SURGERY
7.1 APICAL SURGERY WITH ERBIUM LASER TECHNOLOGY
Rolando Crippa
When microorganisms enter the pulpal space, they contaminate the main root canal, proliferating through the dentinal tubules and ramifications of the root canal system until they reach the periapical area [1]. At this site, the resorption lacunae formed in the apical cement by the inflammatory reaction can be colonized by microorganisms [2,3].
In spite of a meticulous chemo-mechanical preparation, intracanal medicaments and antibiotic therapy, the literature counts many endodontic failures.
The major causes are:
- anatomical difficulties in cleaning the root canal;
- secondary infections caused by coronal microleakage or communications between the periapical region and the oral mucosa, such as periodontal pockets and sinuses (communications fundamental for the maintenance of periapical infections) or other situations that give a bacterial infection [1,2].
Recent studies using anaerobic techniques have revealed that the composition of the root canal microbial flora after treatment failure differs from that normally found in necrotic pulp. The wide variety of combinations of bacteria in necrotic pulp averages four to seven species per canal, predominantly anaerobic, with approximately equal proportions of Gram-negative and Gram-positive bacteria [4].
In contrast, the microbial flora detected in apical periodontitis of treated teeth counts one, two or sometimes three microbial species, with predominantly Gram-positive microorganisms (Enterococcus faecalis, Actinomyces spp. and Propionibacterium spp) facultative o strict anaerobes [4] and other times it comprises fungal species such as Candida albicans. Its presence is often an event caused by a physically or chemically canalar treatment [1].
The microorganisms could produce a polysaccharide layer resistant to intracanal medicaments, to host defence and to antibiotics, protecting themselves from any attacks. Thus, these periapical biofilms remained at this site act as a continuous irritation, releasing by-products such as endotoxins; in these cases the microbial biofilms can only be removed and disrupted physically and not chemically.
It’s clear that the only orthograde retreatment is not an effective therapeutic procedure for the treatment of teeth with chronic periapical lesions. Surgical endodontics seems to be the only method useable with teeth with periapical lesions when orthograde treatment is unsuccessful [3,12].
Apicoectomy in odontology indicates the surgical root-end resection when there are periapical lesions.
An accurate differential diagnosis is the only way to give a medical indication to apicoectomy. Usually the indication is the apical osteitis of a non vital tooth [14]. Its aim is delete granulomatous tissue created in the apical area. Its presence depends on chronic inflammatory process of periapical tissue caused by toxins and bacterial action, products of pulp necrosis in the apical delta and on irritant action of sealing materials in according to different endodontic techniques.
Clinical indications to get an apicoectomy are manifold:
- the presence of an apical granuloma in a tooth bearing an insurmountable anatomic difficulty in the canal inspection;
- the failure of the endodontic therapy on a tooth with an apical granuloma;
- a transversal fracture of the apical third of the dental root;
- the dental apex perforation and an ineffective endodontic closing;
- the breaking of spinning or manual tools at the third apex and the resulting retention or partial leakage of them which prevent a correct apical closing;
- difficulties in sealing depending on anatomical apical anomalies (curves, apical delta, apical infundibular, etc…);
- chronic infective flare-up and defective drain of apical abscess;
- the presence of radicular pin in tooth affected with apical granuloma which could split the root, trying to remove them, or which can’t be removed;
- the presence of untimely dental apices or apical lysis;
- removal of maxillary cysts involving several root apices;
- excess of endodontic material, come out the root apex causing inflammation and chronic apical infection;
- impossible root canal probing caused by new formation of secondary dentin in a tooth needing conservative or prosthetic treatment.
Several studies reveal many failures in retrograde therapy too. They suggest a new operation instead of the tooth extraction.
A new retrograde treatment, getting with more modern techniques and innovatory sealing, could reach the healing of several teeth that failed in the first time.
In addition a better disinfection of the radicular canal final area and of periapical area, with a more adequate closing of the apical foramen, might enhance the success of the therapy [3].
In the light of these scientific discoveries the use of laser surgery improves the number of therapeutic approaches in the endodontic retrograde treatment [5].
The first laser use in endodontics was reported by Weichman & Johnson (1971) who attempted to seal the apical foramen in vitro by means of a high power-infrared (CO2) laser [5].
Further studies have examined the capability of different lasers in sterilizing radicular canals.
It was used the Nd:YAG laser (1064nm), XeCI laser (308nm) [7], Diode laser (810nm) (Moritz et al. 1997), the Er:YAG laser (2640nm) (Gomi et al.), the Nd:YAG laser (1340nm) (Blum et al. 1997). All of them had demonstrate a great bactericidal capability related to their specific power settings.
In 1983 Eriksson & Albrektsson found that the threshold level for bone survival was 47°C for 1 min and that too high power might cause thermal injury to paradontal tissues. Thus it is necessary to select an appropriate laser parameter [5].
Furthermore for reducing the temperature increase is better using laser in pulse modality.
The Nd:YAG laser, for its thin optical fiber usable in radicular canals, has the mayor use in these cases.
Results demonstrate a great anti-inflammatory power such as it might give a great aid to traditional orthograde endodontic treatment.
Thanks these results several studies in vitro [7,8,9] have proved the Nd:YAG laser capability in reducing the bacterial leakage in selected roots (after an apicectomy and a retrograde filling).
After the establishing of laser antibacterial capacity, the laser Er:YAG in vitro studies tried to obtain smooth radicular sites, without impurities and carbonised sides.
They reach the goal using 50-90mJ power, in pulse modality, at wet and dry fields.
Other researches have demonstrated that Er:YAG laser irradiation was more effective in removing the smear layer and debris on root canal walls than the Ar or Nd:YAG laser (Takahashi et al. 1996, Matsuoka et al. 1998, Takeda et al. 1998a,b,c, 1999), however it is almost impossible to clean the lateral canal walls.
In 1988 Miserendino, in his clinical investigations, used CO2 laser for apicectomy but results appeared unsatisfactory.
Thus the Er:YAG laser becomes the more used laser during apicectomies.
Many clinical investigations [17] have demonstrated that the Er:YAG laser use in radicular resection improves the healing and increases the post-treatment compliance.
Although the speed cut is quite inferior than traditional surgery methods (spinning tools), it reduces the probability of a surgery site infection and of a shock in the surrounding tissues [18].
Others have verified that, using this laser, the retrograde cavity preparation is faster and clearer than with ultrasonic [20].
No significant differences indeed were noticed in the sealing of retrograde cavities prepared with laser Erbium:YAG (350mJ and 6Hz) than mechanic tools [19].
A research, using the scanning electron microscope (SEM) and the optic microscope, has weighted effects at apical seal after an apicectomy with Er:YAG and Nd:YAG lasers [11].
Results show that, the use of the Er:YAG laser for the apicectomy and Nd:YAG for the retrograde cavity preparation is a good choice to obtaining the reduction of the dentinal permeability.
Clinical case A
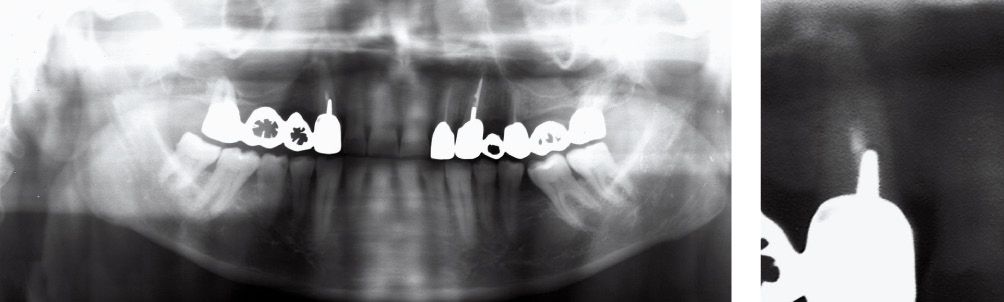
Figure A-1 • Adult male patient presenting an apical granuloma and wrong way of the tooth 1.3. The initial X-ray reveals a prosthetic covering and a reconstruction by endocanalar screw.
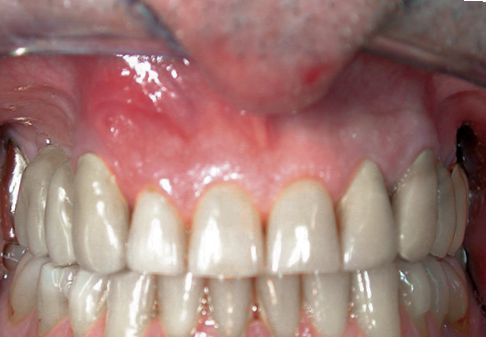
Figure A-2 • Clinical examination: a vestibular fistula of tooth 1.3 is present.
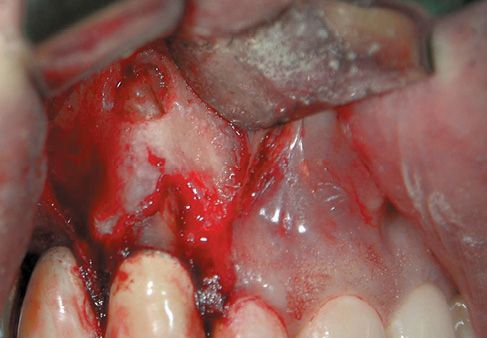
Figure A-3 • Opening of the access edge by using CO2 laser, power 2.0W in superpulsed mode. There are a wide apical lesion and a bony dehiscence near the middle third of the root caused by a false canal.
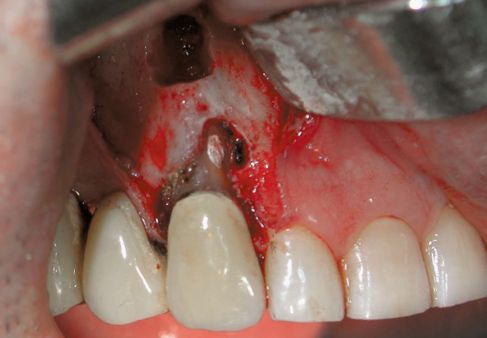
Figure A-4 • Apicoectomy was performed using an Er:YAG laser 2940nm, 600 micron fibre, power 400mJ, frequency 14pps. Cleaning of the wrong way at the middle third and retrograde filling of apex and wrong way using SuperEba cement. Decontamination of the entire area by using CO2 laser (SP mode, power 1W).
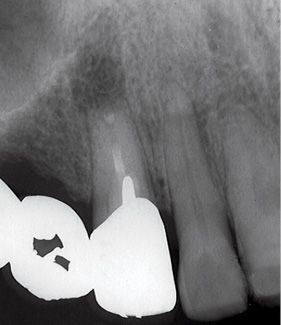
Figure A-5 • Post-operative X-ray: note the SuperEba apical seal and the false way closing.
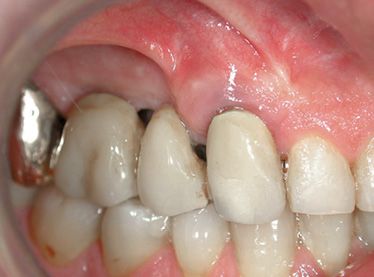
Figure A-6 • Three weeks later: good periodontal health.
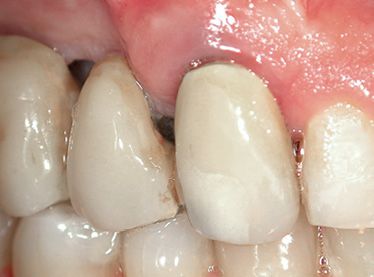
Figure A-7 • Note the gingival margin of tooth 1.3. The sticking mucosa appears like orange peel.
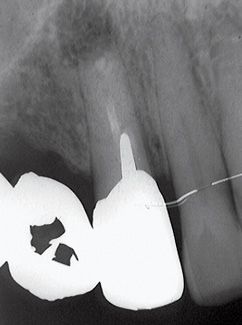
Figure A-8 • Control X-ray three months later.

Figure A-9 • Control X-ray six months later.
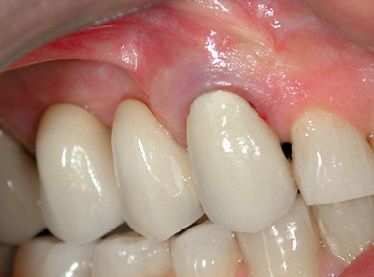
Figure A-10 • Cementation of the new ceramic prosthesis. Note the preserved anatomy of gingival tissue.
Clinical case B
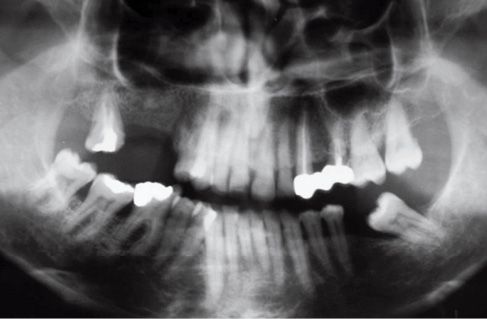
Figure B-1 • Adult female patient presenting an apical granuloma of tooth 2.3.
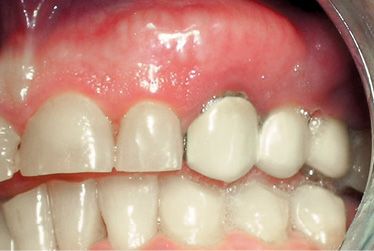
Figure B-2 • Clinical appearance before treatment.
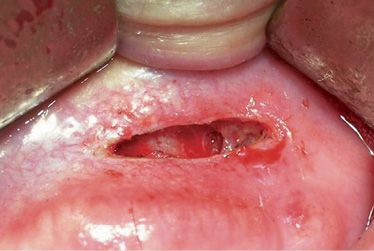
Figure B-3 • Cut made by ErCr:YSGG laser, using 400 micron fibre, length 6mm, power 1.7W, 7% of H2O and 11% of air.
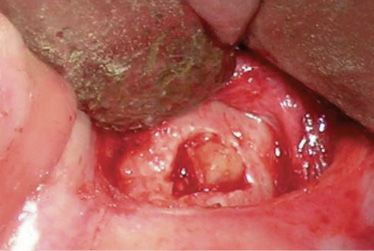
Figure B-4 • A good haemostasis gives a good view of the operating field.
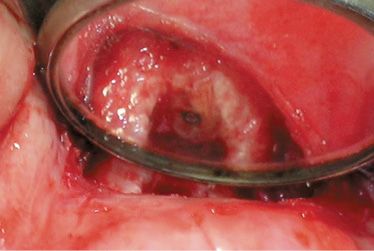
Figure B-5 • After surgery the apex has been decontaminated, cutted off by using the laser, then retrofilled with guttapercha.
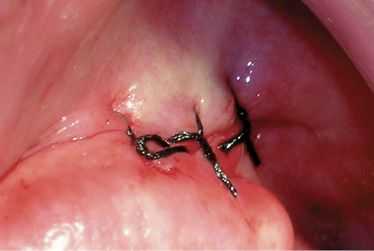
Figure B-6 • The suture made with silk stitch.
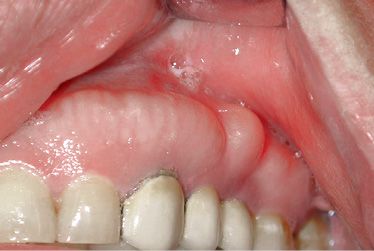
Figure B-7 • Clinical outcome 1 month later: healing of gingival tissues.
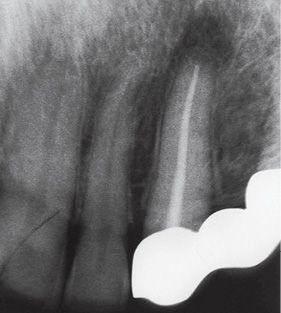
Figure B-8 • The X-ray control reveals a good apical bone repair.
Clinical case C
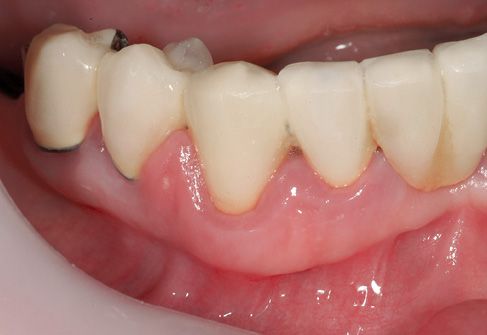
Figure C-1 • Female patient presenting an apical granuloma of tooth 4.2. It is a bridge pillar, it is endodontically treated and reconstructed by a metal pin.
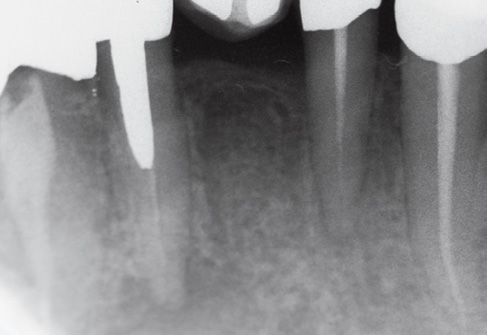
Figure C-2 • The X-ray investigation reveals an apical lesion.
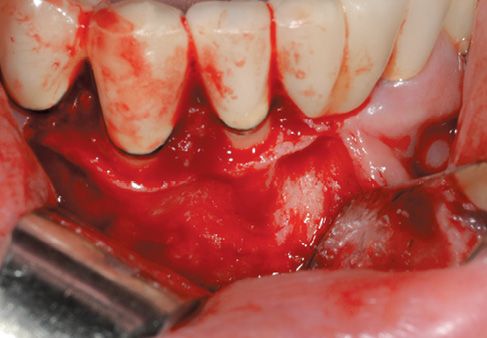
Figure C-3 • Surgical edge prepared with ErCr:YSGG laser, using 400micron fibre, S program (soft tissue), power 2.0 W, 5% of H2O and 20% of air.
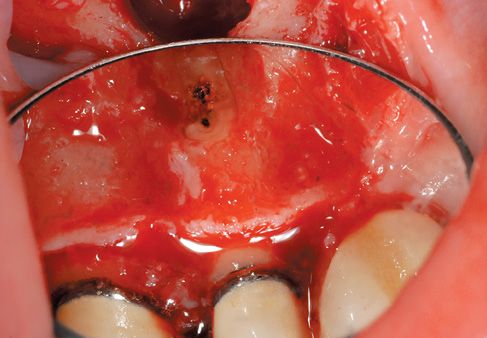
Figure C-4 • The apicoectomy is executed with laser power 6.0W, program H (hard tissue), 20% H2O, 40% air. The guttapercha condensation and the decontamination are performed using these settings: 2.0W power, 0% H2O and 20% air.
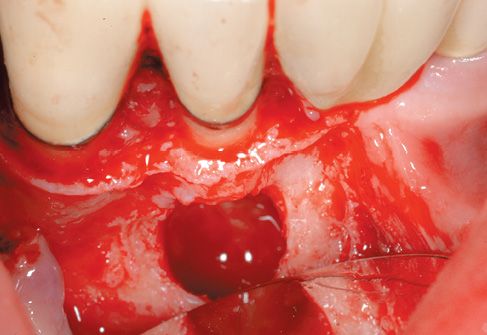
Figure C-5 • The bony cavity after surgery.
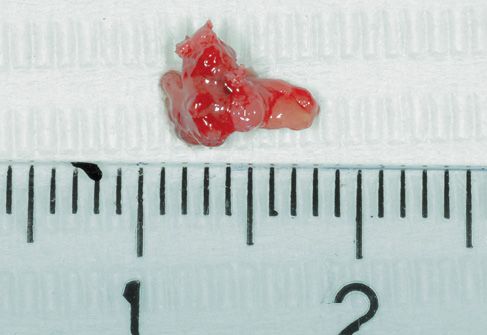
Figure C-6 • Bioptic specimen after excision.
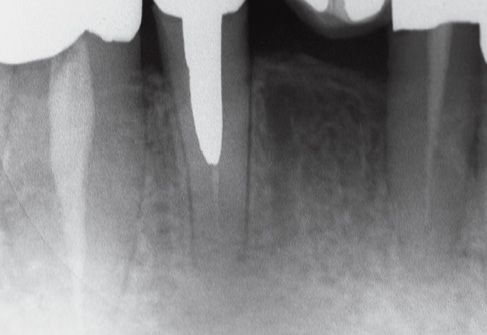
Figure C-7 • Post-operative radiological control.
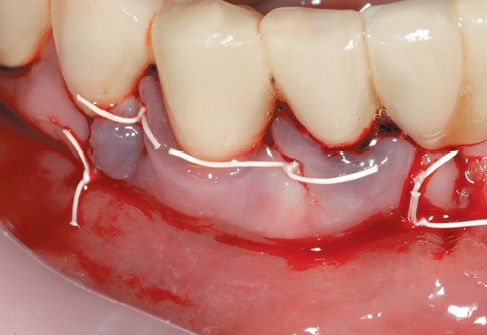
Figure C-8 • Suture of the edge with single teflon stiches 4.0.
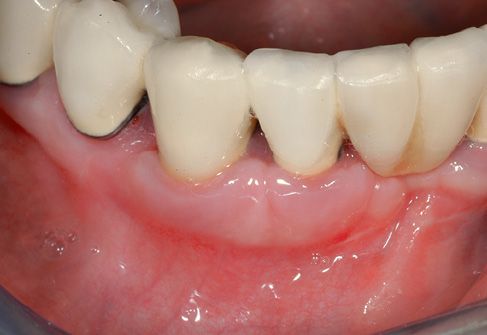
Figure C-9 • Healing of gingival tissues and clinical outcome.
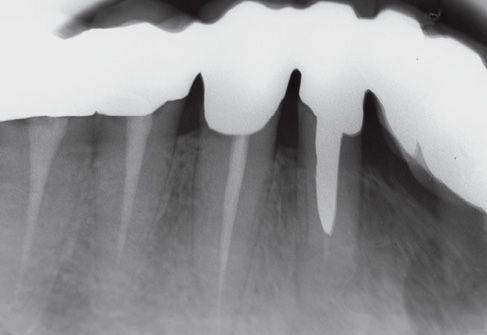
Figure C-10 • Clinical and radiological control after 1 month.
Discussion
The laser is usable for each step that characterizes the endodontic surgery (surgical cut of the mucosa, osteoplastic surgery, apical resection, retrograde cavity preparation). Especially some Erbium lasers are provided with flexible terminals in different sizes. Their lengths enable the canal cleaning for several millimeters. Their effect is comparable to the ultrasonics one that are used for the cleaning of canals after their resection. The sealing of these canals may be done using well known sealants, particularly Supereba cements.
The use of Erbium laser in apicoectomy offers to the operator some advantages: operating in an bloodless and decontaminated field where the tissues are biostimulated by laser.
About patient benefits there’s no cutter vibration, a faster healing and a less post-operating edema. Today the laser use for apicoectomy presents many advantages and during next years the technologies improvement and the expenses decreasing surely will replace traditional methods in endodontic surgery.
REFERENCES
1. Ferreira FBA, Ferreira AL, Gomez BPFA, Souza-Filjo FJ. Resolution of persistent periapical infection by endodontic surgery. International Endodontic Journal 2004; 37:61-69.
2. Siqueira JF, Jr, Lopes HP. Bacteria on the apical root surfaces of untreated teeth with periradicular lesions: a scanning electron microscopy study. International Endodontic Journal 2001; 34:216-20.
