4
The dentoalveolar gingival unit
With the growing trend in smile-enhancement therapies, more people are striving for that perfect smile. Indeed, a harmonious dentogingival junction is one that fits perfectly with the rest of the face. Not every small modification from the theoretical ideal leads to a lessening of the smile’s esthetic value; such deviation may personalize the smile and keep it from becoming too monotonous. In addition, it is interesting to recall that a perfect symmetry between the two halves of the human face does not exist, and that the midline of the face and the midline of the dental arch correspond in only 70% of patients. The vertical maxillary and mandibular axes line up in only 75% of patients (Magne and Belser, 2003).
The vertical dimension of the dentogingival junction, consisting of the sulcus depth, the junctional epithelium (JE), and the connective tissue attachment (CTA), is physiologically predetermined and constant. The level of this “biological width” is dependent on the location of the crest of the alveolar bone (Figs 4.1a, b) and includes only the JE and CTA. Changing the level of the alveolar bone moves the entire dentogingival junction at an apical or coronal level (Landolt and Blatz, 2008).
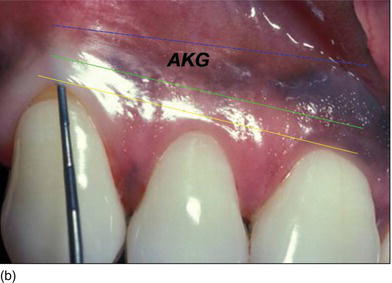
Figure 4.1 (a) A diagram of the different components of the dentoalveolar gingival unit. (Courtesy of Dr. M. Legall.) (b) Fully keratinized gingiva from the FGM to the MGJ, and defining the amount of attached keratinized gingiva with the probe.
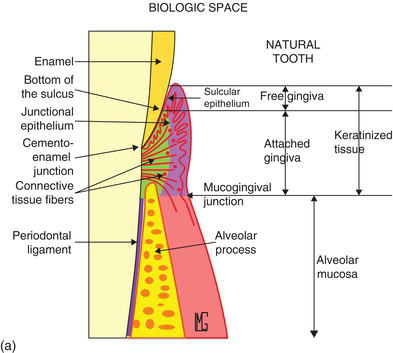
The composition of an esthetic and functional dentoalveolar gingival unit (DAGU) or periodontal restorative interface (PRI) must take into account the periodontal health of the bone, the gingiva, the interdental papillae (the pink esthetic score, or PES), the teeth (the white esthetic score, or WES), and the biological space (JE+CTA).
The gingiva
The esthetics of the anterior maxillary region of the dentition depends largely on the appearance of the gingival tissues surrounding the teeth. Traditionally, the physiological gingival architecture has been described as having a scalloped contour (Prichard, 1961) around the four surfaces of the tooth, in accordance with the course of the cemento-enamel junction (CEJ) (Schroeder, 1991) and is thus concave apically in the free surfaces and convex at the tip of the papilla.
Esthetic considerations
The gingival margins of the maxillary anterior dentition should be at their correct level. Asymmetrical gingival tissues can significantly affect the harmonious appearance of both natural or prosthetic dentition. The zenith is defined as the most apical point of the gingival marginal scallop (Figs 4.2a–c). The proper placement of the gingival zenith should be at the peak of the parabolic curvature of the gingival margin, which for the central incisors, canines, and premolars should specifically be located slightly distal to the middle of the long axis on these teeth. This produces the subtle inclination of the distal root that is paramount for the foundation of a beautiful smile. The zenith for the lateral incisors is located at the midline of the long axis of the tooth.
The gingival margin of lateral incisor teeth can range from being at the same level or 0.5–2.0 mm coronal to the zeniths of the central incisor and the canine teeth (Feigenbaum, 1991). The height of the gingival margin for the lateral incisors should be 1 mm shorter than the gingival margins of the adjacent teeth (Kurtzman and Silverstein, 2008). In other words, the gingival margin of the lateral incisor should be 1 mm coronal to the line connecting the gingival margins of the central incisor and the canine.
Figure 4.2 (a) The gingival zenith does not exist on thelateral incisor, but it is limited and is slightly distal on the cuspid. (b) The gingival zenith on the central incisor is located distal to the midline. (c) The gingival level of the lateral incisor is below the line joining the adjacent teeth and could vary from one side to the other.
