Restorative and Esthetic Dental Materials
Learning Outcomes
On completion of this chapter, the student will be able to achieve the following objectives:
• Pronounce, define, and spell the Key Terms.
• Discuss how a dental material is evaluated before it is marketed to the profession.
• List the properties of dental materials and ways that they affect their application.
• Discuss the differences between direct and indirect restorative materials.
• Describe the factors that affect how dental materials are manufactured for the oral cavity.
• Describe the properties of amalgam and its application in restoring teeth.
• Describe the properties of composite resin materials and their application in restoring teeth.
• Describe the properties of glass ionomers and their application in restoring teeth.
• Describe the properties of temporary restorative materials and their application in restoring teeth.
• Discuss the use of tooth-whitening products.
• Describe the properties of gold alloys and their application in restoring teeth.
• Describe the properties of porcelain and its application in restoring teeth.
Performance Outcomes
On completion of this chapter, the student will be able to achieve competency standards in the following skills:
Electronic Resources
![]() Additional information related to content in Chapter 43 can be found on the companion Evolve Web site.
Additional information related to content in Chapter 43 can be found on the companion Evolve Web site.
![]() and the Multimedia Procedures DVD
and the Multimedia Procedures DVD
Key Terms
Adhere To stick or glue two items together.
Alloy A mixture of two or more metals that are dissolved with each other when in the liquid state.
Amalgam (uh-MAL-gum) An alloy, with one of the constituents being mercury.
Auto-cured Hardened or set by a chemical reaction of two materials.
Ceramic Hard, brittle, heat- and corrosive-resistant material such as clay.
Coupling agent Agent that strengthens resin by bonding filler to the resin matrix.
Cured Preserved or finished by a chemical or physical process.
Dual-cured Prepared, preserved, or finished by a chemical or physical process.
Esthetic (aesthetic) (es-THET-ik) Artistically pleasing and beautiful appearance.
Filler Inorganic material that adds strength and other characteristics to composite resins.
Force Causes a physical change through energy and strength.
Galvanic (gal-VA-nik) An electrical current that takes place when two different or dissimilar metals come together.
Gold A soft, yellow, corrosive-resistant metal that is used in the making of indirect restorations.
Irregular Not straight, uniform, or symmetric.
Malleability (mal-ee-uh-BIL-uh-tee) The ability of a material to withstand permanent deformation under compressive stress without sustaining permanent damage.
Matrix (MAY-triks) Foundation that binds a substance together; continuous phases (organic polymer) in which particles of filler are dispersed in composite resin.
Microleakage (mye-kroe-LEE-kuj) Microscopic space located at the interface of the tooth structure and the sealant or restoration.
Palladium (puh-LAY-dee-um) Soft, steel-white, tarnish-resistant metal that occurs naturally with platinum.
Pestle (PES-ul, PES-tul) An object that is moved vertically to pound or pulverize a material.
Platinum (PLAT-i-num) Silver-white noble metal that does not corrode in air.
Porcelain (POR-se-lun) Hard, white, translucent ceramic material fabricated by firing and then glazing it to match the tooth color.
Restorative (re-STOR-uh-tiv) To restore or bring back to its natural appearance.
Retention (ree-TEN-shun) The act of holding something by means of an adhesive, or mechanical locking, or both.
Spherical (SFEER-i-kul) Round.
Strain Distortion or change produced as a result of stress.
Stress Internal reaction or resistance to an externally applied force.
Viscosity (vis-KOS-i-tee) Physical property of fluids responsible for resistance to flow.
Wetting Covering or soaking something with a liquid.
Dental materials fulfill an important role in the way dentistry is delivered today. Selection of the appropriate dental material for a procedure will depend on several variables: the extent of decay; the type of defect in the tooth; the condition of the entire mouth; the location of the restoration; and cost factors.
The restorative and esthetic dental materials used most commonly in dentistry today include the following:
Restorative is a term in dentistry that describes the ability to remove decay or disease and bring back the proper function of a tooth. Esthetic is a term that refers to recreating the tooth or teeth with an artistically pleasing appearance.
The types of dental materials introduced in this chapter are restorative and esthetic materials most often used by the general dentist. An understanding of these dental materials will provide you with the knowledge necessary to recognize the types of procedures practiced by the general dentist.
As a clinical dental assistant, it is essential that you learn the general characteristics of each type of dental material, the selection criteria, and ways to prepare the restorative and esthetic materials for a procedure. It will be your responsibility to assemble the setup for each material while being attentive to the manufacturer’s recommended proportions and mixing techniques.
Standardization of Dental Materials
When a new dental material is developed, the product must undergo a strict evaluation and assessment before it is marketed to the profession. The Council on Dental Materials, Instruments and Equipment was formed as a subcommittee by the American Dental Association (ADA) in combination with federal organizations to ensure that standards and strict specifications are followed by dental material manufacturing companies when new dental materials are developed.
Criteria for a New Dental Material
• It must not be poisonous or harmful to the body.
• It must not be harmful or irritating to the tissues of the oral cavity.
• It must help protect the tooth and tissues of the oral cavity.
• It must resemble the natural dentition as closely as possible so as to be esthetically pleasing.
• It must be easily formed and placed in the mouth to restore the mouth’s natural contour.
• It must conform and function despite limited access, wet conditions, and poor visibility.
Properties of Dental Materials
The types of dental materials used to restore teeth must respond to and withstand specific factors associated with oral conditions. The following characteristics enhance the ability of dental materials to withstand the oral environment and allow for easy application.
Mechanical Properties
The average biting and chewing force in the posterior area of the mouth of people with natural teeth is approximately 170 pounds (77 kg). This is on the order of 28,000 pounds of pressure per square inch (psi) on a single cusp of a molar tooth. Materials used in restoring posterior teeth must have sufficient strength to withstand such force.
A force is any push or pull on matter. In turn, force can create a stress and a strain. Stress is the reaction within the material that can cause distortion. Strain is the change produced within the material that occurs as the result of stress.
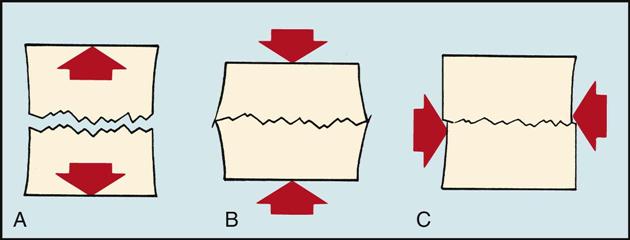
Types of Stress and Strain
• Tensile stress pulls and stretches the material. A tug-of-war is an example of tensile stress (Fig. 43-1, A).
• Compressive stress pushes the material together. Chewing is an example of compressive stress (Fig. 43-1, B).
• Shear stress is the breakdown of material as the result of something sliding over two areas. Cutting with scissors is an example of shear stress (Fig. 43-1, C).
Thermal Change
When a person drinks hot coffee and then immediately eats ice cream, the temperature in the mouth can change from 150° F to 100° F (66° C to 38° C) within seconds. These thermal changes are of major concern for two reasons: (1) contraction and expansion, and (2) the need to protect the pulp from thermal shock from extreme differences in temperature.
Contraction and Expansion
When temperature changes occur, each type of dental material will contract or expand at its own rate. It is essential that the tooth structure and the restorative material have, as nearly as possible, the same rate of contraction and expansion. Significantly different rates of contraction and expansion can cause the dental material to pull away from the tooth, which can result in microleakage, or a faulty restoration. This minute separation of material and tooth could allow fluids, debris, and microorganisms to enter between the restoration and the wall of a cavity preparation.
Electrical Properties
An electrical current (also referred to as galvanic action) can take place in the oral cavity when two different or dissimilar metals are present (Fig. 43-2). Conditions that allow these electrical currents include the following:
Corrosive Properties
Corrosion is the reaction of metals that occurs within a metal when it is exposed to corrosive factors such as temperature, humidity, and saline. Certain foods contain metallic forms that cause corrosion of a dental material (Fig. 43-3). Most corrosion, however, involves surface discoloration and can be removed easily with the use of polishing agents.
Solubility
Solubility is the degree to which a substance will dissolve in a given amount of another substance. For example, sand has low solubility because it does not dissolve easily; sugar has high solubility because it does dissolve easily. The solubility of a material placed in the mouth is a matter of concern (Fig. 43-4). A material that dissolves easily in the oral environment is of limited use because it will wash away and leave the tooth structure exposed.
Application Properties
For a dental material to have specific mechanical properties, steps must occur in the application of the dental material. Techniques used in the application help create the following effects.
Flow
When decay is removed and healthy tooth structure is prepared to receive a permanent filling material, the dental material must be pliable enough to be placed in the preparation. Dental materials are designed to have a certain amount of flow for placement; this allows the material to fill in the preparation of the tooth.
Adhesion
Adhesion is the strength that causes unlike materials to adhere to each other. Adhesion between a dental material and the tooth structure is a major concern. Without proper adhesion, microleakage can occur, and the restoration could come out. Characteristics of dental materials that can affect the adhesion process are wetting, viscosity, surface characteristics, and film thickness.
Wetting is the ability of a liquid to flow over the surface and come into contact with the small irregularities that may be present. For example, water has high wetting ability because it flows easily.
Viscosity is the property of a liquid that causes it not to flow easily. A liquid with high viscosity, such as maple syrup, does not flow easily and is not effective in wetting a surface.
Surface characteristics influence the wetting ability of the material. A liquid flows more easily on a rough surface than on a very smooth surface. For example, water flows easily over sandpaper; however, on wax paper, water beads up and does not flow as readily.
For adhesion to take place and remain permanent, the dental material selected that will join the tooth and restoration will require a thin film thickness. It is important to remember that the thinner the film thickness, the stronger the adhesive junction. For example, the ideal film thickness for cementing a permanent restoration is 25 mm or less.
Retention
Retention is the ability to hold two things firmly together when they will not adhere to each other naturally. For example, amalgam (silver fillings) and cast metals will not naturally adhere directly to tooth structure. Retention is an extremely important concept in dentistry because dental restorations, casting, and appliances must be held in place with the use of materials and retention methods.
Because amalgam and tooth structure do not adhere to each other, a traditional cavity preparation must include a retention form. The dentist will use a bur or a hand cutting instrument to produce this retention form (creating a crevice in the tooth that the material can lock into). Even though retention holds the material in place, it does not completely seal the two structures together. Microleakage can occur between the material and the tooth if an additional material such as a bonding material is not placed to create a seal.
Curing
Curing is a setting process of a dental material that is initiated by a chemical reaction or by light in a blue wave spectrum. Curing of most dental materials is the process of placing a paste-type material into the tooth preparation for adaptation. After the material is in place, it is then cured to harden.
An auto-cured or self-cured material hardens as the result of a chemical reaction of the materials once mixed together. The curing action proceeds from start to finish by itself. Because of urgency, the material must be mixed and placed within working and setting times for that material.
A light-cured material does not harden until it has been exposed to a curing light (Fig. 43-5). This allows a more flexible working time.
With a dual-cured material, some curing takes place as the material is mixed. However, the final cure does not occur until the material has been exposed to a curing light.
Direct Restorative and Esthetic Materials
Direct restorative and esthetic materials are applied to a tooth or teeth while the material is pliable and can still be adapted, carved, and finished. Materials used in these types of restorative and esthetic procedures are amalgam, composite resins, glass ionomers, temporary restorative materials, and tooth-whitening products.
Amalgam
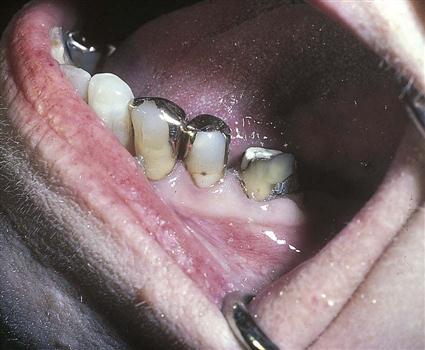
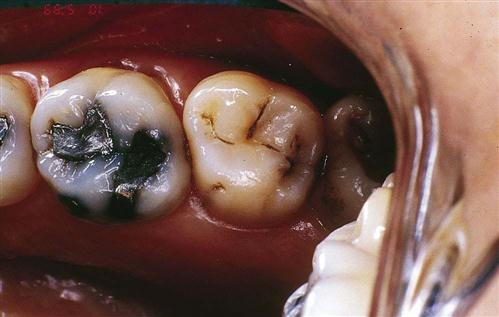
For longer than 150 years, billions of dental amalgam fillings have been used to restore decayed teeth. Amalgam is the technical name for “silver fillings” (Fig. 43-6). Actually, amalgam consists of several different metals, with silver being the predominant metal. These metals are in powder form and are mixed with mercury to form a soft, pliable mixture. When placed into a prepared tooth, condensed, carved, and allowed to harden, the mixture becomes a permanent amalgam restoration.
Dental amalgam is a safe, affordable, and durable material used predominantly to restore premolars and molars.
Indications for Using Dental Amalgam
• Stress-bearing areas of the mouth
• Small to medium-sized cavities in the posterior teeth
• Severe destruction of tooth structure
• As a foundation for cast-metal, metal-ceramic, and ceramic restorations
• When a patient’s commitment to personal oral hygiene is poor
Contraindications to the Use of Dental Amalgam
Composition of Dental Amalgam
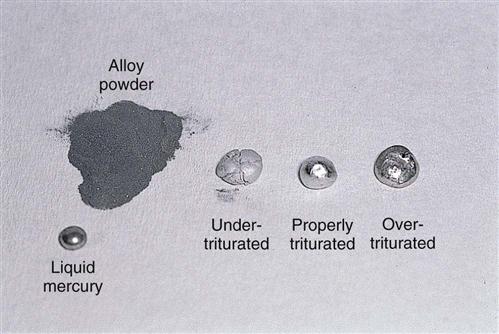
Dental amalgam is the end result of mixing approximately equal parts of mercury (43 to 54 percent) and an amalgam alloy powder (46 to 57 percent) (Fig. 43-7). The alloy powder is a combination of metals. Amalgam alloy powder is composed of the following metals:
• Silver, which gives it its strength
• Tin, for its workability and strength
The main differences in the composition and classification of dental amalgam alloy powders are based on (1) alloy particle shape and size, (2) copper content, and (3) zinc content.
High-Copper Alloys
High-copper alloys, frequently used in dentistry, are named this because they contain a higher percentage of copper than previous alloys. High-copper alloys are classified according to their particle shape: spherical (round particles) or irregular (rough, lathe-cut particles) (Fig. 43-8). These particle shapes influence the trituration and working characteristics (condensing and carving) of the resulting amalgam mixture.
A high-copper alloy is made up of 40 to 70 percent silver, 8 to 28 percent copper, and 15 to 30 percent tin. The percentages are expressed as percentages of the composition by weight. Table 43-1 describes the composition and classification of dental amalgam alloy powders.
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses