Dental Amalgam
Objectives
After reading this chapter, the student should be able to:
1. Explain the clinical advantages and disadvantages of using spherical or admixed types of amalgam.
< ?mpslid E1?>< ?mpslid S2?>
2. Describe precapsulated amalgam, and explain why its use is mandatory today.
< ?mpslid E2?>< ?mpslid S3?>
< ?mpslid E3?>< ?mpslid S4?>
< ?mpslid E4?>< ?mpslid S5?>
< ?mpslid E5?>< ?mpslid S6?>
< ?mpslid E6?>< ?mpslid S7?>
7. Understand the rationale for limiting the patient’s and dental personnel’s exposure to mercury.
< ?mpslid E7?>< ?mpslid S8?>
< ?mpslid E8?>
Key Terms
Admixed amalgam
Amalgam
Amalgamation
Condensation
Creep
Dimensional change
Gamma-2 (γ2)
Matrix
Precapsulated
Silver alloy
Spherical amalgam
Trituration
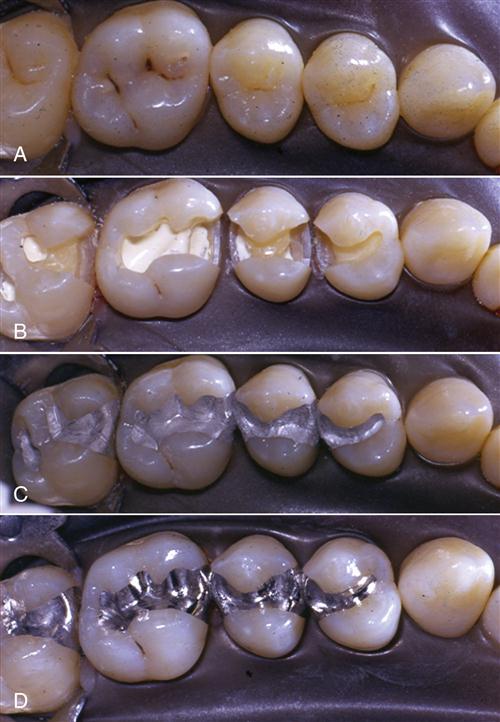
In general, an amalgam is a mixture of a metallic element with the liquid element mercury. Dental amalgam is a mixture of a silver alloy with mercury. When the silver alloy, which is a powder composed mostly of silver, copper, and tin, is mixed with mercury, a chemical reaction ensues. For 1 to 2 minutes after mixing, dental amalgam has a putty-like consistency, but it progresses to a carvable consistency for an additional 2 to 4 minutes. During this time, the amalgam is packed into a cavity preparation in a tooth and carved to the desired shape (Figure 5-1). In the following minutes to hours, the amalgam reaction proceeds, reaching maturity and full strength in about 24 hours.
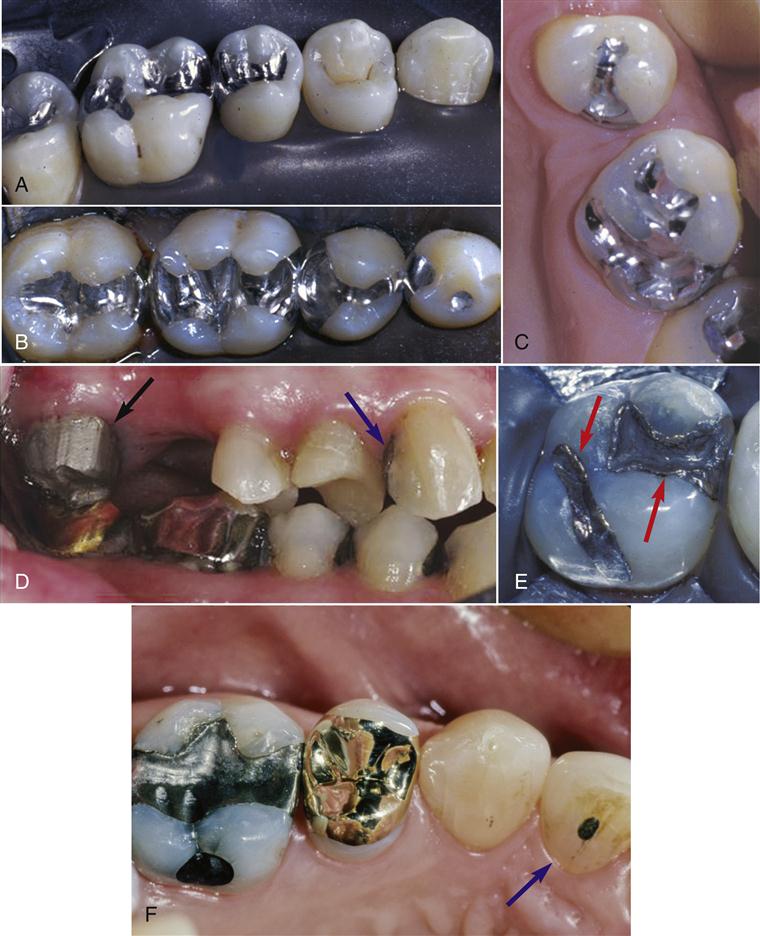
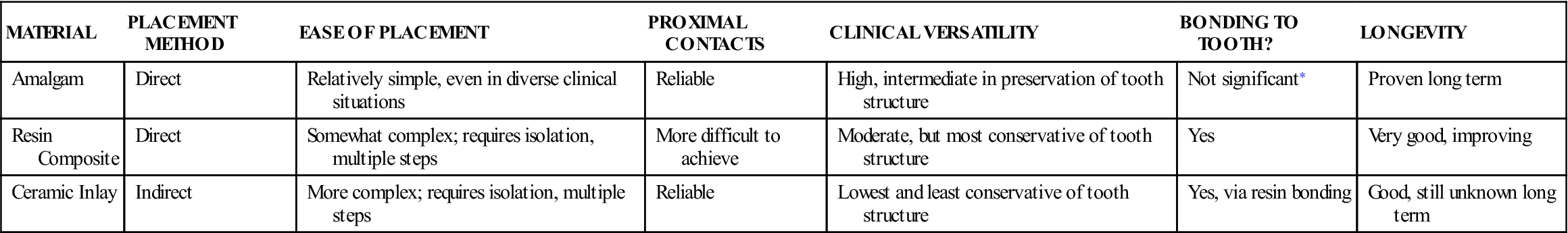
On a global scale, amalgam restorations still account for a significant portion of all dental restorations, although many modern dental practices today use amalgam sparingly. Prior to the development of tooth-colored restorations, amalgams were used to restore teeth throughout the mouth. However, because of their gray color, today these restorations are limited to the posterior teeth where esthetics is less of a concern (Figure 5-2). More esthetic ceramic or direct resin composite materials are now used in situations where esthetics is important. However, these alternative materials have problems such as expense, shorter longevity, and technique sensitivity in placement. Thus, amalgam remains a viable clinical choice where longevity, ease of placement, and clinical performance are paramount, especially when clinical conditions are challenging (Table 5-1). Concerns over mercury toxicity from amalgams remain despite no credible evidence that adverse health effects stem from the mercury in amalgam materials. Concerns about the mercury load on the environment from dental sources also remain.
TABLE 5-1
Dental Amalgam versus Esthetic Alternatives
< ?comst?>
| MATERIAL | PLACEMENT METHOD | EASE OF PLACEMENT | PROXIMAL CONTACTS | CLINICAL VERSATILITY | BONDING TO TOOTH? | LONGEVITY |
| Amalgam | Direct | Relatively simple, even in diverse clinical situations | Reliable | High, intermediate in preservation of tooth structure | Not significant∗ | Proven long term |
| Resin Composite | Direct | Somewhat complex; requires isolation, multiple steps | More difficult to achieve | Moderate, but most conservative of tooth structure | Yes | Very good, improving |
| Ceramic Inlay | Indirect | More complex; requires isolation, multiple steps | Reliable | Lowest and least conservative of tooth structure | Yes, via resin bonding | Good, still unknown long term |
< ?comen?>< ?comst1?>
< ?comst1?>
< ?comen1?>
< ?comst1?>< ?comen1?>∗< ?comst1?>< ?comen1?>Studies with amalgam bonding have been done, but evidence suggests that bond strength is far less than for composites or ceramics, and it is marginally useful clinically.
Silver Alloys for Dental Amalgams
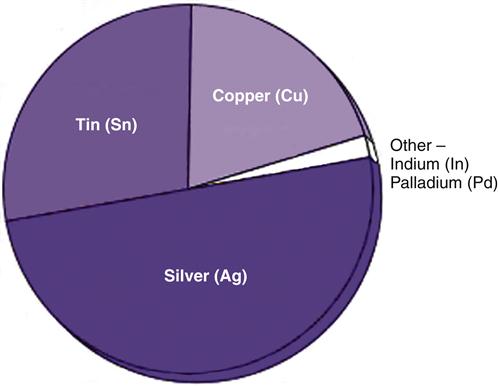
Historically, the silver alloy used in dental amalgams had low (2 to 4 wt %) amounts of copper. These alloys were combined with mercury to form “low-copper amalgam.” However, the development of silver alloys with higher (13% to 30%, generally about 20%) copper (Figure 5-3) has replaced low-copper alloys because high-copper alloys produce amalgams with higher strength, less corrosion, less creep, and better longevity at the margins (edges of the cavity preparation). Other elements such as palladium and indium are sometimes included to reduce corrosion. Today’s high-copper silver alloys are essentially free of zinc (<0.01 wt % per ADA Specification No. 1) because zinc causes a significant, long-term, and clinically unacceptable expansion of the amalgam if it is contaminated with saliva or other moisture during placement.
Particles of the silver alloy powder can be either irregularly shaped, spherical, or a mixture of the two (Figure 5-4). The shape of these particles will significantly influence the setting reaction and manipulation of the amalgam. If a mixture of particles is used, the resulting amalgam is referred to as an admixed amalgam. Amalgams containing irregular particles with or without spherical particles added require greater packing or condensation forces during placement than amalgams with spherical particles alone. Most practitioners feel that amalgams containing irregular particles produce better proximal contacts and are easier to carve. Spherical amalgams, containing only spherical alloy particles, require less mercury and set somewhat faster. Practitioners generally select either a spherical or admixed amalgam, depending on the clinical situation. The importance of proper manipulation of each type cannot be overemphasized. For example, if a condensation force that is appropriate for a spherical amalgam is applied to an admixed amalgam, the restoration will likely contain voids and lack adequate proximal contacts.
Mercury
Mercury is a dense metal (density = 13.5 g/mL) and the only metallic element in the periodic table that is a liquid at room temperature. Mercury’s liquid form facilitates mixing of mercury with the alloy particles to create a putty-like mass that is condensed into a cavity preparation prior to setting. Historically, excess mercury was added to the alloy particles and mixed with a mortar and pestle by hand until the proper consistency was achieved for placement. The excess mercury was removed by “wringing it out” with a cheesecloth. This practice was abandoned because of concerns about mercury toxicity and inconsistencies in the consistency of the putty state. Later, the powder was compressed into tablets of specific mass, and the proper amount of mercury was added with a dispensing device into a reusable mixing capsule. This new method of trituration resulted in consistently strong amalgams and reduced the exposure of the dental staff to mercury. However, the mercury in this dispensing device was exposed to air and dust particles that increased the risk of contamination leading to inferior clinical properties of the mixed amalgam. The risk of spilling significant amounts of mercury during filling or use of the device was another disadvantage.
Today, nearly all practitioners use silver alloy powder and mercury sealed into disposable hard plastic capsules (Figure 5-5). Both the mercury and alloy powder are apportioned by the manufacturer. The mercury is sequestered from the alloy powder by a plastic membrane inside the capsule that is easily ruptured during mixing. Sometimes, a small inert cylinder called a pestle is added to facilitate mixing. This precapsulated method provides the proper ratio of alloy powder to mercury, limits handling of pure mercury by the dental team, produces amalgams with consistent physical properties, and keeps the mercury clean.
