Cements
Objectives
After reading this chapter, the student should be able to:
Cementation
1. List the uses of each type of cement.
< ?mpslid E1?>< ?mpslid S2?>
2. List the components of each type of cement, and indicate their function.
< ?mpslid E2?>< ?mpslid S3?>
< ?mpslid E3?>< ?mpslid S4?>
< ?mpslid E4?>< ?mpslid S5?>
5. Describe the biocompatibility of each type of cement.
< ?mpslid E5?>< ?mpslid S6?>
6. Describe the manipulation of each type of cement.
< ?mpslid E6?>
Bases, Cavity Liners, and Varnishes
1. Compare the uses of high-strength bases, low-strength bases, cavity liners, and varnishes.
< ?mpslid E7?>< ?mpslid S8?>
2. List the components in high-strength bases, low-strength bases, cavity liners, and varnishes.
< ?mpslid E8?>< ?mpslid S9?>
< ?mpslid E9?>< ?mpslid S10?>
< ?mpslid E10?>< ?mpslid S11?>
5. Describe the manipulation of high-strength bases, low-strength bases, cavity liners, and varnishes.
< ?mpslid E11?>
Special Applications of Cement
< ?mpslid E12?>< ?mpslid S13?>
2. Describe clinically important properties of cements used for special applications.
< ?mpslid E13?>
Key Terms
All-ceramic
Chelate
Dentinal tubules
Leached
Luting
Methyl methacrylate
Obtundent
Poly(methyl methacrylate)
Pulp capping
Silicone
Temporary filling
Cementation
Cements are generally hard, brittle materials formed when a powdered oxide or glass is mixed with a liquid. When mixed to a cementing consistency, dental cements are used to retain restorations such as alloy or ceramic crowns and bridges and esthetic inlays, onlays, and veneers. When mixed to a thicker consistency, some cements can be used as temporary filling materials or to provide thermal insulation and mechanical support to teeth restored with other materials, such as amalgam, composites, or gold. Cements classified as low-strength bases or liners provide protection to the pulp from irritants or serve therapeutically as pulp-capping agents. Varnishes are not cements but are used with cements to provide pulpal protection from irritants. Other cements are used for special purposes in endodontics and orthodontics. Cements are classified according to function in Table 7-1.
TABLE 7-1
Summary of Uses of Cements in Restorative Dentistry
< ?comst?>
| FUNCTIONS | CEMENTS |
| Cementation of cast alloy crowns and bridges | Adhesive resin (dual cured), glass ionomer, resin-modified glass ionomer, self-adhesive resin, zinc polycarboxylate |
| Cementation of all-ceramic or indirect composite inlays and onlays | Adhesive resin (dual cured) |
| Cementation of zirconia-based, all-ceramic crowns and bridges | Adhesive resin (dual cured), glass ionomer, resin-modified glass ionomer, self-adhesive resin |
| Cementation of ceramic veneers | Esthetic resin (dual cured or light cured) |
| Cementation of resin-bonded bridges | Adhesive resin (dual cured) |
| Temporary cementation of cast crowns and bridges and cementation of temporary restorations | Zinc oxide–noneugenol, temporary resin |
| High-strength bases | Compomer, glass ionomer, resin-modified glass ionomer, zinc polycarboxylate |
| Temporary fillings | Resin-modified glass ionomer, zinc polycarboxylate |
| Low-strength bases | Calcium hydroxide (self-cured and light cured), glass ionomer, resin-modified glass ionomer |
| Liners | Calcium hydroxide in a suspension |
| Varnishes | Resin in a solvent |
| Special Applications | |
| Cementation of orthodontic bands | Glass ionomer, resin-modified glass ionomer, resin |
| Direct bonding of orthodontic brackets | Resin |
| Root canal sealer | Zinc oxide–eugenol |
< ?comen?>< ?comst1?>
< ?comst1?>
< ?comen1?>
The retention of restorations on prepared teeth is a major function of dental cements. Long-term cementation is required for permanent restorations such as crowns and bridges (see Figures 1-6 and 1-8). Strong cements—such as compomer, glass ionomer, hybrid ionomer, reinforced zinc oxide–eugenol, zinc phosphate, zinc polycarboxylate, or resin-based cement—are used for permanent cementation. Often, a bridge must be cemented temporarily to allow adjustments in fit, occlusion, and esthetics, or temporary restorations, such as aluminum, acrylic, or composite provisional restorations, must be cemented for 4 to 8 weeks until the permanent restoration is ready. In these cases, zinc oxide–eugenol, zinc oxide–noneugenol, or temporary resin cements are used because of their low strength and good handling characteristics. Cements are classified as water-based, oil-based, or resin-based products. More information on zinc phosphate and compomer cements can be found in Appendix 7-1.
Water-Based Cements
Glass Ionomer Cement
Glass ionomer cements are water-based cements used for final cementation of primarily alloy crowns and bridges. A restorative material with a thicker consistency is used for Class V restorations as described in Chapter 4. Examples of commercial products are listed in Table 7-2.
TABLE 7-2
Examples of Traditional Cements Suitable for Final Cementation of Cast Alloy Crowns and Bridges
| CEMENT | PRODUCT | MANUFACTURER |
| Glass ionomer | GC Fuji I | GC America (Alsip, IL) |
| GC Fuji I Capsule | GC America | |
| Ketac-Cem Maxicap | 3M ESPE (St. Paul, MN) | |
| Resin-modified glass ionomer | GC FujiCEM Automix | GC America |
| GC Fuji Plus Capsule | GC America | |
| RelyX Luting Plus Cement | 3M ESPE | |
| Zinc polycarboxylate | Durelon Maxicap | 3M ESPE |
| Hy-Bond | Shofu Dental |
Composition and Reaction
The cement powder is a finely ground aluminosilicate glass, and the viscous liquid is a polycarboxylate copolymer in water. One product (Ketac-Cem Aplicap; 3M ESPE, St. Paul, Minnesota) supplies a powder coated with polyacrylic acid copolymers. It is mixed with a low-viscosity liquid to form the cement. The components of glass ionomer cements react to form a cross-linked gel matrix that surrounds the partially reacted powder particles. Chelation between the polycarboxylate molecules and calcium on the surface of the tooth results in a chemical (adhesive) bond.
Properties
The mechanical properties of glass ionomer cement are compared with other high-strength cements in Table 7-3. Minimum requirements are described by the American National Standards Institute–American Dental Association (ANSI-ADA) Specification No. 96 (ISO 9917 [2000]). The values for compressive and tensile strengths of glass ionomer cements are similar to those of compomer, hybrid ionomer, and zinc phosphate cements. The cement has the nonirritating qualities of zinc polycarboxylate cements. However, a calcium hydroxide base is recommended for pulpal protection when the ionomer cement is used in a deep cavity. Because of fluoride incorporated in the powder, the cement has an anticariogenic effect as it is leached out. Retention of glass ionomer cements is primarily micromechanical, although some chemical bonding occurs.
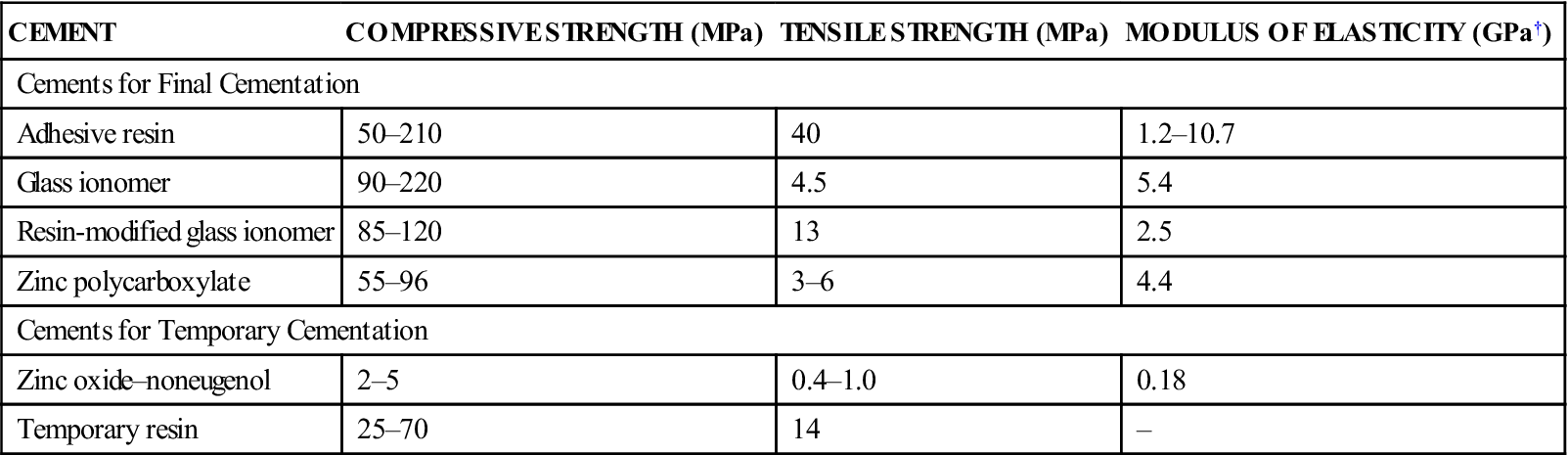
TABLE 7-3
Mechanical Properties of Cements for Final and Temporary Cementation∗
< ?comst?>
| CEMENT | COMPRESSIVE STRENGTH (MPa) | TENSILE STRENGTH (MPa) | MODULUS OF ELASTICITY (GPa†) |
| Cements for Final Cementation | |||
| Adhesive resin | 50–210 | 40 | 1.2–10.7 |
| Glass ionomer | 90–220 | 4.5 | 5.4 |
| Resin-modified glass ionomer | 85–120 | 13 | 2.5 |
| Zinc polycarboxylate | 55–96 | 3–6 | 4.4 |
| Cements for Temporary Cementation | |||
| Zinc oxide–noneugenol | 2–5 | 0.4–1.0 | 0.18 |
| Temporary resin | 25–70 | 14 | – |
< ?comen?>< ?comst1?>
< ?comst1?>
< ?comen1?>
< ?comst1?>< ?comen1?>∗< ?comst1?>< ?comen1?>Properties measured at 24 hours.
< ?comst1?>< ?comen1?>†< ?comst1?>< ?comen1?>GPa = 1000 MPa.
Manipulation
Glass ionomer cements are powder–liquid systems packaged in bottles or capsules. The bottle of powder should be tumbled gently before dispensing. The powder and liquid are dispensed onto a paper pad or glass slab. The powder is divided into two equal portions. The first portion of powder is mixed with a stiff spatula into the liquid before the next portion is added. The mixing time should be 30 to 60 seconds, depending on the product. The cement is used immediately because the working time after mixing is about 2 minutes at 22 °C. Cooling the mixing slab slows the setting reaction and provides additional working time. The cement should not be used once a “skin” forms on the surface or when the consistency becomes noticeably thicker. During application, contact with water should be avoided; the field is isolated completely. The cement sets in the mouth in about 7 minutes from the start of mixing. Encapsulated products (GC Fuji I Capsule, GC America, Alsip, Illinois; Ketac-Cem Aplicaps, 3M ESPE, St. Paul, Minnesota) require mechanical mixing for 10 seconds.
Hybrid Ionomer Cement
Hybrid ionomer cements (resin-modified glass ionomer cements) are water-based cements indicated for permanent cementation of alloy crowns and bridges to tooth structure and core buildups, cementation of posts, and bonding of orthodontic appliances. Some products are not recommended for cementation of lower-strength all-ceramic inlays, onlays, or crowns because of their water sorption, which leads to expansion and can cause cracking of the ceramic restoration. Hybrid ionomer cements can be used for cementation of zirconia-based all-ceramic crowns and bridges. Examples of commercial products are listed in Table 7-2.
Composition and Reaction
One cement powder contains a radiopaque, fluoroaluminosilicate glass and a microencapsulated catalyst system. The liquid is an aqueous solution of polycarboxylic acid modified with pendant methacrylate groups. It also contains 2-hydroxyethyl methacrylate (HEMA) and tartaric acid. Curing results from an acid–base glass ionomer reaction and self-cured polymerization of the methacrylate groups.
