Chapter 6
Gingival Enlargements – Generalised
Aim
This chapter aims to provide an overview of the causes, features and management of generalised gingival swellings, which involve the free and/or attached gingiva and may also extend to the non-keratinised lining oral mucosa.
Outcome
Having read this chapter, the clinician should be able to formulate an appropriate differential diagnosis for generalised gingival enlargements, know which additional clinical investigations to perform to arrive at a definitive diagnosis and be aware of key management strategies either within their practice or in a specialist environment.
Table 6-1 lists the generalised gingival enlargements discussed in this chapter and highlights which are common and uncommon and when the condition may be managed in general practice or should be referred.
| Appearance/ Character | Category | Sub-Category | Incidence | Manage/ Refer |
| Fibrous enlargements | Hereditary gingival fibromatosis | uncommon | manage/ refer | |
| Drug-induced gingival overgrowth | Dilantins (e.g. Phenytoin) | common (13–15% of medicated patients) | manage/refer | |
| Calcium channel blocking drugs (e.g. nifedepine amlodipine felodipine) | common (10–15% of medicated patients) | manage/refer | ||
| Ciclosporin | common (30% of medicated patients) | manage/refer | ||
| Appliance-induced hyperplasia | common | manage | ||
| Delayed gingival retreat | common | manage | ||
| Mucopolysaccharidoses | rare | refer | ||
| Mannosidosis | rare | refer | ||
| Oedematous enlargements | Inflammatory gingival enlargement | Plaque-induced | common | manage |
| Hormonal influence | common | manage | ||
| Hereditary angioedema | uncommon | refer | ||
| Acquired angioedema | uncommon | refer | ||
| Granulomatous enlargements | Sarcoidosis | uncommon | refer | |
| Crohn’s Disease | uncommon | refer | ||
| Orofacial Granulomatosis | uncommon | refer | ||
| Exophytic swellings | Leukaemia | Acute: | ||
| monocytic | uncommon | refer | ||
| myelomonocytic | uncommon | refer | ||
| myeloid | uncommon | refer | ||
| lymphocytic | uncommon | refer | ||
| Pyostomatitis vegetans | uncommon | refer | ||
| Wegener’s granulomatosis | uncommon | refer | ||
| Plasmacytoma | uncommon | refer | ||
| Amyloidosis | uncommon | refer | ||
| Multiple myeloma | uncommon | refer |
Terminology
A variety of terms has been, and is, used to describe generalised gingival enlargements, which can give rise to confusion. These are summarised below:
Gingival hyperplasia – ‘hyperplasia’ is a term that describes tissue enlargement arising from an increase in number of one or more constituent cell types. Hyperplasia can therefore be singular (e.g. connective tissue hyperplasia) or compound (e.g. fibro-epithelial hyperplasia). The term gingival hyperplasia has thus become outdated because it poorly describes the true nature of the swelling.
Gingival hypertrophy – ‘hypertrophy’ describes an increase in tissue size due to an increase in the size of one or more constituent cell types. This can also be singular or compound.
Gingival overgrowth – ‘overgrowth’ is a term used to overcome some of the shortcomings of the above two terms, because it is less specific. The advantage of this term is that it allows for part of, or the entire enlargement to be due to increased production of connective tissue matrix or collagen fibre deposition. ‘Overgrowth’ is usually used in association with drug-induced enlargements that are histologically complex.
Gingival enlargement – the term ‘enlargement’ is used in this chapter, because not all gingival enlargements are true swellings (e.g. delayed gingival retreat), but they do appear clinically as enlarged tissues.
Fibrous Swellings
Hereditary Gingival Fibromatosis (HGF)
Clinical appearance
HGF presents as a generalised pink, firm and often stippled enlargement of the free and attached gingivae, extending to the mucogingival junction (see Chapple and Gilbert, 2002) buccally extending variably into the palate. Classically it affects maxillary tuberosities (Fig 6-1) and retromolar regions of the mandible (Fig 6-2). However, the labial gingivae may be involved (Fig 6-3), and it is important to distinguish this from delayed gingival retreat if planning surgical recontouring. The fibrosis is slowly progressive and may give rise to tooth movement and spacing, or may delay or prevent tooth eruption.
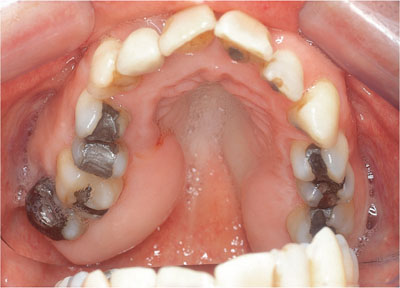
Fig 6-1 Hereditary gingival fibromatosis classically affecting the maxillary tuberosities, where false pocketing had led to early periodontal attachment loss.
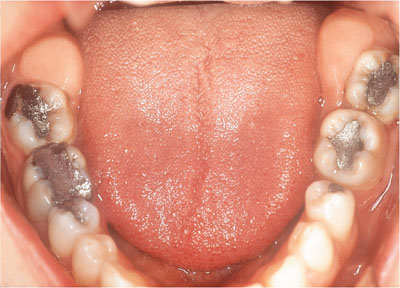
Fig 6-2 Hereditary gingival fibromatosis classically affecting the mandibular retromolar region.
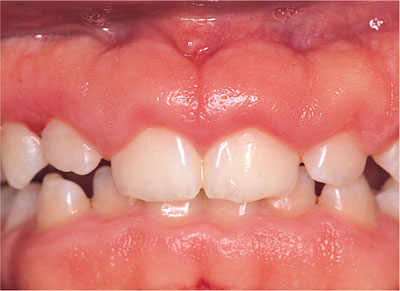
Fig 6-3 Hereditary gingivo-fibromatosis of the labial gingivae in a 12-year- old boy, whose mother, aunt and younger brother were also affected.
Clinical symptoms
The patient’s main concerns will be:
-
Aesthetic.
-
Functional – tissue can overgrow the crowns of teeth if severe. Also teeth may be moved to a position that interferes with normal occlusal function.
-
Discomfort – can arise where fibrosis is actively moving teeth.
Aetiology
Unknown, but two forms are described:
-
Familial HGF – autosomal dominant inheritance recently linked (Hart et al, 1998) to chromosome 2p21 (position 21 of short arm of chromosome 2). Penetrance may be incomplete, i.e. the condition may not always be expressed phenotypically or may vary in severity.
-
Sporadic HGF – a controversial diagnosis that may be autosomal dominant or recessive. It may simply be a familial form that has variable clinical expression, incomplete penetrance or may arise due to a spontaneous mutation within the 2p21 region.
Involvement of non-gingival sites
Gingival fibromatosis is also associated with various rare syndromes:
-
Rutherford syndrome (juvenile hyaline fibrosis, corneal dystrophy, neurosensory hearing loss) – autosomal dominant inheritance.
-
Laband syndrome (nail, ear, nose and bone defects, syndactyly) – autosomal recessive/dominant or spontaneous mutations.
-
Cross syndrome (hypopigmentation, microphthalmia, athetosis) – autosomal recessive.
-
Ramon syndrome (hypertrichosis [excessive hair growth], cherubism, mental retardation) – autosomal recessive.
Hypertrichosis, mental retardation, epilepsy and growth hormone defects are also described by Gorlin et al (1976), and interestingly hypertrichosis and gingival fibrosis are both complications of ciclosporin medication (see below).
Differential diagnosis
-
Delayed gingival retreat.
-
Drug-induced gingival overgrowth.
-
Plaque-induced chronic inflammatory enlargement.
Clinical investigation
HGF is a presumptive diagnosis based on a careful history and clinical findings. Definitive diagnosis currently requires confirmatory histopathology in addition to clinical findings. Excision biopsy is usually by conventional open-faced gingivectomy (Fig 6-4, 6-5), but if severe, an inverse bevel flap may be required (Fig 6-6) with significant undermining by sharp dissection and filleting ± a distal wedge procedure (Fig 6-7).
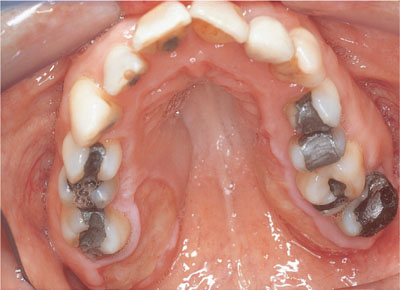
Fig 6-4a HGF from the patient in Fig 6-1, immediately post-open-face gingivectomy.
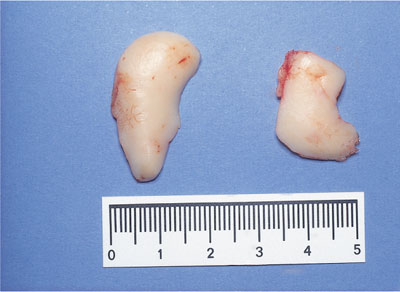
Fig 6-4b The tissue excised from the patient in Fig 6-4a.
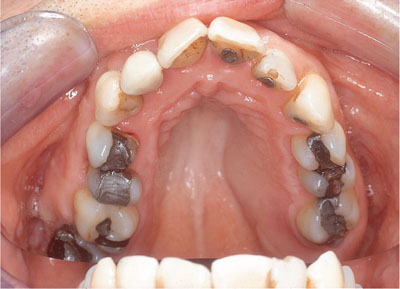
Fig 6-5 The healed tuberosities from 6-4a, two weeks post-surgery. False pocketing has been eliminated. Note the improved angle between the gingival margin UL6 and the tooth, facilitating ease of plaque control.
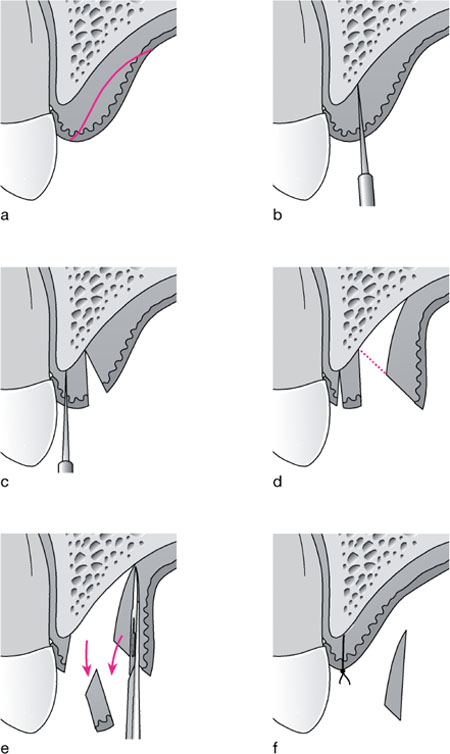
Fig 6-6 Diagram to illustrate the inverse bevel incision prior to flap reflection and ‘filleting’ of the bulk of the sub-mucosal connective tissue.
-
Bulky tissue prior to split-thickness flap reflection.
-
First incision is parallel to a sulcus incision, using an inverse bevel.
-
Second incision isolates a tissue ‘cuff.
-
Split-thickness flap is then raised by sharp dissection allowing surgical access to the underside of the flap.
-
Cuff is removed and underside of flap is debulked by sharp excision of excess connective tissue. Care must be taken to avoid puncturing the flap.
-
Flap is closed after any necessary debridement and removal of the bulky connective tissue from the underside of the flap.
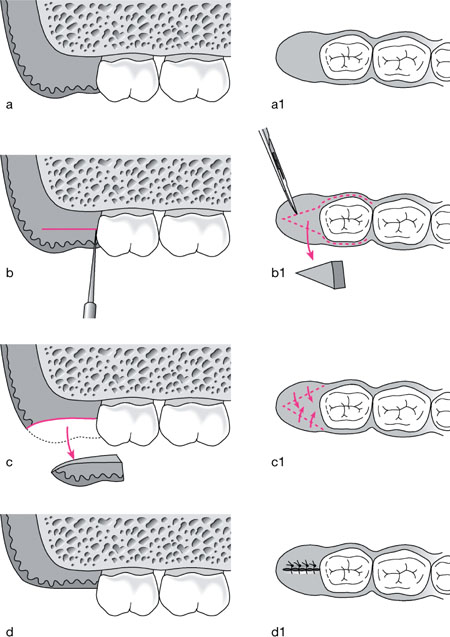
Fig 6-7 Diagram to illustrate the distal wedge procedure. There are several variants, but the most basic literally involves the sharp dissection of a wedge of tissue from the tuberosity/retro-molar region.
a. Enlarged tuberosity with false pocketing distal to UR7.
a1. Occlusal view of ‘a’.
b. A buccal and lingual/palatal incision is made down to the alveolar crest
b1. The wedge is delineated by the dotted lines and buccal and palatal flaps are raised a short distance prior to wedge removal.
c. Wedge is sharp-dissected out.
c1. Buccal and lingual flaps are approximated over ‘dead space’ and in doing so this space is eliminated and the tissue level falls.
d. View of closed defect with tissue level more apically positioned.
d1. Occlusal view with distal wedge removal complete and flaps sutured.
Management
If mild and symptom-free, simply monitor. However, probing is essential to distinguish between true and false pocketing (Fig 6-8). Surgical reduction is likely to require specialist skills for tuberosity and retromolar lesions, as feeder arteries can arise within, and the sublingual space may be compromised during lower molar surgery. Slow recurrence is likely.
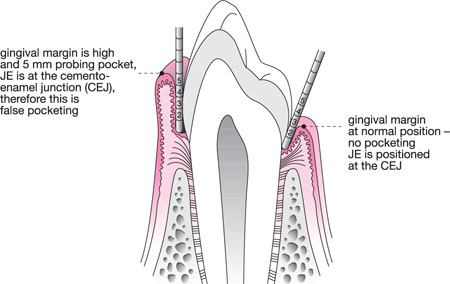
Fig 6-8 Schematic longitudinal section of a premolar and associated periodontal tissues demonstrating a healthy sulcus and false pocketing.
Drug-induced Gingival Overgrowth (DIGO)
Clinical appearance
The appearance of DIGO is variable. Classically the enlargement starts at the interdental papilla and spreads to involve the marginal gingivae. The anterior gingivae are most commonly affected, but posterior enlargement may lead to occlusal surface coverage in severe cases (Fig 6-9). With phenytoin and ciclosporin, in the presence of good plaque control, overgrowth is firm, fibrous and pink (Fig 6-10), but where plaque control is poor it becomes more vascular (Fig 6-11). Overgrowth associated with calcium channel blocking drugs (CCBs) tends to be more vascular (Fig 6-12) and is associated with more severe enlargement with concurrent ciclosporin. Surface keratosis or ulceration may arise following trauma from opposing teeth.
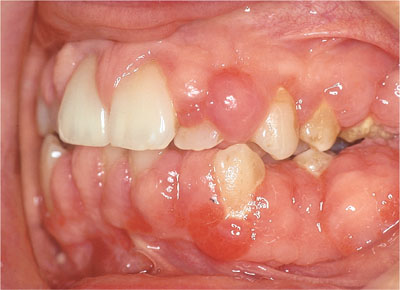
Fig 6-9 Drug-induced gingival overgrowth in a renal transplant patient medicated with ciclosporin. Molar teeth were covered making mastication uncomfortable.
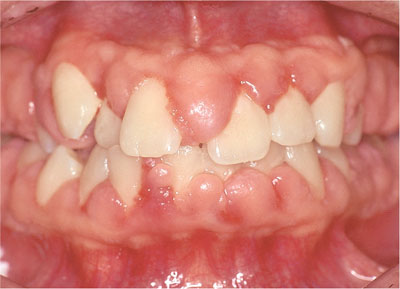
Fig 6-10 Fibrous overgrowth in a patient medicated with ciclosporin.
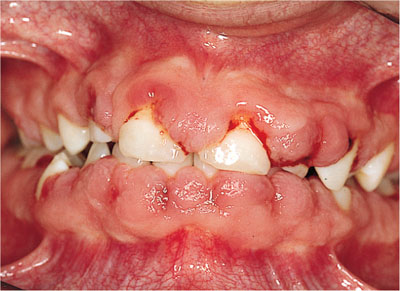
Fig 6-11 DIGO with a more vascular inflammatory component due to poor plaque control. (Fig 7-5 book 11).
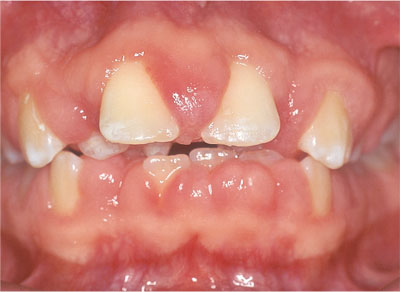
Fig 6-12 Fibro-vascular overgrowth in a patient medicated with ciclosporin and nifedepine.
Clinical symptoms
As for HGF.
Aetiology
DIGO is associated classically with three drug types, but has also been reported with the oral contraceptive, cannabis (Rees, 1992), erythromycin (Valsecchi and Cainelli, 1992) and sodium valproate (Syrjanen & Sryjanen 1979).
-
Calcium channel blocking (CCB) drugs – nifedepine, amlodipine, felodipine, diltiazem hydrochloride.
-
Dilantins – phenytoin
-
Ciclosporin.
CCBs are used to control hypertension, ciclosporin is an immunosuppressive agent used to modulate allograft rejection or in severe erosive mucosal disease (Chapter 8). Phenytoin is an anti-convulsant drug. While there are some common features to their modes of action at an ionic level, the aetiology of DIGO remains poorly understood and complex. Seymour et al (2000) have described a number of risk factors:
|
|
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