3. Gagliani MM, Gorni, FGM, Strohmenger L. Periapical resurgery versus periapical surgery: a 5-year longitudinal comparison. International Endodontic Journal 2005; 38:320–327.
4. Sundqvist G, Figdor D, Persson S, Sjogren U. Microbiologic analysis of teeth with failed endodontic treatment and the outcome of conservative re-treatment. Oral Surgery, Oral Medicine and Oral Pathology 1998; 85:86-93.
5. Kimura Y, Wilder-Smith P, Matsumoto K. Laser in endodontics : a review. International Endodontic Journal 2000; 33:173-185.
6. Miserendino LJ. The laser apicoectomy: endodontic application of the CO2 laser for periapical surgery. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiolog and Endodontics 1988; 66:615-9.
7. Stabholz A, Khayat A, Weeks DA, Neev J, Torabinejad M. Scanning electron microscopic study of the apical dentine surfaces lased with Nd:YAG laser following apicoectomy and retrofill. International Endodontic Journal 1992; 25:288-91.
8. Stabholz A, Khayat A, Ravenshad SH, McCarthy DW, Neev J, Torabinejad M. Effects of Nd:YAG laser on apical seal of teeth after apicoectomy and retrofill. Journal of Endodontics 1992; 18:371-5.
9. Wong WS, Rosemberg PA, Boylan RJ, Schulman A. A comparison of the apical seals achieved using retrograde amalgam fillings and the Nd:YAG laser. Journal Of Endodontics 1994; 20:595-7.
10. Paghdiwala AF. Root resection of endodontically treated teeth by Erbium:Yag laser radiation. Journal Of Endodontics 1993; 19:91-4.
11. Oliveira RG, Gouw-Soares S, Baldochi SL, Eduardo CP. Scanning electron microscopy (SEM) and optical microscopy: effects of Er:YAG and Nd:YAG lasers on apical seals after apicoectomy and retrofill. Photomed Laser Surg 2004; 22:533-6.
12. Danin J, Stromberg T, Forsgren H, Linder LE, Ramskold LO. Clinical management of nonhealing periradicular pathosis. Surgery versus endodontic retreatment. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiolog and Endodontics 1996; 82:213-7.
13. Bosnev. Terminologia Stomatologica Poliglotta. SI, Stampa Medica, 1981.
14. Harnisch H. Apicectomia. Milano, Scienza e tecnica dentistica, 1981.
15. Barnes M. Apicectomia. Milano, Scienza e Tecnica dentale, 1986.
16. Moritz A, Schoop U, Goharkhay K, et al. The bactericidal effect of Nd:YAG, Ho:YAG and Er:YAG laser irradiation in the root canal:an in vitro comparison. J Clinic Laser Med Surg 1999; 17(4):161-64.
17. Komori T, Yokoyama K, Takato T, Matsumoto K. Clinical application of the erbium:YAG laser for apicoectomy. J Endod 1997 Dec;23(12):748-50.
18. Komori T, Yokoyama K, Matsumoto Y, Matsumoto K. Erbium:YAG and holmium:YAG laser root resection of extracted human teeth. J Clin Laser Med Surg 1997 Feb;15(1):9-13.
19. Francischone CE, Padovan LA, Padovan LE, Duarte MA, Fraga Sde C, Curvello VP Apicoectomy with the Er:YAG laser or bur, followed by retrograde root filling with zinc oxide/eugenol or sealer 26. Photomed Laser Surg 2005 Aug;23(4):395-8.
20. Karlovic Z, Pezelj-Ribaric S, Miletic I, Jukic S, Grgurevic J, Anic I. Erbium:YAG Laser versus Ultrasonic in Preparation of Root-End Cavities.J Endod 2005 Nov;31(11):821-823.
7.2 Laser Treatment in Medicine and Oral Pathology
Giovanni Maria Gaeta
The use of Erbium laser in Oral Medicine has determined a real progress in treatment technique of oral mucosa lesions, faster healing and improvement in post-surgical consequences, reduction of microbiological contamination, and more compliance in the patient, obtaining results that were hardly achieved through the use of the former techniques.
The main goals whished by Oral Medicine helped by laser are certainly the treatment wide spread hyperkeratosis and leukoplakias of the soft tissues, treatment of bone lesions, removal of benign and dysplastic or neoplastic lesions, treatment of infectious lesions.
Biological Functioning of Lasers: Selective Phototermolysis
Every surgical procedure is aimed to remove the pathologic lesion with a minimum damage of the surrounding tissues, in order to start a topical healing process without having any brief, medium and long term complications.
This theory, which can be applied and in every surgical procedure, is also suitable for the use of laser lights.
There is a widespread availability of different laser frames, each one with specific features, depending on the wavelengths produced. In addition, this features depend not only from the kind of tissue-section, the working time, the deepness of surgical wounds and the methods of working, but also from the specific absorption of laser waves of every single tissue component. Therefore, laser can be considered, not only an advanced technology which allows the surgical treatment of organic lesions with extreme easiness and precision, but also a selective therapeutic method which allows the identification of the organic and tissue component which can be addressed with absolute accuracy. For this assertion, we can refer to the physical-organic principles of the selective thermolysis theory, elaborated by Anderson and Parrish in the 80’s. Twenty years later, selective thermolysis has changed the goals of the use of laser, especially in the treatment of superficial tissues.
The word selective thermolysis has been coined to describe a thermal tissue damage of biological targets by appropriate selection of wavelength.
The light releases energy only in the areas where it is absorbed. Then, the wave-lengths penetrate into the tissues and are absorbed especially by chromophores, as blood vessels or cells containing melanin and thus generate heat. As soon as heat is generated, it starts to dissipate, because of the conduction or the irradiative transfer. The contrast between the active heating and the passive cooling produces the overheating of the desired area. It is possible to achieve a more selective overheating, when the laser pulse intensity is longer then the cooling time.
Selective photothermolysis is based on three elements: wavelength which penetrates and is absorbed especially by the aimed targets; laser pulse duration inferior or equal to thermal relaxation time; correct fluence to the target to produce the desired temperature rise (related to the size of the lesion). When these conditions are achieved, extremely selective tissue damage can be obtained in thousands of microscopically targets, and there’s no need to focus the laser on every single one of them. Selective thermolysis may achieve a great variety of thermal damages mediated by the heat, as thermal denaturation, mechanical damage due to the fast diffusion of heat or cavitations, or changes in the primary chemical structures (pyrolisis). Indeed this method represents the most precise way of using heat or any energy-source, achieved until now in the whole medical field.
Theoretical Considerations on wound healing
Before re-epithelization of a wound, inflammatory cells and macrophages that occur over the wound were recovered to eliminate potential contaminants (bacteria/yeast) as well as nonviable tissue.
In this process, inflammatory mediators, stimulate angiogenesis, attract additional inflammatory cells, and stimulate the production and release of various growth factors. The total effect stimulates wound healing through epidermal migration and proliferation as well as epithelial remodelling.
Wound healing occurs through a balance of inflammatory mediators and cells. If excessive angiogenesis results, leading to prolonged clinical erythema and delayed wound healing. As an extent of nonviable tissue in the wound, it is reasonable to expect a disruption of optimal wound healing when nonviable tissue is present in the wound.
When we use CO2 laser at 300 mJ, two passes, and the average extent of thermal damage of epithelial tissue is 60-100 micros. When Erbium laser 15/J cm2 is 10-30 micros.
The Erbium laser vaporization produce this effects because the affinity for the water at 2940 nm is 10 times greater 10.600 nm wavelength of CO2 laser. Thus the Erbium removes about the 75% of these nonviable tissues after 48-72 hours from treatment there is a marked reduction of polymorphnucleates and audiophiles and nuclear debris respect CO2 laser. The angiogenesis is present for both treatments. The epithelium reforms 3-4 days faster with Erbium than CO2 at one week the re-epithelization is complete for the two groups but in the CO2 specimens the mild inflammatory infiltrate is greater than the Erbium groups.
The Erbium laser treatment leave soft oral tissue surfaces clean from nonviable tissues, products of denaturation of proteins and pyrolisis and infectious contaminant. The same is after the treatment of hard oral tissues (bone and dentin) with strong reduction of debris and smear layer. (See suggested lectures)
This condition lead to a better and faster healing after surgical procedures, a reduction of post operative complications and side effects and increase the patient’s compliance.
Erbium Laser: YAG
The frequency emission is 2940 nm.
Strongly absorbed by water.
The Erbium laser leads to the possibility of working on all tissues involved in the oral pathology (mucosa, bone and dentin).
It has a great thermal effect because it is very well absorbed by water; it can be used with very short pulses, between 50 and 100 microseconds, which allow mechanical ablation of layer-cells and minimum thermal damage. For this reason it is recommended either for the removal of peduncles and sessile tissues, or vaporisation of oral cavity wide lesions, even in physiologic areas susceptible to thermal changes. Very good post-surgical resolution, with no scars. The more quantity of water is contained within the tissue, the greater the ablative result. Excellent for the removal and molding of bone. The coagulation effect is reduced except when using long pulses and high frequencies. Coagulation effect increase remarkably in smooth Erbium lasers because of the thermal increase.
Laser ray can derive from fibers or articulated arms (Tables 7.2.1-7.2.2).
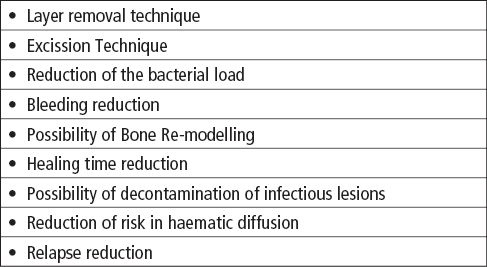
Table 7.2.1 • Rationale for the use of Erbium laser in Oral Medicine.
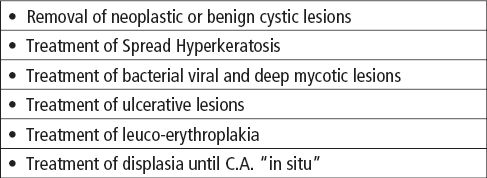
Table 7.2.2 • Suggestions for the use of laser in Oral Medicine.
Premises for the use of laser system in Oral Medicine: biopsy of Oral Lesions
Oral cavity lesions can be faced with several therapeutic attitudes. In same cases they present clinical aspects which even a skilful operator is unable to distinguish.
Whatever therapeutic choice is taken, a sure diagnosis is required, by taking a bioptic sample and doing soon after an histological exam.
If the lesion removal is made by laser, the result may outcome unreadable from an anatomopathologist, or either completely destroyed, if the lesion itself has been vaporised.
The treatment of any kind of lesion might turn ineffective or harmful if it is not preceded by an accurate diagnosis (clinical and histological).
The bioptic sample has to be done after the highlighting of the lesion by vital stain (Toluidine Blue), then carried out in the area of mayor colorimetric contrast using a cold-blade knife (BP 11; 15c; Punch 4/6 mm).
The cytological sample, accomplished by spud, scraper or brush can give false-negatives and results completely useless in hyperkeratosic lesions. Therefore, do not use it to ascertain the malignancy of the lesion itself.
The sample will be fixed in formalin buffered to 10% in liquid ratio biotic sample 10 to 1 and then sent to the anatomopathologist for the staining.
A prescription, written by the doctor who has taken the sample, has to be added, which reports the patient’s registry details, including telephone number, doctor’s details, sample anatomic area, lesion description and the assumed clinic diagnosis.
Ask explicitly for the specific microscopic marker is being researched, in order to address the anatomopathologist to the most suitable technique and histological staining. Only in this case the information received from the anatomopathologist will be completed.
Brush or swab technique on the lesion is undoubtedly advisable in case it results a viral or mycotic infective pathology; however, the anatomopathologist has to be clearly advised about the diagnostic suspect. In case it is necessary to search for the virus or the mycete, plunge in the specific culture medium after the sample.
As for the infective lesions considered potentially malignant, it is possible to request the typing of the mycotic or viral strain.
Suggestions for a wise therapeutic and diagnostic behaviour.
- Any lesion in oral cavity which clinically appears as ulceration, erosion, plaque, nodule or vegetating hyperplasia, has to be considered suspected until it is verified the opposite.
- If there isn’t any irritant or trauma that can be highlighted nearby the suspected lesion to validate it, biopsy must be done.
- On the contrary, if it’s possible to identify a trauma (cut edge, fractured crown, and overhanging restoration etc.), this will be removed.
- The patient will be checked after one week, and if healing signs of the lesion are found, next check will be performed after some days, until it will be completely recovered. In case the opposite happens, biopsy has to be done.
- Do not use palliative therapies as mouth rinses, cortisone ointments or anti-inflammatory. These could result in a symptomatologic cure and disguise the patient’s pathology.
- If any doubt exists, patient must undertake biopsy or be sent to a specialised health centre.
- Excision biopsy should be avoided in non-equipped health centres, because incomplete excision, performed without considering the edges of extension, may exacerbate prognosis, alterating the anatomopathological report.
- Incision biopsy must be decided, or, at least, the patient must be addressed to the specific health centre.
- For neoplastic lesions larger than 3cm, perform more than one bioptic sample, depending on the clinical aspect and the vital stain of the lesion itself.
- The bioptic fragment must never be divided in pieces. The preparation could get unreadable, and then the diagnostic report may result unsuitable (Table 7.2.3).
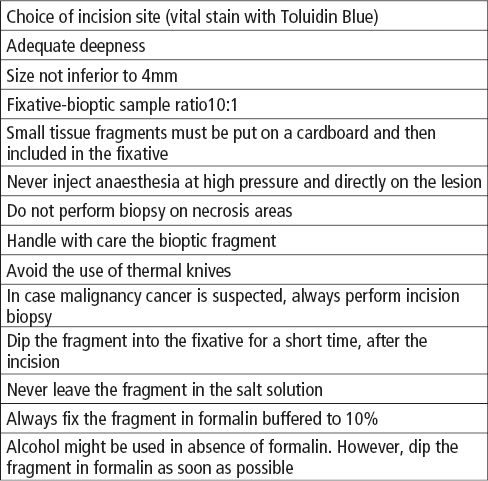
Table 7.2.3 • How to perform biopsy correctly.
Infectious Disease
Herpetic Lesions (Herpes simplex 1).
Herpes simplex viruses (HSVs) are DNA viruses that cause acute skin and mucosa infections and present as grouped vesicles on an erythematous base. Rarely, these viruses can cause serious illness. Most infections are recurrent and tend to reappear at or near the same location. Herpes labialis is the most common infection caused by HSV type 1 (HSV-1).
Intimate contact between a susceptible person (no preantibodies) and an individual who is actively shedding the virus is required for HSV infection to occur. Contact must involve mucous membranes or open or abraded skin.
HSV invades and replicates in neurons as well as in epidermal and dermal cells. Virions travel from the initial site of infection to the sensory dorsal root ganglion, where latency is established. Viral replication in the sensory ganglia leads to recurrent clinical outbreaks. These outbreaks can be induced by various stimuli, such as trauma, ultraviolet radiation, and extremes in temperature, stress, immunosuppression, or hormonal fluctuations.
Herpetic Gingivostomatitis is one of the primary manifestations of Herpes Simplex1 virus infection.
Clinically, Herpetic Gingivostomatitis appears with subjective and objective symptoms. Subjective symptoms are 3-4 day high fever, painful lymphadenopathy and malaise. Objective lesions are erosive lesions in the first half of oral cavity, including frictional mucosa, (gums and hard palate) with 1 mm- monomorphic lesions on perioral and face skin, which tend to join. Primary herpetic gingivostomatitis usually resolves within approximately 14 days.
Treatment of herpes generally consists of episodic courses of oral acyclovir and prodrugs valacyclovir and famciclovir.
Oral Candidiasis
Candida albicans infection appears in the oral cavity as spores or hyphas. Hyphas Mycelium is more aggressive, as they penetrate deeply into the tissue, resulting in clinical lesions. However, three general factors may lead to mycotic infection: the immune status of the host, the oral mucosal environment, the strain of microrganism. The pathogenicity of Oral Candida may depend on its capacity of recognising the polyptychial mucosal epithelium, adhering to epithelium cells, forming hyphas and producing enzymes (proteases) which perform a keratolytic effect.
Clinical Aspects
Oral candidiasis occurs with variable clinical aspects:
Pseudomembranous candidiasis
Erythematous candidiasis
Atrophic candidiasis (median rhomboid glossitis)
The diagnosis of oral candidiasis is most frequently made on the basis of clinical appearance, colture examination, and bioptic examination using PAS staining or fluorescence examination.
Treatment of candida infections primarily is accomplished with appropriate antifungal drugs.
Human Papillomavirus
HPVs are members of the papovavirus group, which are icosahedral viruses that contain circular DNA. The name papova is derived from the first 2 letters of their common clinical features, as follows: papillomatous lesions, polyomas (varied tumor induction), and vacuolization of infected cells.
More than 100 types of HPV have been discovered. The infectivity of HPV is poorly understood. Some types may act in concert with cofactors, such as diminished immunity or sun exposure.
The effects of HPVs in the oral cavity are usually considered to be benign, though certain types have been associated with malignancies of the anus, the genital tract, and other areas, including the oral cavity.
The virus penetrates the mucosal epithelium and invades the cells of the basal layer, where the viral circular DNA inserts into the host DNA.
Most oral lesions caused by HPV are benign lesions with rare malignant potential. Lesions are usually painless. They are generally not ulcerated, though they may be secondarily ulcerated by trauma. Lesions may be more common and severe in patients who are immunocompromised than in other patients.
Lesions are usually treated with excisional biopsy.
Erbium:YAG Treatment of Infectious Lesions
The Erbium laser may be used to ablate or decontaminate infective lesions.
In herpetic lesions treatment must be performed in the first stage of the illness, (vesicular stage), when the viral load has a maximum concentration.
Usually the lesion is irradiated for 1 minute with high-medium power. The emission pulse with Erbium laser is between 100 and 150 mJ, with a considerable length, (maximum heat deposit) and frequency between 5 and 15 Hz. If it is possible to change the pulse length, the right pulse is from 500 to 1000 microseconds. Laser should be moved on the lesion surface from a 4-6 mm distance.
The ablative effect of Erbium laser causes rupture and bleeding of the vesicular lesion; therefore, once removed the tectum, the bottom will be treated.
Candidiasis lesions which can be treated with laser are all the chronic lesions, either of hyperplastic, atrophic and recurrent forms and the ones occurring in immunosuppressed patients (associates with medical treatment). Deep tissue, where usually mycotic hyphas penetrate, must be de-contaminated.
In hyperplastic forms, excision and vaporisation of the lesion will be performed, using surgical-like methods (excision).
For atrophic and recurrent lesions the treatment will be the same used for viral lesions. With the Erbium laser the emission pulse is between 100 and 150 mJ, with a length of 1000 microsec, frequency between 10 and 30 Hz.
For pseudo-membranous lesions, after removing the pseudo-membrane with vaporisation, the erythematous area will be treated using viral decontamination methods.
Papillomavirus lesions are treated with thermal excision for peducolate or wart-like forms, and with vaporisation in plan-forms.
Deep removal is advised to contrast the recurrence high tendency, performing a complete ablation of the epithelium until periosteum (frictional mucosa lesions) or muscular layer (non frictional mucosa lesions). These kinds of lesions usually need 2-3 treatments, performed every 7 days.
Leukoplakia and Frictional Hyperkeratosis
Oral leukoplakia (OL) is a white patch or plaque that cannot be rubbed off, cannot be characterized clinically or histologically as any other condition, and is not associated with any physical or chemical causative agent except tobacco. Therefore, a process of exclusion establishes the diagnosis of the disease. In general, the term leukoplakia implies only the clinical feature of a persistent, adherent white plaque; therefore, reserve the term for idiopathic lesions when investigations fail to reveal any cause. The term carries absolutely no histologic connotation, although, inevitably, some form of disturbance of the surface epithelium is characteristic.
Follow-up studies suggest that cancer is more likely to occur in individuals with idiopathic leukoplakia than in individuals who do not have this condition. Thus, idiopathic leukoplakia is considered a premalignant lesion.
The aetiology of most cases of OL is unknown (idiopathic). In other cases, the initiation of the condition may depend on extrinsic local factors and/or intrinsic predisposing factors. Factors most frequently blamed for the development of idiopathic leukoplakia include tobacco use, alcohol consumption, chronic irritation, candidiasis, vitamin deficiency, endocrine disturbances, and possibly a virus.
Three stages of OL have been described.
The earliest lesion is no palpable, faintly translucent, and has white discoloration.
Next, localized or diffuse, slightly elevated plaques with an irregular outline develop. These lesions are opaque white and may have a fine, granular texture.
In some instances, the lesions progress to thickened, white lesions, showing induration, fissuring, and ulcer formation.
Clinically, OL falls into 1 of 2 main groups.
The most common are uniformly white plaques (homogenous OL) prevalent in the buccal mucosa, which usually have low premalignant potential.
Far more serious is speckled or verrucous leukoplakia, which has a stronger malignant potential than homogenous leukoplakia. Speckled leukoplakia consists of white flecks or fine nodules on an atrophic erythematous base. These lesions can be regarded as a combination of or a transition between leukoplakia and erythroplakia, which is flat or depressed below the level of the surrounding mucosal red patch, is uncommon in the mouth, and carries the highest risk of malignant transformation.
Five clinical criteria demonstrate a particularly high risk of malignant change.
- The verrucous type is considered high risk.
- Erosion or ulceration within the lesion is highly suggestive of malignancy.
- The presence of a nodule indicates malignant potential.
- A lesion that is hard in its periphery is predictive of malignant change.
- OL of the anterior floor of the mouth and under surface of the tongue is strongly associated with malignant potential.
In all cases, the relative risk of malignant potential is determined by the presence of epithelial displasia upon histological examination.
In persons who smoke, the combustion end-products brought about by burning tobacco and heat (eg, tobacco tars and resins) are irritating substances capable of producing leukoplakic alterations of the oral mucosa.
Surgical excision of OL may be considered. Frequent clinical observation accompanied by photographic records is recommended.
Because of the unpredictable behaviour of dysplastic lesions, immediately obtain a biopsy on any areas that are suggestive or that change in appearance.
Cryotherapy ablation and carbon dioxide laser ablation are also used. The area heals rapidly, and apparently healthy mucosa is left behind. However, uncertainty remains regarding the risk of invasive carcinomas subsequently arising in sites previously treated.
Topical retinoids are ineffective.
Systemic retinoids may be effective, but they have toxic effects.
Erbium:YAG Treatment of Leukoplakia or Hyperkeratosic lesions
After the histological diagnosis, excision or vaporisation technique is used. The lesion is vaporised with Erbium laser using a hand piece of 05-1 mm diameter spot size. Spot diameter can be increased along with the working distance.
The lesion is treated by dividing it in dials and using the overlapping technique (spot overlapping and layer excision, until the lesion is completely removed in every single dial). Usually the ablation is performed until the connective tissue is completely removed.
In some cases the removal must be achieved more deeply, until periosteum on adhering mucosa and muscular stratum on free mucosa.
All the smokes must be removed during vaporisation. In order to reduce bleeding, it is advisable to de-focus the ray, increasing the distance from the lesion to 5 cm, using a long pulse and high frequency (stronger thermal effect), without using sprays.
Erbium laser parameters are: Pulse: 250-350 mJ; Pulse Lenght: 500-1000 microsec; Frequency: 20-30 Hz.
Avoid using water and air as blood vessels may be opened by the emission pressure, and the visibility in the operating area may result obfuscated.
These procedures do not need of sutur/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


