CHAPTER 49
Salivary Gland Pathology
Michael Grau, Jr.,1 Markus S. Hill,1 Billy Turley,1 Vincent Slovan,1 Christopher J. Haggerty2 and Robert M. Laughlin1
1Department of Oral and Maxillofacial Surgery, Naval Medical Center San Diego, San Diego, California, USA
2Private Practice, Lakewood Oral and Maxillofacial Surgery Specialists, Lees Summit; and Department of Oral and Maxillofacial Surgery, University of Missouri–Kansas City, Kansas City, Missouri, USA
Mucocele (Mucous Extravasation Phenomenon) Excision
Local excision of a damaged minor salivary gland and associated soft tissue.
Indications
- Soft tissue swelling associated with damage or rupture of a minor salivary gland duct
Contraindications
- Significant medical comorbidities (i.e., anticoagulated patients)
Anatomy
Mucocele: A damaged or ruptured minor salivary gland duct leading to extravasation of mucus within the surrounding soft tissue.
Technique
- Local anesthesia is administered specific to the location of the mucocele. For lower lip mucoceles, bilateral mental nerve or inferior alveolar nerve blocks are performed.
-
An elliptical excision of the mucocele, with its associated overlying mucosa and glandular tissue/damaged minor salivary gland, is performed down to the muscle layer (Figure 49.3). Care is taken to preserve the integrity of the vermillion border of the lip, to remove the entire involved minor salivary gland, and to minimize iatrogenic damage to adjacent minor salivary glands.
- Deep 4-0 Vicryl sutures are placed depending on the size and depth of the surgical defect.
- Interrupted 4-0 chromic sutures are used to reapproximate the mucosa (Figure 49.4). Care is taken to not deform the lower lip with inappropriate suture placement.
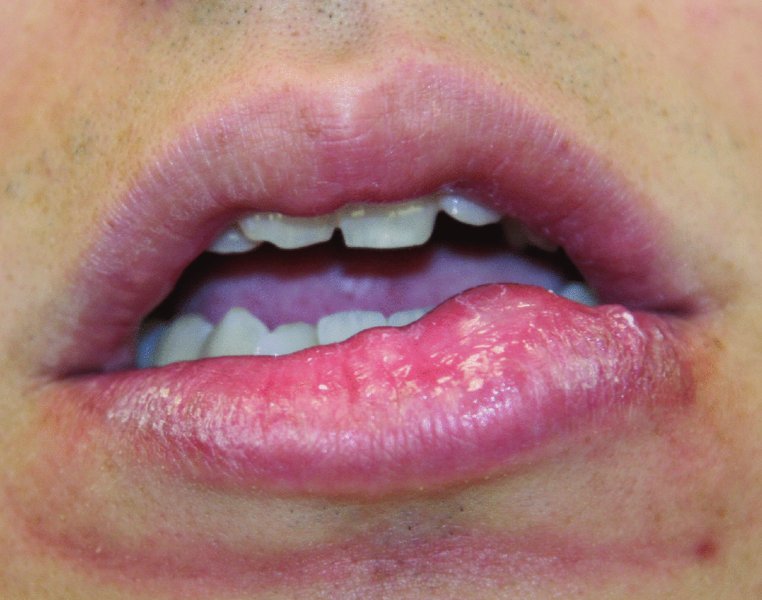
Figure 49.1. Asymptomatic mass to the lower lip.
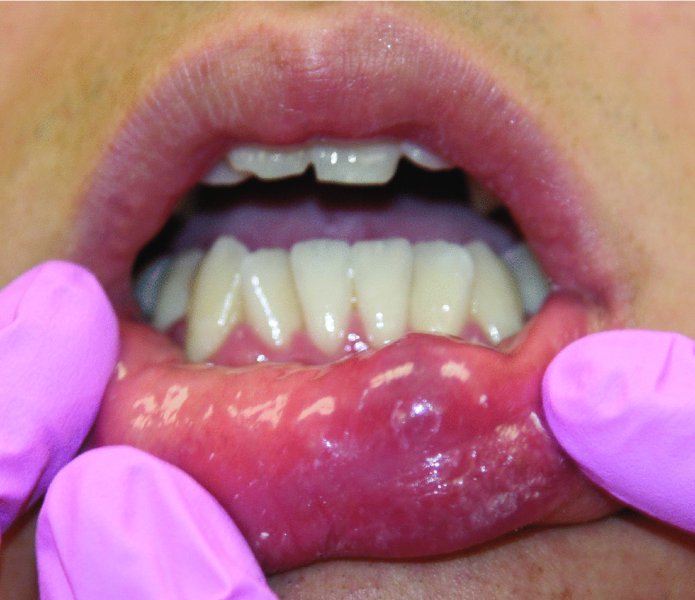
Figure 49.2. Mucoceles typically present as a soft, fluid-filled vesicle with a purple hue, within the lower lip.
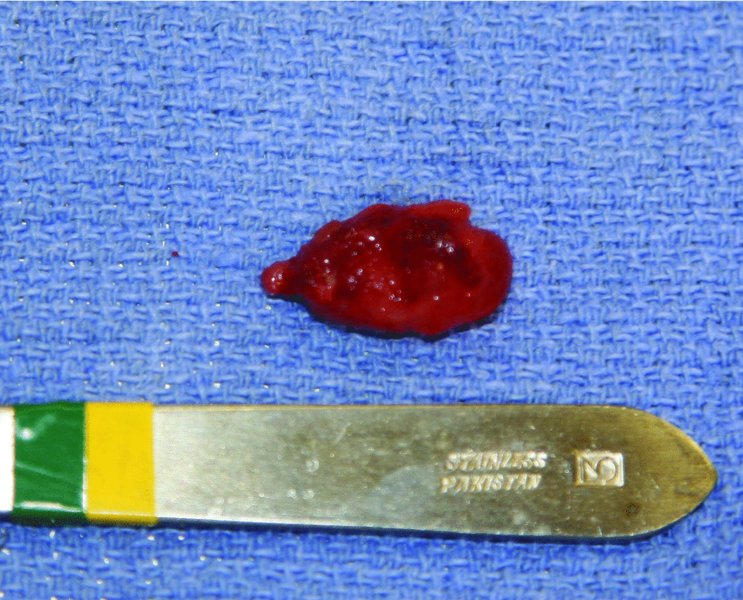
Figure 49.3. An elliptical excision of the mucocele with its associated overlying mucosa and glandular tissue/damaged minor salivary gland.
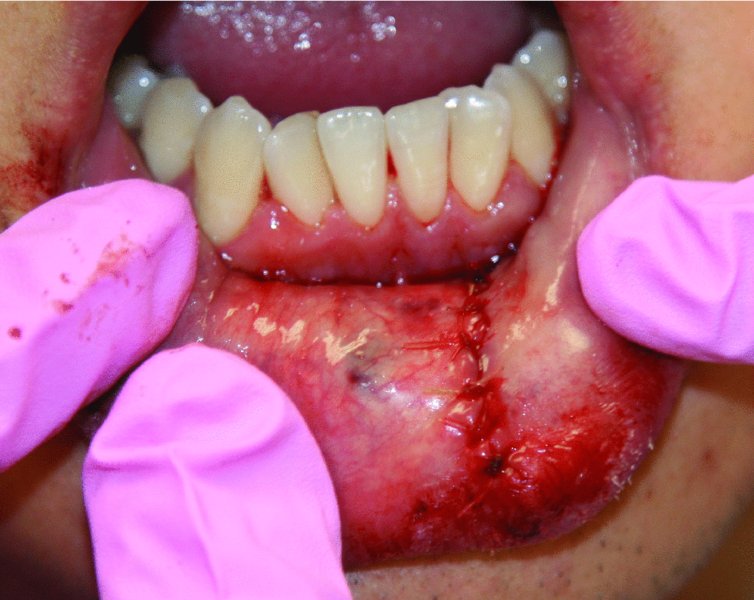
Figure 49.4. Interrupted resorbable sutures are used to reapproximate the mucosa.
Postoperative Management
- Pain medication is prescribed based on the invasiveness of the procedure.
- If the lower lip is the site of excision, ice is applied to the affected area off and on for the first 24 hours.
- A 2-week follow-up is advised to assess the healing of the site and to review the pathology results with the patient.
Complications
- Lower lip asymmetry: Results from inappropriate suture placement. If identified at the time of the procedure, the asymmetry may be corrected with suture removal and resuturing of the lower lip. If identified late, the asymmetry may require revision surgery.
- Recurrence: Due to incomplete removal of the involved minor salivary gland or from damage to adjacent minor salivary glands. Prior to the procedure, the possibility of recurrence should be discussed with the patient.
Key Points
- Although mucoceles may occur at any location containing minor salivary glands (i.e., the soft palate, retromolar pad, ventral tongue, floor of mouth, and buccal mucosa), over 80% of mucoceles are found within the lower lip.
- The majority of mucoceles occur within the first three decades of life.
- Mucoceles typically present as a blue, purple, gray, or normal-colored painless, soft, fluid-filled vesicle on the lower lip. Mucoceles may undergo a fibrotic transformation from chronic inflammation and lip biting.
- Mucoceles infrequently exceed 1.0 cm in their greatest dimension.
- Drainage of mucoceles or incomplete excision of the damaged gland will result in a recurrence of the mucocele.
- Recurrence rates of mucoceles are related to incomplete excision of the involved minor salivary gland and/or damage to adjacent minor salivary glands during the excisional procedure.
- All removed tissue is sent for pathological examination.
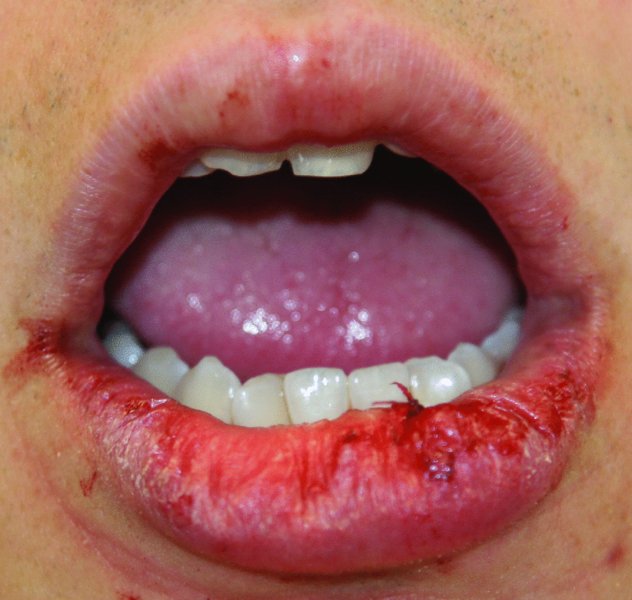
Figure 49.5. The lip is checked for asymmetries. Any asymmetries are corrected with suture removal and resuturing of the lip.
Sublingual Gland Excision
A surgical method for excising an infected or pathologic sublingual gland with or without an associated ranula.
Indications
- Ranula formation
- Sialadenitis
- Sublingual pathology
Contraindications
- Significant medical comorbidities
- Relative contraindication: active infection
Anatomy
- Ranulas: Extravasation mucoceles that arise from the sublingual gland due to damage or rupture of a main duct or the acini after obstruction.
- Plunging ranulas: A ranula that extends through the mylohyoid muscle and extends into the neck, with or without intraoral swelling.
- Sublingual gland: The sublingual gland is the smallest of the major salivary glands and produces 3–5% of the total salivary flow to the oral cavity. The sublingual saliva exits the gland via small ducts of Rivinas. These ducts may empty directly into the floor of the mouth, join Wharton’s duct directly, or coalesce into a larger, common sublingual (Bartholin’s) duct before joining the submandibular duct.
- Lingual nerve: A large, flat nerve that is located within the lateral floor of mouth, superior to the submandibular gland. The lingual nerve descends from the mandibular division of the trigeminal nerve near the lingula, and it courses from lateral to medial as it nears the tongue. During its descension, the lingual nerve loops underneath the submandibular duct, first crossing from the lateral side, and then again passing the duct as the nerve ascends on the medial side to innervate the dorsal tongue.
Technique: Intraoral Ranula and Sublingual Gland Excision
- The procedure may be performed with local anesthesia alone, intravenous sedation, or general anesthesia with endotracheal intubation.
-
Local anesthesia is administered in the form of bilateral nerve blocks and local infiltration. A lacrimal probe is inserted into Wharton’s duct (Figure 49.7) to allow for the identification and retraction of the duct. Traction sutures may be placed within the periphery of the tongue to aid in retraction.
- An elliptical incision is made around the periphery of the ranula through the oral mucosa only. Care is taken to not perforate the ranula itself.
- Blunt dissection with a fine hemostat is used to identify the associated sublingual gland (Figure 49.7). During dissection, the surgeon should be cognizant of the location of Wharton’s duct (easily identifiable if a lacrimal probe was placed) and the lingual nerve medial to the sublingual gland.
- An Allis clamp is used to grasp and elevate the sublingual gland as it is freed from its surrounding tissue with blunt dissection (Figure 49.8).
- The site is copiously irrigated, and the lacrimal probe is removed. The defect is evaluated for hemostasis.
The mucosal defect may be reapproximated with interrupted resorbable sutures or may be left open to granulate secondarily (Figure 49.10).
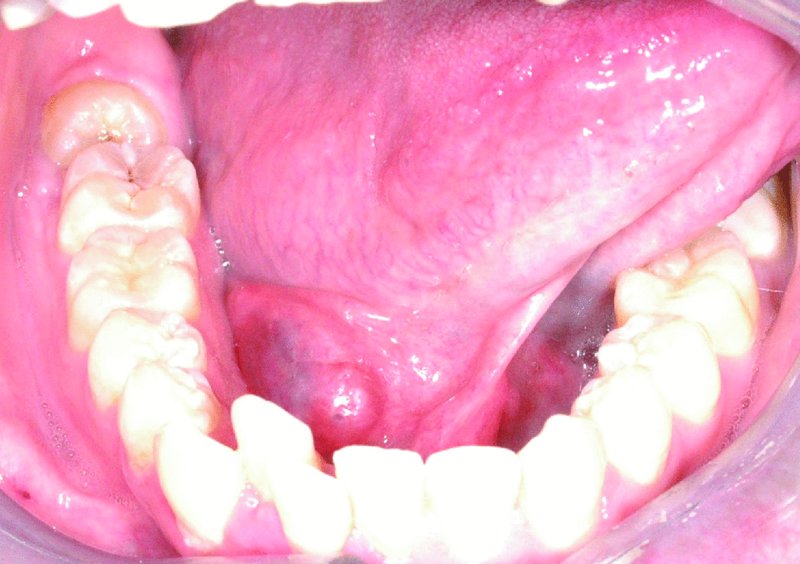
Figure 49.6. A 2.5 cm soft, painless, bluish swelling to the right floor of the mouth.
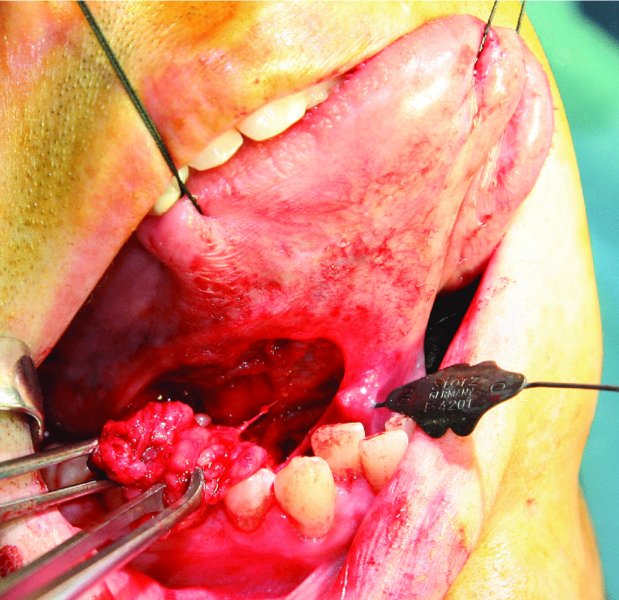
Figure 49.7. Traction sutures and lacrimal probe in place. An elliptical incision was made over the ranula to allow for excision of the ranula and sublingual gland in continuity.
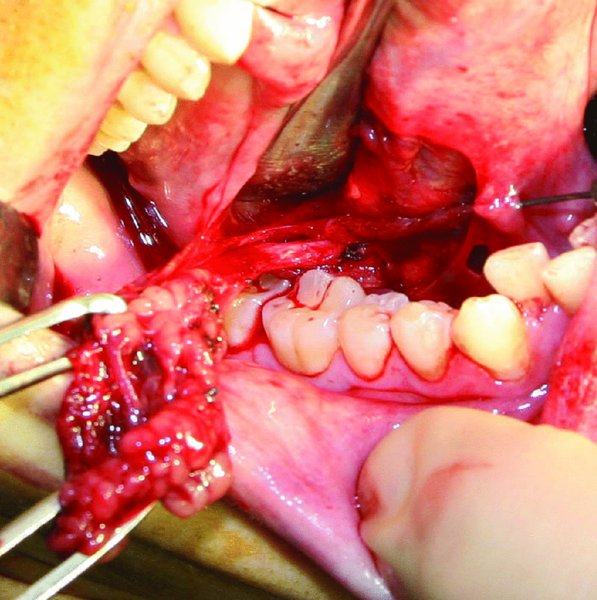
Figure 49.8. Allis clamps are used to grasp the sublingual gland. Blunt dissection is used to free the gland from its surrounding tissue. Wharton’s duct is identified along the medial aspect of the dissection.
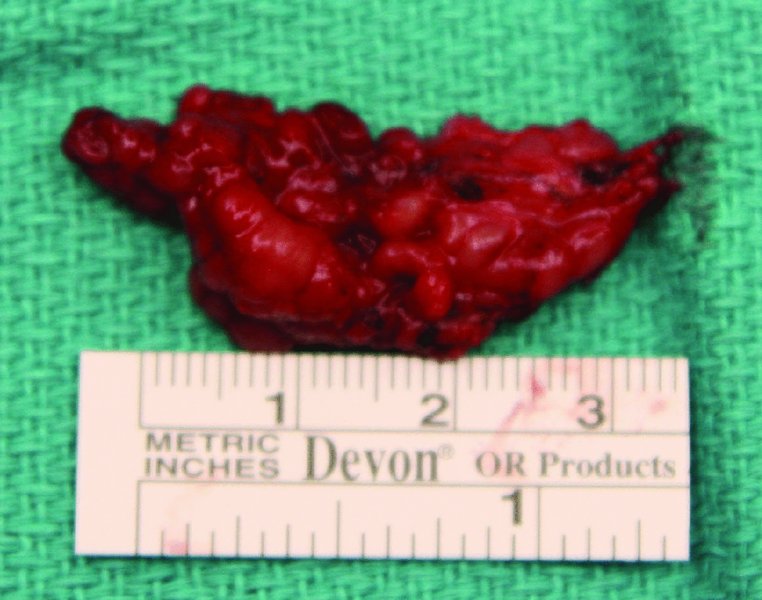
Figure 49.9. Excised right sublingual gland.
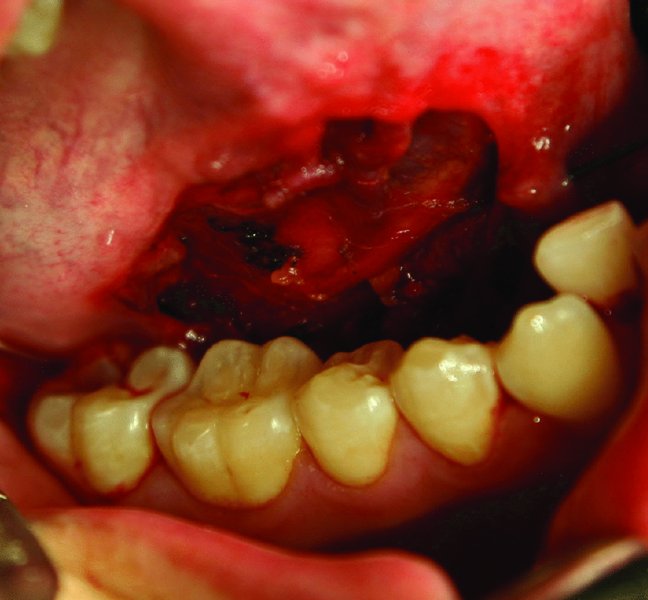
Figure 49.10. The intraoral defect is copiously irrigated and checked for hemostasis. The mucosa surrounding the defect may be reapproximated with interrupted resorbable sutures or allowed to heal secondarily.
Technique: Intraoral Sublingual Gland Excision without Associated Ranula
- Follow steps 1–2 of the “Technique: Intraoral Ranula and Sublingual Gland Excision” section.
- A lingual gingival sulcular incision is initiated along the posterior teeth. The incision may be carried to the midline if additional exposure of the floor of the mouth is necessary.
- A subperiosteal tissue plane is developed, and a full-thickness tissue flap is developed along the lingual aspect of the body or ramus of the mandible.
- The sublingual gland is palpated medial to the periosteal reflection, superior to the mylohyoid muscle. The periosteum overlying the sublingual gland is penetrated with blunt dissection, and the sublingual gland is identified.
- The ensuing sublingual gland removal is described in steps 4–6 of the “Technique: Intraoral Ranula and Sublingual Gland Excision” section.
- The incision is closed primarily with interrupted interproximal resorbable sutures.
Technique: Transcutaneous Plunging Ranula and Sublingual Gland Excision
- The approach to the submandibular triangle is described in steps 1–6 of the “Submandibular Gland Excision” section.
- Once the ranula is identified, blunt dissection is used to mobilize the ranula from its surrounding tissue. Blunt dissection and mobilization continue superiorly until the plunging ranula is freed from the anterior and posterior bellies of the digastric muscles, the hyoglossus muscle, and the herniation through the mylohyoid muscle is identified.
- The dehiscence through the mylohyoid muscle is enlarged, and the attachment of the plunging ranula to the ipsilateral sublingual gland is identified. Blunt dissection is used to free the sublingual gland from its associated tissue. Care is taken to avoid iatrogenic damage to the lingual nerve and Wharton duct medial to the gland.
- The plunging ranula and the associated sublingual gland are delivered through the transcutaneous incision. The dehiscence in the mylohyoid muscle is repaired with interrupted resorbable sutures.
- The incision is irrigated, hemostasis is confirmed, and the wound is closed in a layered fashion. Drains are placed dependent on the size of the plunging ranula excised.
Postoperative Management
- Broad-spectrum antibiotics are prescribed for 5 days postoperatively.
- Saline rinses are begun the day after surgery and continued until the complete mucosalization of the defect or incision site.
- Analgesics are prescribed based on the invasiveness of the procedure.
- Follow-up appointments are typically at 1 week, 3 weeks, and 6 weeks to assess surgical site healing and to assess lingual nerve and Wharton’s duct function.
Complications
- Hematoma formation: Minimized with meticulous dissection and careful inspection of the surgical site prior to procedure completion. Managed with hematoma evacuation with or without drain placement.
- Damage to the lingual nerve (CN V3): Iatrogenic damage to this structure as it courses medial to the sublingual gland will result in a sensory disturbance to the tongue. Most injuries are stretch injuries and improve with time. Witnessed nerve transections should be repaired primarily at the initial surgery.
-
Damage to Wharton’s duct: Minimized by cannulation of Wharton’s duct with a lacrimal probe prior to surgical exploration of the floor of the mouth. Damage to Wharton’s duct may lead to stenosis, obstructive sialadenitis, and sialocele formation. Observed transections or lacerations are treated with intraoperative sialodochoplasty or intraoral cannulation with plastic tubing. The tubing should be left in place for several weeks to promote epithelialization around the stent to reestablish duct continuity (Figures 49.24 and 49.25).
- Ranula recurrence: Recurrence rates are directly related to the procedure performed. Complete sublingual gland excision has the lowest recurrence rate, whereas marsupialization alone is associated with the highest recurrence rate.
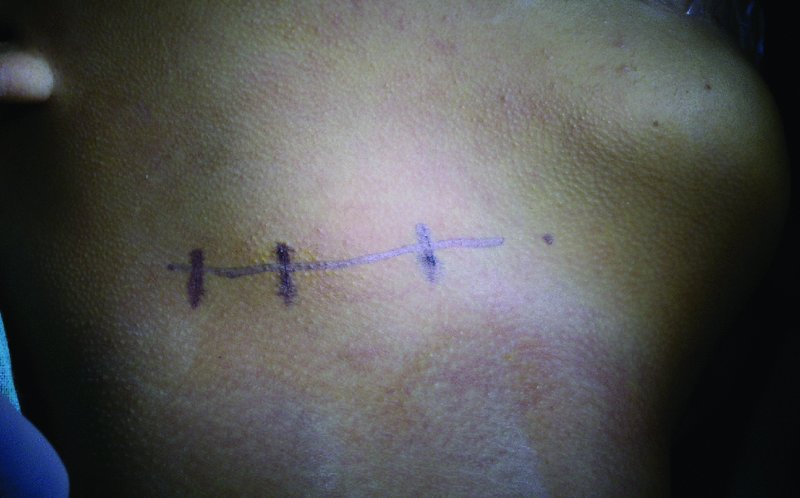
Figure 49.11. The proposed incision is marked approximately 3 cm below the inferior border of the mandible, extending anterior to the anterior border of the sternocleidomastoid muscle.
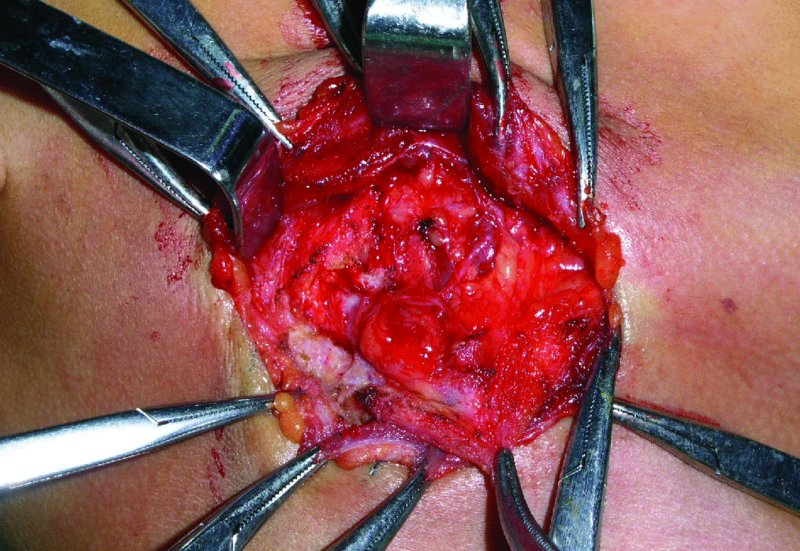
Figure 49.12. The initial incision is through skin, subcutaneous tissues, and platysma.
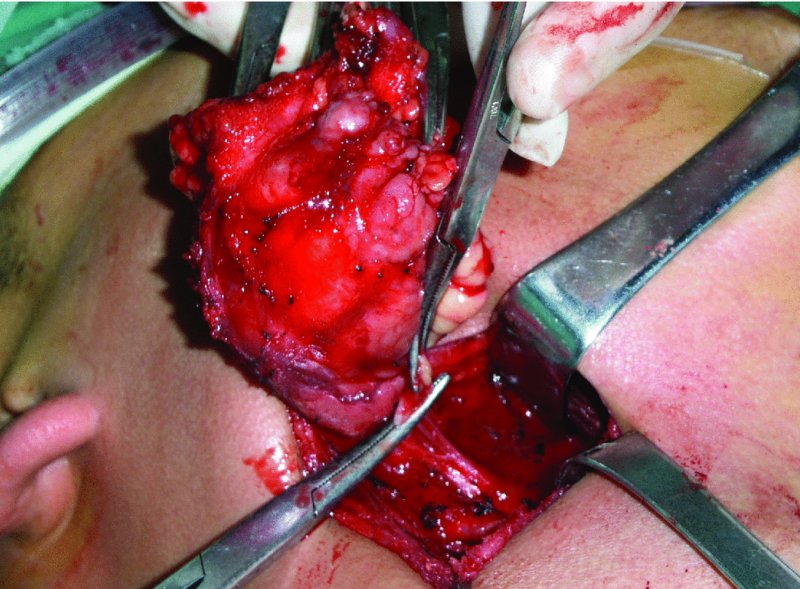
Figure 49.13. The diseased gland is freed from all attachments. The duct and vessels are divided and ligated. The lingual nerve retracts superiorly after division from the ganglion.
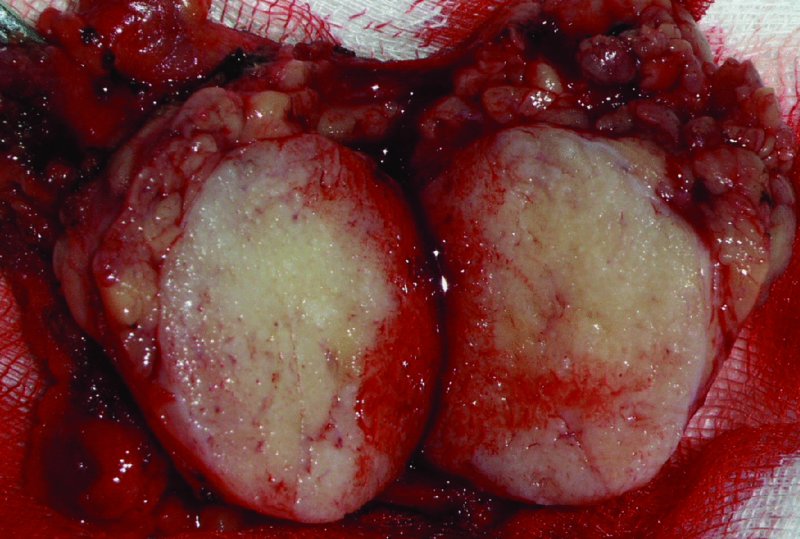
Figure 49.14. The diseased gland is examined prior to being sent for final pathology. Classic “cut-potato” appearance of a pleomorphic adenoma.
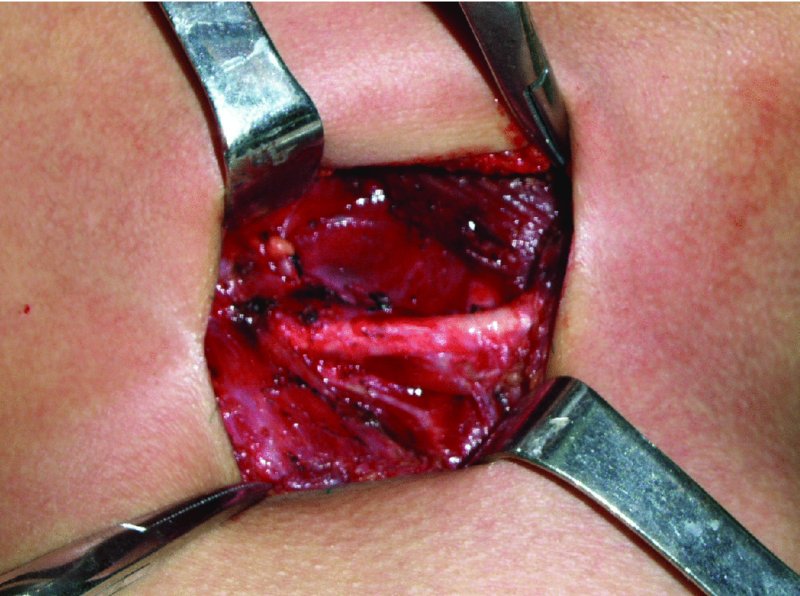
Figure 49.15. View of the surgical field after resection. Note the mylohyoid muscle and digastric tendon.
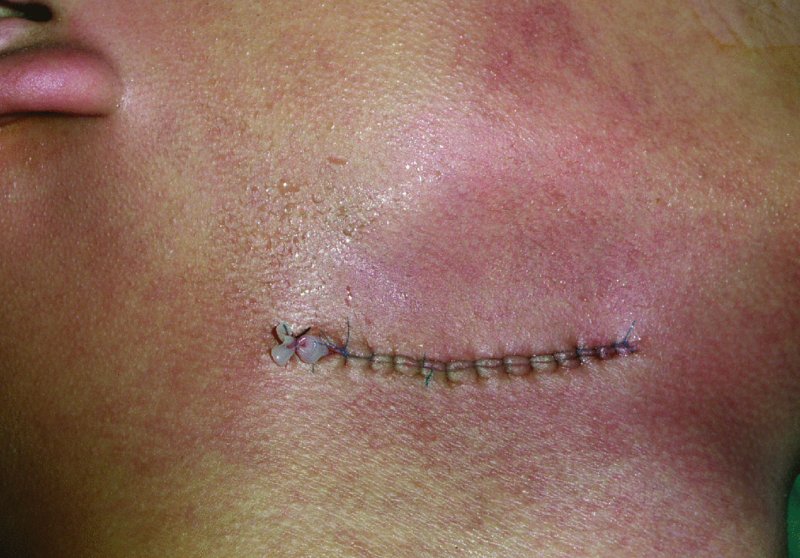
Figure 49.16. Surgical site is closed in layers. A passive Penrose drain is in place.
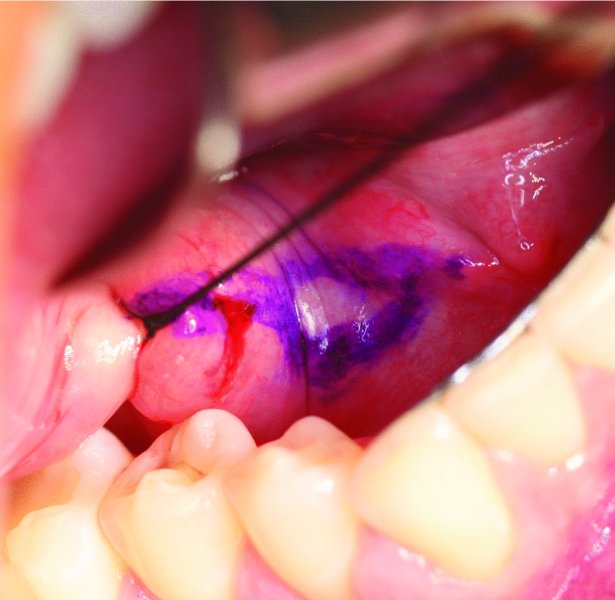
Figure 49.17. The location of the palpable stone is marked in purple. A 2-0 silk suture is placed distal to the stone and around the duct to prevent retrograde migration of the stone toward the hilum of the gland.
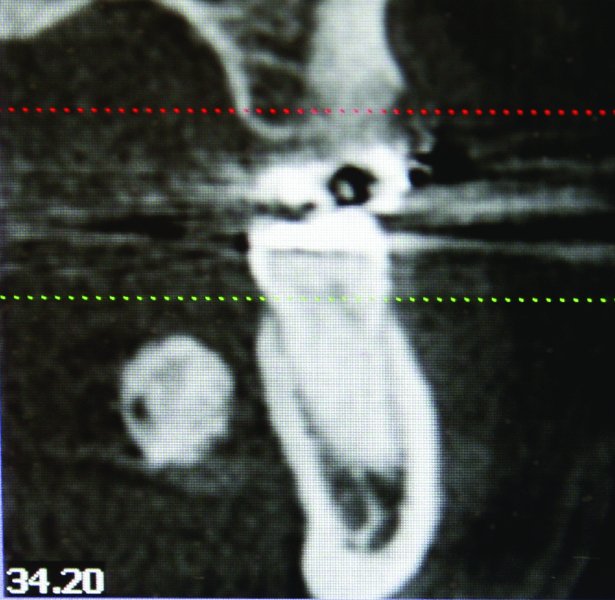
Figure 49.18. Coronal cone beam computed tomography scan demonstrating a radiolucent mass associated with the soft tissue of the left floor of the mouth.
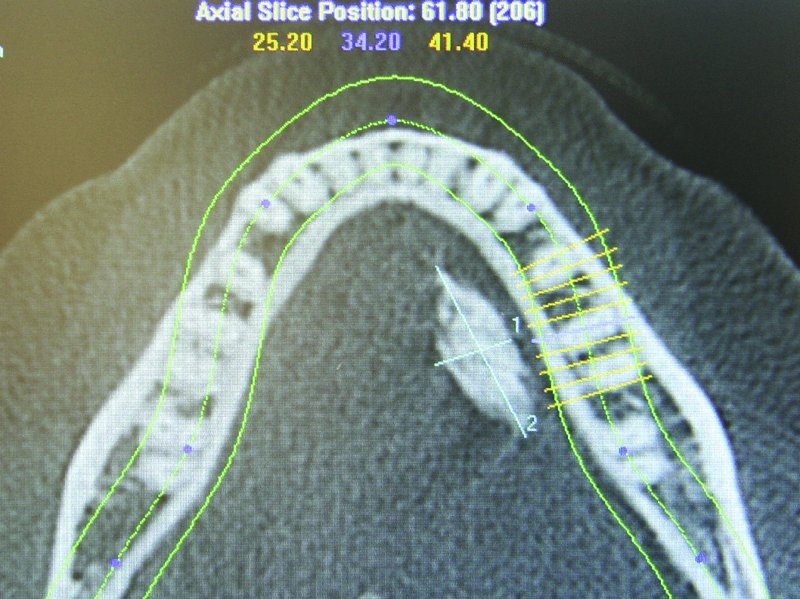
Figure 49.19. Axial cone beam computed tomography scan demonstrating a 23 mm (length) by 12 mm (width) sialolith located within the left submandibular (Wharton’s) duct.
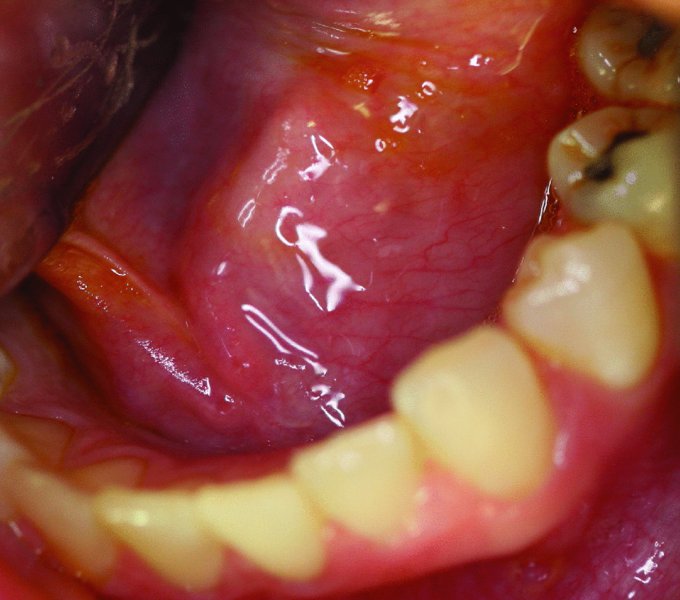
Figure 49.20. Obvious floor-of-mouth mass. Wharton’s duct was found to be functional.
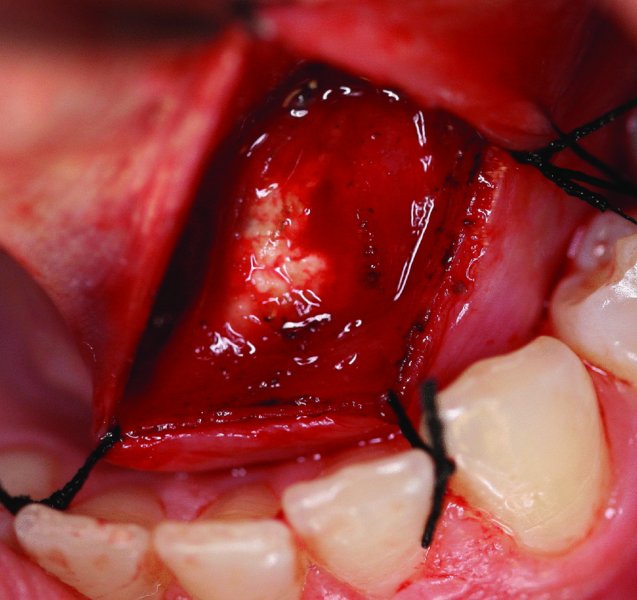
Figure 49.21. The superficial floor-of-mouth mucosa directly over the stone is transected via sharp dissection incising parallel to the duct. Traction sutures are placed to aid in visualization.
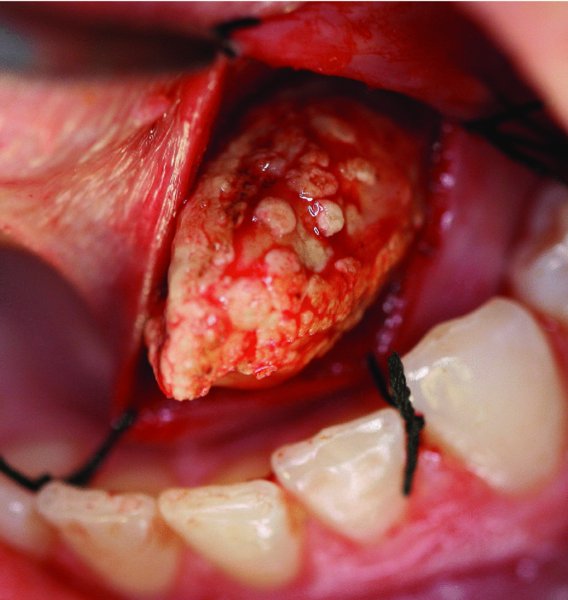
Figure 49.22. The sialolith is delivered from the left submandibular duct.
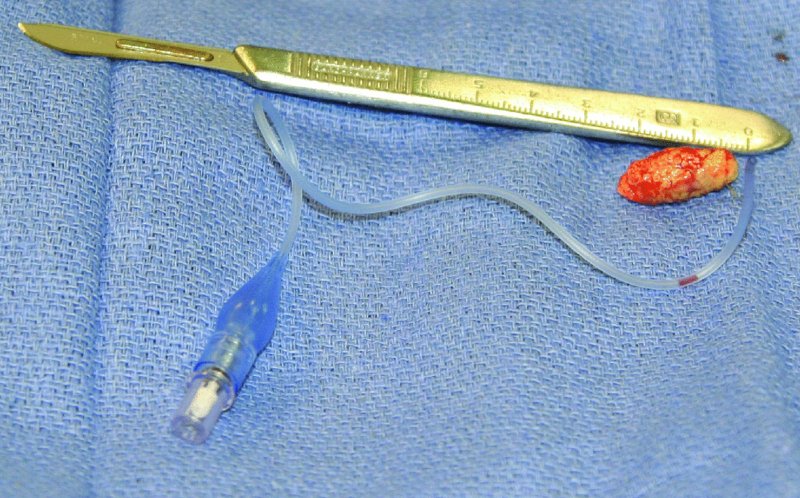
Figure 49.23. The 23 mm × 12 mm sialolith and the sterile tubing from an endotracheal tube cuff. The sterile tubing from the endotracheal tube cuff will be used as a catheter to maintain the original patency of the damaged submandibular duct.
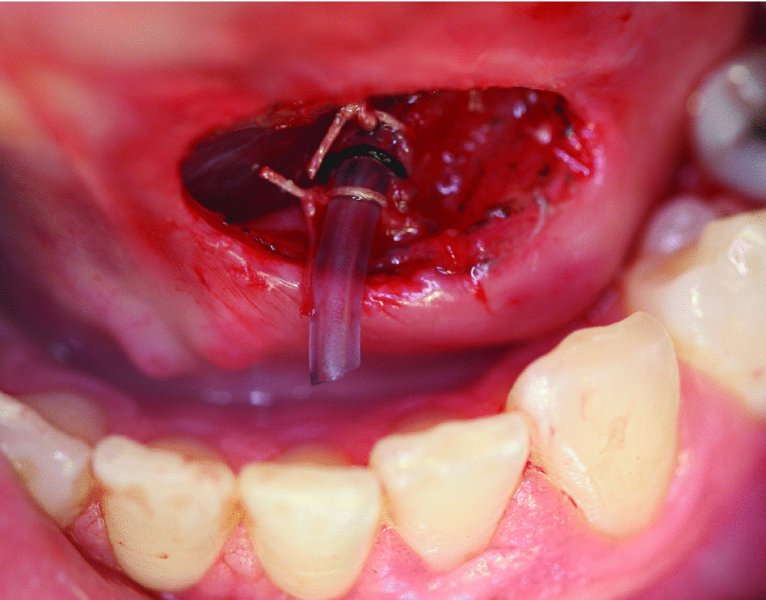
Figure 49.24. Sterile tubing is positioned within the dilated submandibular duct and passively inserted to the hilum of the submandibular gland. The tubing is secured to the floor-of-mouth tissues.
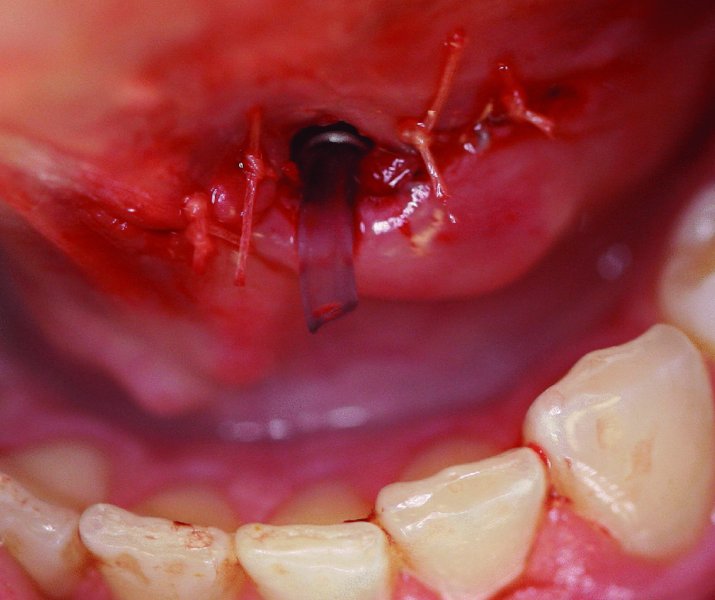
Figure 49.25. The orifice of the duct is sewn to the floor-of-mouth tissues.
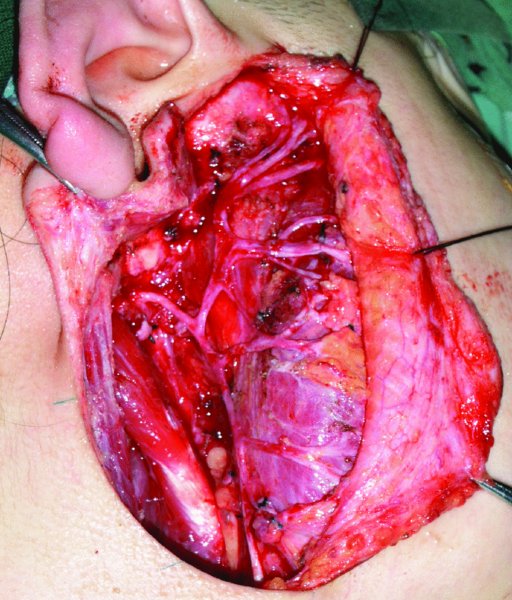
Figure 49.26. Exposure of the facial nerve after removal of the superficial lobe of the parotid.
Key Points
- Multiple treatment modalities exist for ranula management. Surgica/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