Properties
The values for compressive and tensile strengths of hybrid ionomer cements are similar to those of glass ionomer cements (see Table 7-3). Hybrid ionomer cements have no measurable solubility when tested by lactic acid erosion. The fracture toughness is higher than that of other water-based cements but lower than that of resin cements. Fluoride release is similar to the glass ionomer cements. The early pH is about 3.5 and gradually rises. These cements have minimal postoperative sensitivity. The bond strength to moist dentin is 14 MPa and is much higher than that of most water-based cements.
Manipulation
Hybrid ionomer cements are powder–liquid systems packaged in bottles or capsules or paste–paste systems. With the powder–liquid products, the powder is fluffed before dispensing. The liquid should be dispensed by holding the vial vertically to the mixing pad. The powder is incorporated into the liquid within 30 seconds to give a mousse-like consistency. The working time is 2.5 minutes. The cement is applied to a clean, dry tooth that is not desiccated. No coating agent is needed. HEMA is a known contact allergen; therefore, protective gloves and a no-touch technique are mandatory. A paste–paste product (GC FujiCEM; GC America, Alsip, Illinois) is packaged in an auto-mixed dispenser (Figure 7-1).
Zinc Polycarboxylate Cements
Zinc polycarboxylate cements are water-based cements used as final cements for retention of crowns and bridges. They are not as strong as zinc phosphate cements, but they are less irritating to the pulp. Examples of commercial products are listed in Table 7-2.
Composition and Reaction
Zinc polycarboxylate cements are supplied usually as a powder and a liquid. The powder is mainly zinc oxide, and the liquid is a viscous solution of polyacrylic acid in water. One product (Tylok Plus; DENTSPLY Caulk, Milford, Delaware) is supplied as a powder to be mixed with tap water. Its powder consists of zinc oxide coated with solid polyacrylic acid. The zinc oxide and the polyacrylic acid react to form a zinc polyacrylate that surrounds the partially reacted zinc oxide powder particles. The reaction is accelerated by heat.
Properties
The important properties of zinc polycarboxylate cements are moderate viscosity, moderate strength, ability to bond to enamel, and mild acidity. Minimum requirements are described in ANSI-ADA Specification No. 96 (ISO 9917 [2000]).
Mixed polycarboxylate cement appears to be too viscous (thick), but it flows readily when applied to the surfaces to be cemented. The compressive strength of polycarboxylate cement (see Table 7-3) is less than that of glass ionomer cement; however, it provides clinically satisfactory retention for well-fitting restorations.
Zinc polycarboxylate cements are slightly acidic (low pH) when first mixed, but the acid is weakly dissociated. Histologic reactions are similar to those of zinc oxide–eugenol cements, but more reparative dentin is observed with the polycarboxylates. Polycarboxylate cements bond well to sandblasted gold alloys, although clinical studies have not demonstrated improved retention with these cements.
Manipulation
The powder bottle is shaken gently. The powder is dispensed with a scoop onto a disposable paper pad or a glass slab, which can be cooled to permit a longer working time. The viscous liquid is dispensed from the dropper bottle in uniform drops. About 90% of the powder is added immediately to the liquid and mixed for 30 to 60 seconds, depending on the product. The remainder of the powder is added to adjust the consistency. A small area of the mixing surface is used, and the cement is mixed with a stiff spatula. The proper consistency is creamy. The cement should be used immediately because the working time is short (about 3 minutes after mixing at 22 °C). The cement is no longer usable when it loses its luster and becomes stringy or starts to “cobweb.” Durelon Maxicap (3M ESPE, St. Paul, MN) is supplied in unit-dose capsules.
Oil-Based Cements
Zinc Oxide–Eugenol Cements
Zinc oxide–eugenol cements are oil-based cements that have an obtundent (sedative) effect on the pulp and are especially useful for cementation on prepared teeth with exposed dentinal tubules. The addition of reinforcing agents to zinc oxide–eugenol cement has resulted in permanent luting cements. Temporary cements are not so strong but are useful for short-term cementation of temporary stainless steel crowns and permanent restorations. Zinc oxide–noneugenol cements (see Table 7-4) are also oil-based cements used for short-term cementation of temporary acrylic crowns and completed cast restorations. They are weak and easily cleaned from the casting.
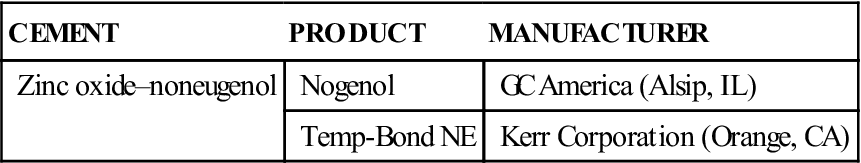
TABLE 7-4
< ?comst?>
| CEMENT | PRODUCT | MANUFACTURER |
| Zinc oxide–noneugenol | Nogenol | GC America (Alsip, IL) |
| Temp-Bond NE | Kerr Corporation (Orange, CA) |
< ?comen?>< ?comst1?>
< ?comst1?>
< ?comen1?>
Composition and Reaction
The zinc oxide–eugenol cement powder (Type I) contains zinc oxide (69%); rosin (29%) to reduce brittleness; and zinc acetate, an accelerator. The liquid is eugenol or a mixture of eugenol and other oils. The powder reacts with the eugenol in the presence of moisture to form an amorphous chelate of zinc eugenolate. The zinc oxide–noneugenol cements (Type I) are formulated with oils other than eugenol for patients sensitive to eugenol.
The polymer-reinforced zinc oxide–eugenol cements (Type II) contain 80% zinc oxide and 20% acrylic resin in the powder and eugenol in the liquid. The ethoxybenzoic acid (EBA)-alumina–reinforced cements contain 70% zinc oxide and 30% alumina in the powder. The liquid is 62.5% EBA and 37.5% eugenol. The EBA in the liquid promotes the formation of a stronger, crystalline matrix. Water and heat accelerate the setting reaction of these cements.
Properties
The moderate strength and low acidic quality of the zinc oxide–eugenol cements are important properties. Minimum requirements are described by ANSI-ADA Specification No. 30 (ISO 3107 [2000]).
The compressive strengths of the permanent and temporary zinc oxide–eugenol cements are listed in Table 7-3. The permanent zinc oxide–eugenol cements are not as strong as other cements but have been shown to be clinically successful for final cementation of crowns and bridges that have good retention. The temporary cements are weaker, a desirable feature for cementation of temporary crowns or for temporary cementation of completed crown and bridge restorations that must be removed easily.
The pH of the zinc oxide–eugenol cements is neutral. Because of the sedative nature of these cements, they do not require a protective varnish or cavity liner. Retention is the result of micro-mechanical interlocking of the restoration and prepared tooth.
Manipulation
The permanent zinc oxide–eugenol cements (Type II) are powder–liquid systems. The powder bottle is shaken gently, then the powder is dispensed with the supplied scoop and the liquid with a dropper. It is mixed on a glass slab or treated paper pad with a metal spatula. The powder is incorporated into the liquid all at once and mixed for 30 seconds. The mix initially is like putty, but continued mixing for 30 seconds more causes the polymer-reinforced cement mix to become fluid. The EBA-alumina–reinforced cement should be stropped for 60 seconds with broad strokes of the spatula after the initial 30-second mixing to obtain a suitable consistency. The working time of the EBA-alumina cements is long (about 22 minutes), unless moisture is present on the slab. In the mouth, the zinc oxide–eugenol cements set quickly because of the moisture and heat.
The temporary cements (Type I) are typically two-paste systems. Equal lengths of the accelerator and base pastes are dispensed on a paper pad or glass slab. The pastes are colored differently. Mixing is continued until a uniform color is achieved. Some products are available in unit-dose packages.
Zinc oxide–eugenol cements are difficult to remove from the tissues and mixing surfaces after setting. The patient’s lips and adjacent teeth are coated with silicone grease before application of the cement. Glass slabs and spatulas are wiped clean before the cement sets. Oil of orange is a solvent useful in removal of set cement.
Resin-Based Cements
Esthetic Resin Cements
Esthetic resin cements are tooth-colored or translucent resins available in a variety of shades, and they are used for bonding of all-ceramic and indirect composite restorations. These cements require a bonding agent for adhesion to tooth structure and separate primers for bonding to ceramic substrates. Examples of commercial products are listed in Table 7-5.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses