla
• Often appears radiographically as a unilocular dentigerous cyst
• Responds to enucleation
CALCIFYING EPITHELIAL ODONTOGENIC TUMOUR → Summaries pp. 157, 190
This tumour, with bizarre microscopic features, is often termed a Pindborg tumour, after its discoverer. Though rare, it is important, because it has been mistaken for a poorly differentiated carcinoma microscopically.
Clinically, adults are mainly affected at an age of about 40 or more. The typical site is the posterior body of the mandible, which is twice as frequently involved as the maxilla. Symptoms are usually lacking until a swelling appears. Radiographs show a radiolucent area with poorly defined margins. Despite the name, a lesion may be completely radiolucent or develop increasing radiopacity as it matures.
Pathology
Calcifying epithelial odontogenic tumours consist of sheets or strands of epithelial cells in a connective tissue stroma (Fig. 8.19). The epithelial cells are polyhedral and typically have distinct outlines and intercellular bridges. Gross variation in nuclear size, including giant nuclei, is frequently striking (Fig. 8.20). These nuclei are usually hyperchromatic and, though mitoses are rare, produce an alarming resemblance to a poorly differentiated carcinoma. However, unlike most carcinomas, a stromal inflammatory reaction is typically absent. Within the tumour, there are typically homogeneous hyaline areas, with the staining characteristics of amyloid. These may calcify and form concentric rings in and around degenerating epithelial cells and may form large masses. Clear cells may also be present.
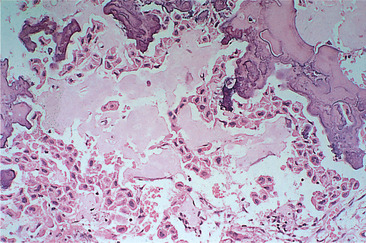 |
| Fig. 8.19
Calcifying epithelial odontogenic tumour (Pindborg tumour). The tumour is composed of strands and sheets of polyhedral epithelial cells lying in pale pink-staining amyloid-like material. Some of this material has mineralised, stains a darker blue colour and gives rise to radiopacities within the lesion.
|
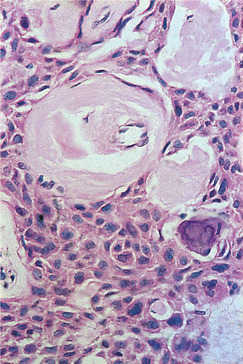 |
| Fig. 8.20
Calcifying epithelial odontogenic tumour. At higher power the epithelial cells are seen to have sharply defined cell membranes resembling squamous epithelium and pleomorphic hyperchromatic nuclei. The pale pink material is amyloid-like.
|
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