Reaction of Silver Alloy with Mercury
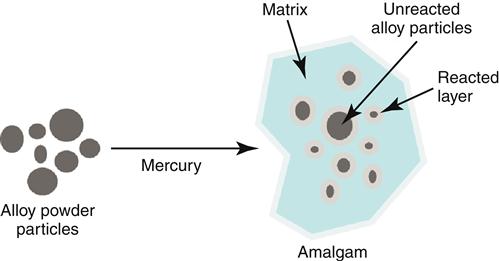
The reaction of the silver alloy with mercury is called amalgamation. When high-copper alloy particles contact the mercury, the copper, silver, tin and other elements in the alloy dissolve into the mercury much as sugar would dissolve in water (Figure 5-6). Dissolution rates vary for each element and occur from the surface of the alloy particles. Almost immediately, new solid products begin to crystallize as the dissolved elements react with the mercury. The crystallization of new products continues until all the liquid mercury is consumed, and the amalgam becomes stiffer and eventually hardens completely. The ratio of alloy to mercury is formulated so that the hardening of the amalgam occurs before all of the original alloy particles can dissolve. Thus, the set amalgam contains many of the original alloy particles surrounded by several new products of mercury with tin, copper, or silver (see Figure 5-6). The reaction products are collectively called the matrix of the amalgam. In the set mass of most contemporary amalgams, the original particles comprise more than 50% of the volume and are important to the strength and corrosion resistance of the clinical restoration.
Once the amalgamation reaction is complete, little or no mercury remains unreacted. In its reacted state, mercury is not easily released from the amalgam. This fact is important because free mercury can pose a health risk if it occurs in sufficient concentrations. In practice, minute amounts of mercury vapor (approximately 1 to 2 µg per day) are released from dental amalgams as a result of chewing. Higher release may occur during the setting reaction, during removal of old amalgams, or if the amalgam is heated above 80 °C (Figure 5-7). To further limit mercury release during the setting reaction, newer mercury-indium liquid alloys have been substituted for pure mercury in a few products.
Early amalgams were much lower in copper (2 to 4 wt %) than contemporary amalgams. In low copper amalgams, the chemical reaction produced a tin-mercury product in the matrix called gamma-2 (γ2). Gamma-2 had poor corrosion properties that significantly shortened the clinical lifetime of the amalgams. The current high-copper amalgams have eliminated the gamma-2 product; high-copper amalgams have greatly increased resistance to corrosion and marginal breakdown in clinical use.
Properties of Amalgam
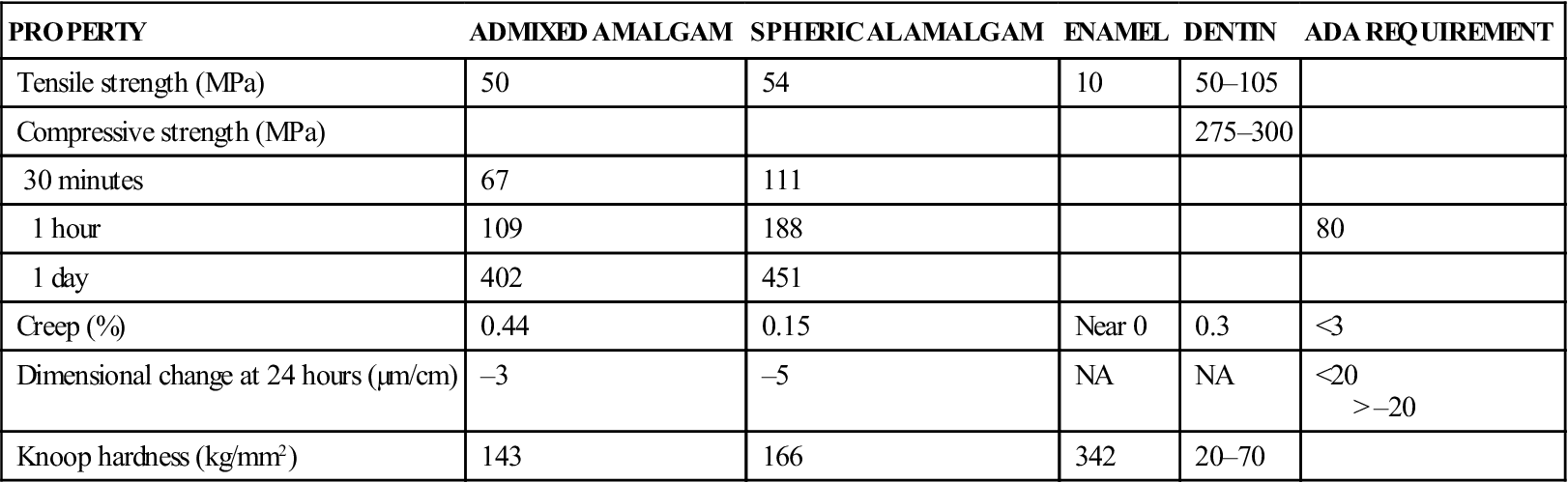
Clinically relevant properties of amalgam include strength, dimensional change, creep, tarnish, and corrosion. Some of these properties are listed for spherical and admixed high-copper amalgams in Table 5-2. Several examples of commercial products are listed in Table 5-3.
TABLE 5-2
< ?comst?>
| PROPERTY | ADMIXED AMALGAM | SPHERICAL AMALGAM | ENAMEL | DENTIN | ADA REQUIREMENT |
| Tensile strength (MPa) | 50 | 54 | 10 | 50–105 | |
| Compressive strength (MPa) | 275–300 | ||||
| 30 minutes | 67 | 111 | |||
| 1 hour | 109 | 188 | 80 | ||
| 1 day | 402 | 451 | |||
| Creep (%) | 0.44 | 0.15 | Near 0 | 0.3 | <3 |
| Dimensional change at 24 hours (µm/cm) | –3 | –5 | NA | NA | <20 > –20 |
| Knoop hardness (kg/mm2) | 143 | 166 | 342 | 20–70 |
< ?comen?>< ?comst1?>
< ?comst1?>
< ?comen1?>
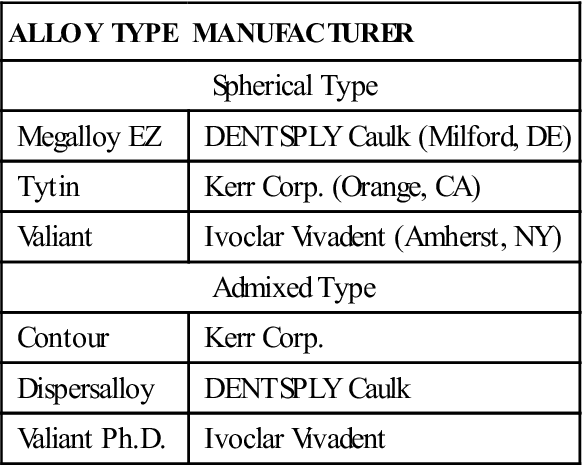
TABLE 5-3
Examples of Commercial Amalgam Restorative Materials
< ?comst?>
| ALLOY TYPE | MANUFACTURER |
| Spherical Type | |
| Megalloy EZ | DENTSPLY Caulk (Milford, DE) |
| Tytin | Kerr Corp. (Orange, CA) |
| Valiant | Ivoclar Vivadent (Amherst, NY) |
| Admixed Type | |
| Contour | Kerr Corp. |
| Dispersalloy | DENTSPLY Caulk |
| Valiant Ph.D. | Ivoclar Vivadent |
< ?comen?>< ?comst1?>
< ?comst1?>
< ?comen1?>
Strength
The strength of an amalgam must withstand oral mechanical forces. Insuff/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses