CHAPTER 5. Gingivitis and periodontitis
THE NORMAL PERIODONTAL TISSUES
The gingival tissues comprise a series of anatomical regions (Box 5.1).
Box 5.1
Anatomical regions of normal healthy gingival tissue
• Junctional epithelium. Extends from the amelocemental junction to the floor of the gingival sulcus and forms the epithelial attachment to the tooth surface
• Sulcular epithelium. Lines the gingival sulcus and joins the epithelial attachment to the free gingiva
• Free gingiva. Coronal to the amelocemental junction and attached gingiva and includes the tips of the interdental papillae
• Attached gingiva. Extends apically from the free gingiva to the mucogingival junction and is bound down to the superficial periodontal fibres and periosteum.
Histologically, gingival epithelium is stratified squamous: the free and attached gingiva are parakeratinised. The epithelial attachment consists of flattened stratified squamous epithelium adhering to enamel (Fig. 5.1). The attachment apparatus consists of a basal lamina and hemidesmosomes. The attachment to the enamel appears histologically as a clear cuticle (Fig. 5.2). It forms an effective seal, protecting the underlying connective tissues. The epithelial attachment is so firmly adherent to the tooth surface that mechanical damage, which would otherwise pull the epithelium from the tooth, tears apart the epithelial cells (Fig. 5.2). This epithelium has a relatively high turnover rate and the epithelial attachment is probably actively maintained and reformed. This enables the attachment to migrate apically until it becomes attached to cementum when periodontitis develops.
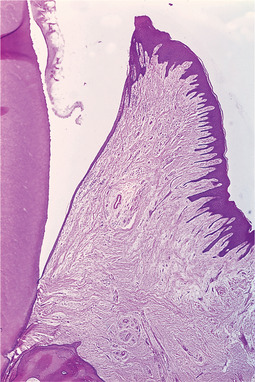 |
| Fig. 5.1
Gingival sulcus and epithelial attachment. This sagittal section of a specimen from a woman of 27 shows the normal appearances. Enamel removed by decalcification of the specimen has left the triangular space. The epithelial attachment forms a line from the top of the papilla to the amelocemental junction; its enamel surface, the actual line of attachment, is sharply defined. The gingival sulcus, minute in extent, is formed where the papilla curves away from the line of the enamel surface. There is hardly any inflammatory infiltrate.
|
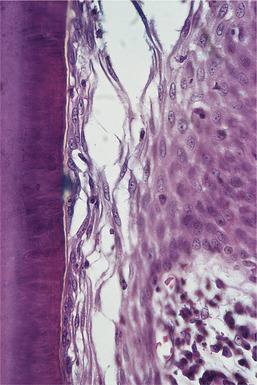 |
| Fig. 5.2
The strength of the epithelial attachment. On this tooth the epithelial attachment has migrated on to the surface of the cementum as a result of periodontal disease. The epithelium has been torn away from the tooth but the tear is within the junctional epithelium, leaving some of its cells still adherent to the cementum and attached by a clear cuticle. A similar strength is seen when the junctional epithelium is attached to enamel.
|
The attached gingiva is firmly bound down to the underlying bone to form a tough mucoperiosteum. Its stippled appearance is due to the intersections of its underlying epithelial ridges at the junction with the connective tissue.
The alveolar mucosa is continuous with the attached gingiva, but is sharply demarcated by its smooth surface, darker colour and increasing separation from the bone.
Gingival and periodontal fibres
The principal fibres of the periodontal ligament form fibrous bundles interspersed by loose connective tissue containing blood vessels and nerves. These components provide a viscoelastic supportive mechanism resisting both compressive and tensile forces on the teeth. The normal thickness of the periodontal ligament is about 0.1–0.3 mm.
The principal fibres are arranged in a series of fairly well-defined groups (Box 5.2).
Box 5.2
Principal fibres of the periodontal ligament
• Oblique fibres form a suspensory ligament from socket to root in coronal to apical directions
• Horizontal fibres form a dense group attaching neck of tooth to rim of socket
• Transeptal fibres of the horizontal group are not attached to alveolar bone but pass superficially to it and join adjacent teeth together. They protect the interdental gingiva by resisting forces that would otherwise separate the teeth and open the contact points
• Gingival fibres form a cuff round the neck of the tooth supporting the soft tissues. They resist separation of the gingivae from the tooth and help to prevent formation of pockets
The periodontal ligament fibres are embedded in cementum at their inner ends and in the lamina dura at their outer ends. New fibres replacing those which have aged, or forming in response to new functional stresses, are attached by apposition of further layers of cementum which becomes thicker with age. The lamina dura is a layer of compact bone continuous, at the mouths of the sockets, with the cortical bone of the jaw. The radiographic appearance of the lamina dura has limited diagnostic value in periodontics as its presence largely depends on the alignment of the beam.
Gingival crevicular fluid (exudate)
In health, a minute amount of fluid can be collected from the gingival margins. It differs in composition from saliva, which contains IgA, while gingival fluid has a variable but significant content of IgG, IgM and leucocytes. Unlike saliva, gingival fluid is not a secretion (there are no glands in this region), but is inflammatory exudate. It therefore increases greatly with the degree of inflammation.
NOMENCLATURE AND CLASSIFICATION OF PERIODONTAL DISEASE
The term periodontal disease usually refers only to plaque-related inflammatory disease of the dental supporting tissues. A wide variety of diseases of the oral mucosa can also affect the gingivae occasionally, so that conditions as diverse as tuberculosis or lichen planus can produce lesions in this area. Such conditions do not play any significant part in the development of periodontal disease in its commonly accepted sense.
Chronic gingivitis is, with few exceptions, the preliminary stage to the development of chronic periodontitis, but clinically no sharp dividing line can be drawn between chronic gingivitis and the onset of periodontitis. Nevertheless, the distinction is important in that chronic gingivitis can generally be cured by plaque control. By contrast, loss of bone and tooth support due to chronic periodontitis are largely irreversible.
The main clinical types of periodontal disease are grouped together on the basis of what seems to be the predominant pathological process (Box 5.3).
Box 5.3
Types of periodontal disease
I. Gingivitis and periodontitis
• Chronic gingivitis (common plaque-related type)
• Chronic adult periodontitis (common, plaque-related type)
II. Less common types of gingivitis
III. Uncommon types of periodontitis
• Prepubertal periodontitis
• Juvenile periodontitis
• Rapidly progressive periodontitis
• Acute leukaemic periodontal destruction
IV. Miscellaneous periodontal disorders
• Gingival hyperplasia
Familial
Drug-related
• Periodontal atrophy recession
• Periodontal (lateral) abscess
• Pericoronitis
CHRONIC GINGIVITIS
Clinical features
Chronic gingivitis is asymptomatic, low-grade inflammation of the gingivae. The latter become red and slightly swollen with oedema (Fig. 5.3). Plaque, deposited along the gingival margins, is readily detectable. In most patients, chronic gingivitis is due to local factors and, in particular, ineffective toothbrushing. It should be curable by effective oral hygiene.
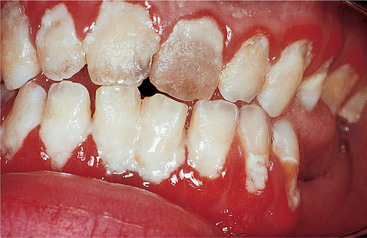 |
| Fig. 5.3
Gingivitis. A florid, bright-red band of gingival inflammation results from very poor oral hygiene. Thick accumulations of plaque are visible on all tooth surfaces.
|
Chronic hyperplastic gingivitis is a term sometimes given to chronic gingivitis in which inflammtory oedema is prominent. However, there is no soft tissue hyperplasia. Maintenance of strict oral hygiene brings resolution.
Systemic disorders can exacerbate chronic gingivitis but rarely play a significant role (Box 5.4).
Box 5.4
Factors contributing to or exacerbating chronic gingivitis
Local
• Poor tooth cleaning technique
• Dental irregularities providing stagnation areas
• Restorations or appliances causing stagnation areas
Systemic
• Pregnancy
• Down’s syndrome
• Poorly-controlled diabetes mellitus
Pathology
Gingivitis is an inflammatory response to plaque bacteria. By definition, inflammation is restricted to the gingival margins and does not affect the periodontal ligament or bone (Fig. 5.4).
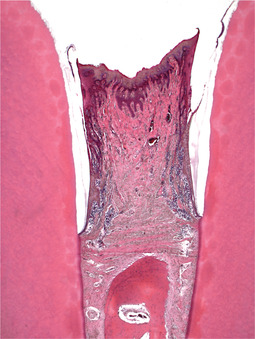 |
| Fig. 5.4
Chronic gingivitis. The epithelial attachment remains at the level of the amelocemental junction and inflammatory cells are concentrated below the junctional epithelium and extend into the deeper gingiva, around the interradicular fibres. The alveolar crest is not resorbed.
|
The development of gingivitis has been arbitrarily divided into three histological stages (Boxes 5.5 and 5.6), while the fourth stage refers to chronic periodontitis. It must be appreciated that these stages are artificially distinguished, being largely based on animal studies.
Box 5.5
‘Initial stage’ chronic gingivitis
• Develops within 24–48 hours of exposure to plaque
• Plaque related to gingival sulcus
• Vasodilatation
• Infiltration predominantly of neutrophils
• Leakage of exudate into gingival sulcus
Box 5.6
Established chronic gingivitis
• Dense, predominantly plasmacytic infiltrate
• Infiltrate fills but limited to interdental papillae
• Destruction of superficial connective tissue fibres
• Deepened gingival crevice
• Epithelial attachment remains at or near amelocemental junction
• Alveolar bone and periodontal ligament remain intact
Variations in plaque bacteria with stage of disease
Healthy (uninflamed) gingivae.
The plaque is supragingival and thin (10 to 20 cells thick). Gram-positive bacteria predominate and include Actinomyces species, Rothia, viridans streptococci and S. epidermidis. In elderly patients in periodontal health, Gram-positive bacteria, particularly streptococci, form the largest single group (50% of the predominant cultivable flora), while Gram-negative bacteria only account for 30%. The latter include porphyromonas and fusobacterium species.
Early (and experimental) gingivitis.
If toothbrushing is neglected for several days, plaque grows in thickness and is typically 100 to 300 cells thick. In the earliest stages, bacteria proliferate but the plaque remains Gram-positive in character and Actinomyces species become predominant.
Chronic gingivitis.
With the passage of time, persistence of plaque leads to chronic inflammation and Gram-negative organisms become increasingly prominent. Veillonella, Fusobacterium and Campylobacter species become conspicuous.
Calculus
Calculus is calcified plaque. The calcification is less significant than the bacterial content which induces inflammation and, later, tissue destruction. However, calculus distorts the gingival crevice and, by extending the stagnation area, probably promotes even greater bacterial proliferation (Fig. 5.5). Supragingival calculus mainly forms opposite the orifices of the major salivary glands in the lower incisor and upper first molar areas. It cannot be removed by the patient and provides a rough, plaque-retentive surface.
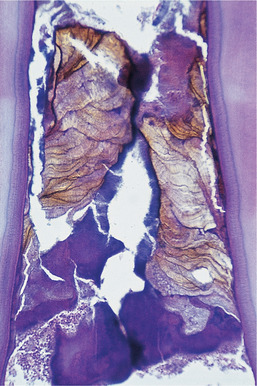 |
| Fig. 5.5
Subgingival calculus showing the layered structure resulting from incremental deposition. The calculus has a brown colour as a result of blood and bacterial pigment within it. A thick layer of plaque adheres to the deep surface.
|
Several compounds, such as pyrophosphates, added to dentifrices have been shown to reduce calculus formation to variable degrees. However, any clinical benefit has not been unequivocally established.
Management
Chronic gingivitis is readily recognisable from the clinical features already described. Gentle probing shows absence of pocketing and radiographs show intact crestal alveolar bone. The diagnosis is confirmed by resolution of gingivitis when effective oral hygiene measures (including calculus removal, effective toothbrushing and interdental cleaning habits) become established (Figs 5.6 and 5.7). Any exacerbating factors must be dealt with if possible.
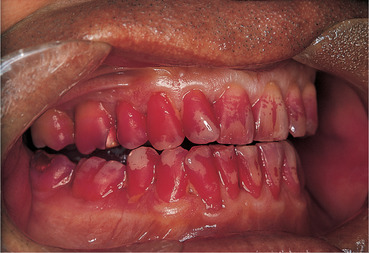 |
| Fig. 5.6
Accumulation of plaque stained after 24 hours by disclosing solution.
|
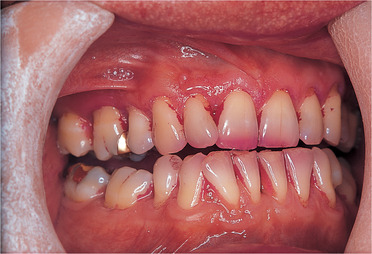 |
| Fig. 5.7
The effects of tooth brushing in the same patient. Plaque remains interdentally in most areas, explaining why gingivitis is often localised here.
|
Pregnancy gingivitis
Pre-existing gingivitis may become more severe in the first two months of pregnancy (see Fig. 31.9). Inflammatory erythema and oedema typically become more severe and a localised vascular lesion (a pregnancy tumour) may develop. The latter has the same structure as a pyogenic granuloma (Ch. 19).
Pregnancy gingivitis can be much ameliorated or abolished by a strict oral hygiene regimen and improves after parturition. However, if a pregnancy tumour persists, it should be excised.
Down’s syndrome
In Down’s syndrome, gingivitis is exacerbated by excessive plaque formation and difficulties in establishing effective toothbrushing habits. The multiple immunodeficiencies typical of this disorder contribute. Progress to periodontitis and early tooth loss are frequent consequences (see Figs 33.3 and 33.4).
Diabetes mellitus
In poorly-controlled diabetes, periodontal health may deteriorate sharply. In well-controlled diabetes, acceleration of gingivitis may not be noticeable. However, it appears that, overall, diabetics suffer somewhat more severe periodontal disease and slightly earlier tooth loss than healthy, control populations. The neutrophil defect typical of this disease is probably the main factor involved.
CHRONIC ADULT PERIODONTITIS
Clinical features
Chronic periodontitis (the ‘advanced lesion’) is the chief cause of tooth loss in later adult life, but symptoms are typically minimal. Many patients remain unaware of the disease until teeth become loose.
Gingivitis persists and patients may complain of gingival bleeding or an unpleasant taste. Later, gingival recession or loosening of the teeth may become conspicuous. Periodontitis is a potent and common cause of foul-smelling breath (halitosis). The congested gingival margins become purplish-red, flabby and swollen. The interdental papillae ‘split’ and can be separated from the teeth. Later, the papillae are destroyed and the gingival margin tends to become straight with a swollen, rounded edge.
Pressure on the gingival margins causes bleeding and sometimes pus can be expressed from round the necks of the teeth. Bacterial plaque and calculus are widespread.
Loss of attachment leads to pocketing which allows a probe to be passed down between teeth and gingiva. In pockets, the rough surface of subgingival calculus can be felt. Eventually, teeth become increasingly loose and dull to percussion.
Radiography
The earliest change is loss of definition and blunting of the tips of the alveolar crests. Bone resorption usually progresses in a regular manner and its level remains the same along a row of teeth. A straight line can be drawn along the crest of the alveolar bone (horizontal bone loss –Fig. 5.8). Complex patterns of bone loss may also be seen, probably due to the effects of variable degrees of loss of attachment superimposed upon the underlying anatomical features. Thus, in areas where bone is thin, destruction results in horizontal bone loss, but where alveolar bone is thicker, partial destruction gives rise to vertical or angular bone defects.
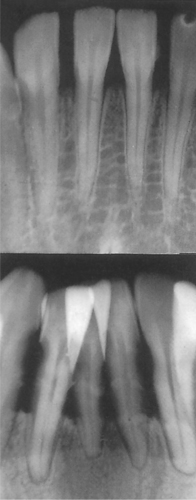 |
| Fig. 5.8
Early and severe horizontal bone loss in advanced chronic adult periodontitis.
|
Aetiology and pathology
Conditions promoting stagnation and maintenance of infection at the gingival margins have been discussed earlier. Persistence of infection at the gingival margins leads to progressive inflammation and destruction of the supporting tissues. Usually, local factors are most important in the progress of chronic periodontitis but, for reasons which are unclear, there is wide individual response.
The main features of the pathology of chronic periodontitis are summarised in Box 5.7.
Box 5.7
Pathological processes in chronic periodontitis
• Chronic inflammation
• Destruction of periodontal ligament fibres
• Resorption of alveolar bone
• Migration of the epithelial attachment towards the apex
• Formation of pockets around the teeth
• Formation of subgingival plaque and calculus
Plaque
Bacterial plaque, a dense mat of bacteria (Box 5.8), extends from the gingival margins into pockets.
Box 5.8
Important bacteria associated with periodontal disease
Subgingival calculus
Extension and calcification of plaque leads to formation of subgingival calculus within periodontal pockets. The deposits are thin, more widely distributed, harder, darker and more firmly attached than supragingival calculus. Calculus appears laminated histologically, with altered staining, probably due to breakdown products from blood cells oozing into the pockets. Subgingival calculus helps to perpetuate chronic periodontitis. It forms a reservoir of bacteria, helping to sustain inflammation, and acts as a barrier to healing.
Chronic inflammation
Plasma cells typically predominate but are accompanied by lymphocytes and neutrophils which migrate into pockets. Inflammatory cells infiltrate the connective tissue and spread between the principal fibres (Figs 5.9 and 5.10). Dense masses of these cells accumulate, especially under the epithelium in the connective tissue apposed to plaque and calculus.
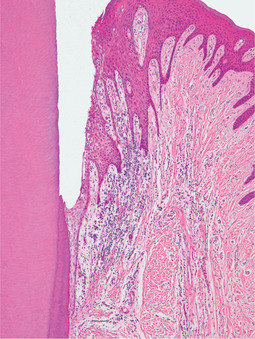 |
| Fig. 5.9
Transition from chronic gingivitis to periodontitis. No pocket has yet formed but the epithelial attachment has extended on to the cementum and inflammation has induced epithelial hyperplasia, as evidenced by rete hyperplasia.
|
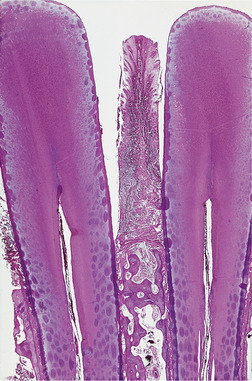 |
| Fig. 5.10
Established chronic periodontitis. In this woman of 33, periodontal pockets have extended well beyond the amelocemental junction and inflammatory cells fill the interdental gingiva.
|
Pocketing
Pocketing, a characteristic feature of chronic periodontitis, is due to the slower destruction of gingival soft tissues than of periodontal ligament and bone (Fig. 5.11). Less frequently, where gingival tissue is thin, it may be lost at the same rate as the supporting tissues and gingival recession results. Pockets round the teeth provide a protected environment in which bacteria grow more freely. There is no effective drainage and the wall of the pocket presents a large area where bacteria or their products can irritate the tissues. Pockets also favour the growth of anaerobes which probably contribute greatly to tissue destruction.
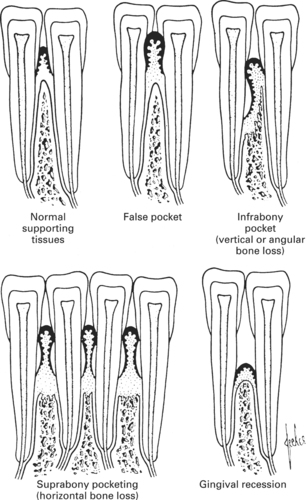 |
| Fig. 5.11
Pocket formation in periodontal disease. A simplified diagram to show the relationship between periodontal soft tissues and alveolar bone in the different presentations of periodontal disease.
|
Pockets surround the teeth. One wall is formed by cementum, the other soft tissue replacing the destroyed bone and periodontal fibres. The outer wall consists of connective tissue infiltrated by chronic inflammatory cells and lined by epithelium continuous with the gingival epithelium at the pocket mouth. The epithelial attachment forms the deep boundary. The epithelium is often hyperplastic, but blood vessels may extend through it almost to its surface.
‘Ulceration’ of the lining is often described but rarely seen histologically.
Epithelial migration
The epithelial attachment migrates from enamel on to cementum, forming the floor of the pocket (Figs 5.12 and 5.13). The attachment to cementum is strong and a clear refractile cuticle (see Fig. 5.2) can sometimes be seen joining the epithelium to the root surface. The length of the epithelial attachment is variable but may extend several millimetres.
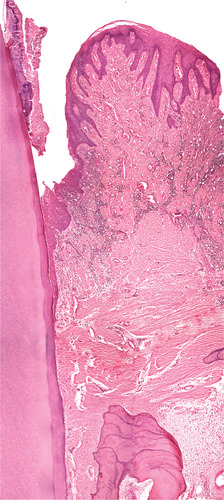 |
| Fig. 5.12
Chronic periodontitis. The amelocemental junction lies at the top of the picture covered by plaque and calculus that extends into the upper pocket. The epithelium of the pocket is hyperplastic and irregular but the epithelial attachment is tighly apposed to the cementum. The alveolar crest shows resting and reversal lines indicating remodelling. A broad band of uninflamed densely fibrous tissue, almost scar tissue, separates the bone from the inflamed tissue.
|
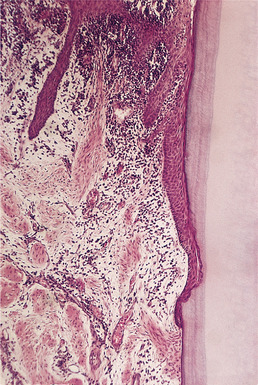 |
| Fig. 5.13
Chronic periodontitis. Higher-power view showing the epithelial attachment lying against cementum. Note how collagen has been lost from the areas containing inflammatory cells.
|
Destruction of periodontal fibres
Periodontal fibres are destroyed progressively from the gingival margin down to the level of the floor of the pocket. More deeply, the fibres retain their normal appearance.
Destruction of alveolar bone
Bone destruction starts at the alveolar crest. The usual level of the remaining bone is just deep to the floor of the pocket where the superficial fibres of the periodontal ligament are attached. The zone of inflammation is separated from the underlying bone by a zone of fibrous tissue. This so-called fibrous walling-off is typical of chronic periodontitis and invariably present (see Fig. 5.12). It has been suggested that the inflammatory lesion may extend beyond this to affect the alveolar bone and remaining periodontal ligament during periods of active disease, but histological evidence of this is lacking. Osteoclasts are rarely seen, probably because their action is intermittent and the rate of bone destruction extremely slow.
Osteoporosis and premature alveolar bone loss
Confirmation that osteoporosis may be related to premature alveolar bone loss has been handicapped by the difficulties in measuring bone mineral density (BMD) accurately. However, it has been confirmed that diminished bone mineral density, measured by dual X-ray absorptiometry in several parts of the skeleton, showed a highly significant correlation with the thickness of the mandibular alveolar process in the first premolar region.
Other studies have also shown that age-related bone loss is greater in women than men after the age of 50 years. There is also a significant increase in mandibular cortical porosity and concomittant decline in bone mass, with an estimated loss of bone mineral content of 1.5% per year in women, but only 0.9% in men.
Role of specific bacteria in destructive periodontitis
With formation of pockets and subgingival plaque there is enormous proliferation of bacteria of all kinds. Subgingival plaque appears to have a dense zone attached to the tooth surface where the bacteria are mainly Gram-positive. By contrast, plaque related to the pocket wall is less densely packed but involves many Gram-negative bacteria, including anaerobes and spirochaetes. Bacteria particularly associated with destructive disease include Porphyromonas gingivalis, Actinobacillus actinomycetemcomitans, B. intermedius, Wolinella recta, spirochaetes with axial filaments, or in other cases Eikenella corrodens, and other corroding bacteria. The latter’s colonies cause pitting (‘corrosion’) of the surface of culture media.
Also present are Fusobacterium nucleatum and as yet unidentified anaerobic vibrios and spirochaetes. Where there is severe and relatively acute bone loss associated with severe inflammation, in young adults, for example, Actinobacillus actinomycetemcomitans and Porphyromonas gingivalis appear to be particularly important. Where inflammation is minimal in spite of extensive bone loss there may be greater numbers of Prevotella melaninogenica and Eikenella corrodens. Many of the bacteria implicated in destructive periodontitis are dangerous pathogens and in other body sites can cause severe or lethal systemic infections. For example, Capnocytophaga ochraceus, Selenomonas sputigena and Eikenella corrodens have been implicated in life-threatening infections, particularly in patients with granulocytopenia.
In human periodontal disease, it remains unclear which particular types of bacteria are responsible for tissue destruction. However, inoculation of specific bacteria into the mouths of germ-free animals has yielded somewhat unexpected results. Thus, various Gram-positive bacteria typically produce large amounts of plaque, but Gram-negative bacteria do not. Certain bacteria of either group can induce or accelerate periodontal tissue damage, but there is typically little inflammation or lymphocytic infiltration of the tissues. In Gram-positive infections also, few osteoclasts are seen. However, A. viscosus in particular produces a bone-resorbing factor. Gram-negative bacteria also stimulate osteoclastic activity and numerous osteoclasts are seen at the sites of tissue destruction.
That individual species of bacteria may be responsible for periodontal tissue destruction is also suggested by the finding in monkeys that the severity of periodontal damage correlates well with the numbers of Porphyromonas gingivalis which can be isolated. Further, antimicrobial treatment that suppressed P. gingivalis led to healing and bone regeneration. Greater numbers of P. gingivalis also appear to be associated with enhanced periodontal disease activity in humans.
Bacterial mediators of tissue damage
Periodontal destruction may result from the actions of bacterial products or indirectly from immunologically mediated processes or both in varying degree. Bacterial products (virulence factors) that may contribute to tissue damage include enzymes, toxins and bone-resorbing factors.
Enzymes.
Destruction of periodontal fibres may be due to collagenases. Prevotella species, for example, produce collagenase, trypsin, fibrinolysin, hyaluronidase, chondroitin sulphatase, heparinase, aribonuclease and deoxyribonuclease. Cytotoxic metabolic products such as indole, ammonia and hydrogen sulphide may also contribute. Porphyromonas species, especially P. gingivalis, are regularly associated with human destructive periodontitis.
Toxins.
Certain strains of A. actinomycetemcomitans produce a powerful leukotoxin, while P. gingivalis and P. intermedius produce an epitheliotoxin.
Endotoxin, a lipopolysaccharide (LPS) component of Gram-negative bacteria cell walls, is released after cell lysis. It can liberate lysosomal enzymes from phagocytes and activate complement via the alternative pathway. Endotoxins can therefore mediate an inflammatory response. Endotoxins from some bacteria can enhance osteoclastic resorption to variable degrees and, in this respect, endotoxin from P. gingivalis is ten times more potent than that from A. actinomycetemcomitans. The Gram-positive cell wall component, lipoteichoic acid (LTA) is also a potent mediator of inflammation and enhances bone resorption.
Bone resorbing factors.
A. viscosus produces factors which induce decalcification of bone in vitro. Lipopolysaccharide can also induce bone resorption under experimental conditions, as can crude soluble extracts of human supragingival plaque and some other bacterial products. Bacterial factors can also cause bone resorption by stimulating release of, or acting synergistically with lymphokines, osteoclast activating factors and prostaglandin E 2.
Interference with host defence mechanisms
Other virulence factors act by interfering with host defences. Substances cytotoxic for human leucocytes (leukotoxins) are produced by A. actinomycetemcomitans in particular. This bacterium is involved in juvenile periodontitis and can also cause osteomyelitis and infective endocarditis in humans. Other bacterial factors potentially capable of damaging host defences are immunoglobulin proteases. Neisseria gonorrhoeae can produce IgA proteases which help it overcome mucosal defences and contribut/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