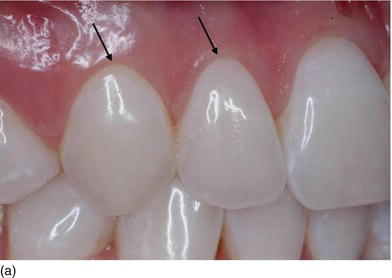
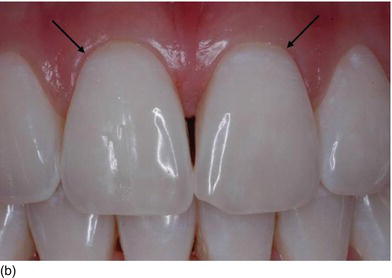
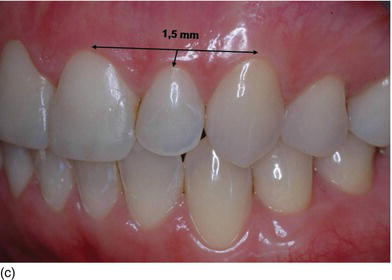
The gingival zenith of the canine is at the same level or slightly more apical to the gingival zenith of the central incisors. When the patient is looking straight ahead, the gingival zenith of the lateral incisor is typically below (81.1%) or on the gingival line joining the cuspid and the central incisor zenith point (15%).
More recently, it has been found that the heights of the gingival tissues over the maxillary central incisors should be slightly higher (1 mm apically) than the heights of the tissues over the maxillary lateral incisors. The heights of the maxillary canines should be at the same level apically as the central incisors, or slightly more apical. The gingival zeniths should be located at the distolabial line angles, thus creating a “raised eyebrow” over the central incisors. A directional gingival contour asymmetry has been demonstrated, with the right side higher than the left side (Charruel et al., 2008).
A recent study (Mattos and Santana, 2008) has also shown that the gingival zenith is not universally displaced toward the distal aspect. The frequency and magnitude of distal displacement is dependent on the crown tooth position and its root orientation. It is more pronounced in central incisors than in lateral incisors, in which, in turn, it is more prominent than in canines. In other words, the distal position of the zenith was very frequent on the central incisor, frequent on the lateral incisor, and rare on the canine.
Consideration of these findings may improve clinical management of the dentogingival complex (DGC) and enhance periodontal surgical procedures, as well as conventional or implant restorative measures in the anterior maxillary dentition.
The gingival biotype
The gingiva can present a range of characteristics:
- Frame: harmony or disharmony, in relation to tooth eruption on the ridge (Figs 4.3a–c).
- Color: pink or pigmented, in relation to ethnicity (Figs 4.3d, e).
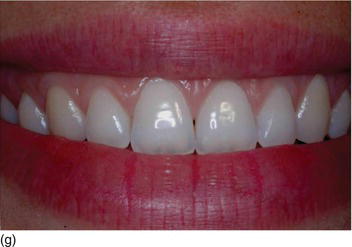
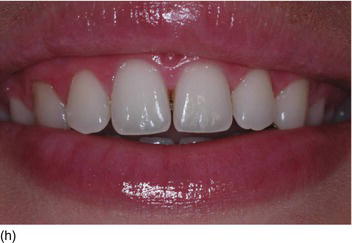
- Texture: normal or inflamed, in relation to oral hygiene (Figs 4.3f, g).
- Biotype: thick, medium, or thin, in relation tothe cortical bone plate (Weisgold and Coslet, 1977).
- minimal disparity between the location of the gingival margin and the peak of the papilla
- a flatter, thicker underlying osseous form
- denser, more fibrotic soft tissue
- a larger amount of keratinized attached gingiva, and
- a square tooth shape.
- a distinct disparity between the location of the gingival margin and the peak of the papilla
- a scalloped osseous form, and often dehiscence and fenestration
- delicate, friable soft tissue
- a minimal amount of keratinized attached gingiva, and
- a triangular tooth shape.
Another biotype classification has been proposed by Maynard and Wilson (1980) and is based on classification of the gingiva/alveolar same thickness (Figs 4.4d, e).
Figure 4.3 (a) The gingival contour on the lower anterior teeth, in harmony and without any inflammation. (b) Gingival and dental disharmony before orthodontic treatment. (c) The right mandibular incisor is erupting buccally with less keratinized gingiva and the left one lingually with an excess amount of keratinized gingiva.
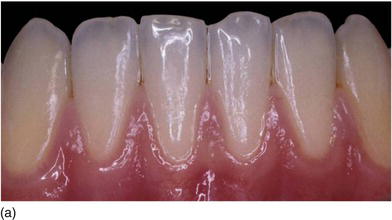
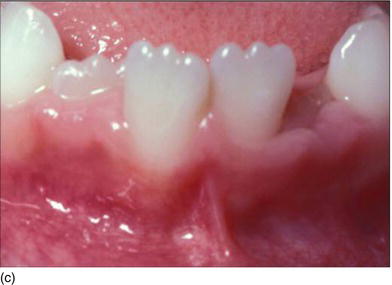
Figure 4.3 (d) Pink attached gingiva on a blond Caucasian patient. (e) Pigmented attached gingiva on a black African patient. (f) Noninflamed gingiva with a healthy gingival surface texture. (g) Inflamed gingiva induced by biofilm and calculus.
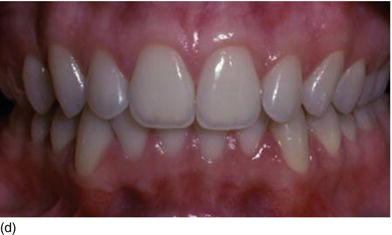
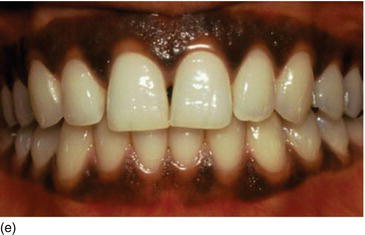
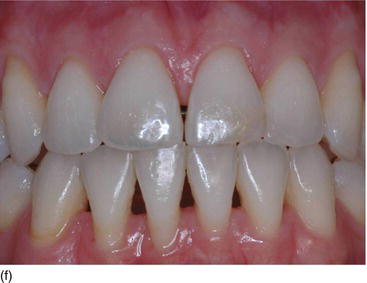
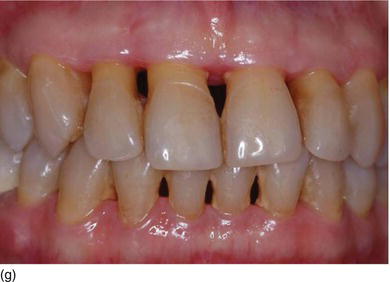
Figure 4.4 (a) The thick, flat gingival biotype.
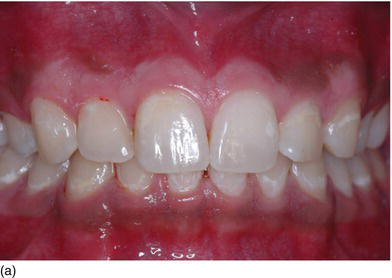
Figure 4.4 (b) The thin, scalloped biotype. (c) The medium gingival biotype.
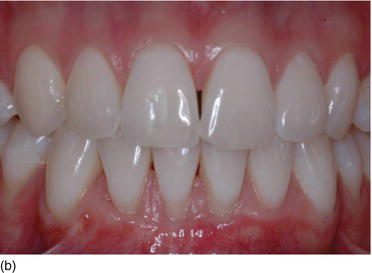
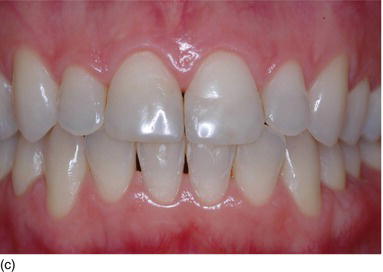
Figure 4.4 (d) Type I, thick gingiva/thick biotype; Type II, thin gingiva/medium biotype. (e) Type III, thick gingiva/medium biotype; Type IV, thin gingiva/thin biotype. (Courtesy of Dr. S. Rocha Bernardes, Curitiba, Brazil.)
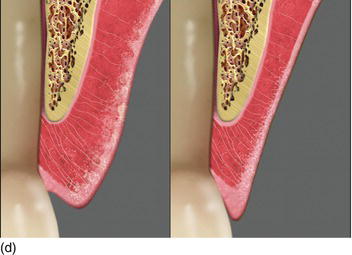
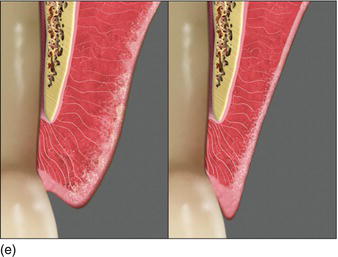
Table 4.1 Bone and gingival biotypes
| Type I | Thick bone | Thick gingiva/Thick bone |
| Type II | Thick bone | Thin gingiva/Medium bone |
| Type III | Thin bone | Thick gingiva/Medium bone |
| Type IV | Thin bone | Thin gingiva/Thin bone |
The bone biotypes I, II, and III correspond to thick and medium periodontium, and only type IV corresponds to thin periodontium. This classification could be interesting in establishing the planning of implant treatment and in evaluation of the esthetic results (Table 4.1).
The presence of an adequate zone of keratinized mucosa was thought to be necessary for the maintenance of gingival health and to prevent the progression of periodontal disease. Lang and Loë (1972) suggested a width of at least 2 mm of keratinized mucosa, of which 1 mm was to be attached.
Subsequently, several authors have challenged this concept, and have shown that gingival health can be maintained with hardly any attached gingiva but with good hygiene (Miyasato et al., 1977; Kennedy et al., 1985).
Restoration on natural teeth has supra-, juxta-, or intrasulcular cervical limits and, in general, the gingival profile dictates the prosthetic emergence profile.
Subgingival restorations around teeth tend to recede over time (Valderhaug, 1980) and this phenomenon has been confirmed by Stetler and Bissada (1987), who reported that a narrow zone of keratinized mucosa in teeth with subgingival restorations is associated with a higher chance of gingival inflammation; this can be extrapolated to implant crown restorations. Thus, it is especially important to have a keratinized tissue zone adjacent to dental implants, because the implant restoration is always located beneath the oral mucosa margin in the esthetic zone, and it should conceal the subgingival part of the abutment. Together with its double submerged and emergent architecture, the abutment/implant restoration contributes to the shaping and formation of the peri-implant mucosa at the level of its scalloped marginal contour and the interdental papillae in harmony with the adjacent teeth (Warrer et al., 1995; Saadoun and Touati, 2007; Bouri et al., 2008).
Although the medium and thick biotypes account for 70% of cases, the more extreme thin biotypes, which make up about 15% of cases, are the most frequently described because of the specific challenges that they represent (Jansen and Weisgold, 1995).
The gingival thickness is related to the gingival height and the thickness of the buccal plate (Chang et al., 2003). The initial thickness of the gingival tissues at the crest may be considered as a significant influence on marginal bone stability around implants within the first year of functioning after implant placement (Chang et al., 1999).
The thicker the buccal plate, the less bone resorption takes place. The thicker the gingiva, the less gingival recession occurs (Schropp et al., 1999). Any marginal bone remodeling around the teeth will result in:
- no gingival variation or marginal soft tissue deformity on a thick, flat biotype, but
- some gingival recession and marginal soft tissue deformity on a thin, scalloped biotype.
For background, see Saadoun et al. (1999) and Rompen et al. (2003).
In a study by Chung et al. (2006), implants placed in areas that lacked keratinized gingiva had a higher susceptibility to tissue breakdown due to plaque accumulation. Despite similar plaque levels, implants placed in nonkeratinized areas showed earlier loss of attachment.
Bouri et al. (2008) reported that implants with a narrow zone of keratinized tissue had a significantly higher chance of probing and/or bleeding (89% versus 31%) and significantly higher mean alveolar bone loss than implants with a wider zone of keratinized mucosa.
The dimensions of the peri-implant mucosa at 1 year were related to the peri-implant biotype in the maxillary anterior region (Kan et al., 2003a):
- Thick biotype – the probe was not visible through the gingival margin (Fig. 4.4f).
- Thin biotype – the probe was visible through the gingival margin (Fig. 4.4g).
Since the above study was conducted, different techniques have been made available to measure soft tissue thickness; namely, visual inspection, transgingival probing (Fig. 4.4h), probe transparency, ultrasonic devices, and cone beam CT imaging (Kao et al., 2008; Fu et al., 2011).
Figure 4.4 (f) The thickness of the marginal tissue hidesthe instrument in the sulcus. (g) The probe appears through the thin marginal tissue. (h) Transgingival probing, measuring soft tissue thickness.
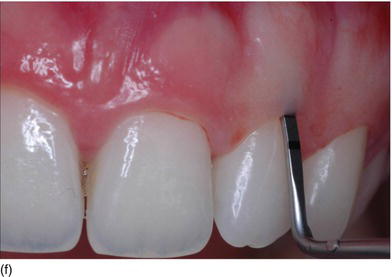
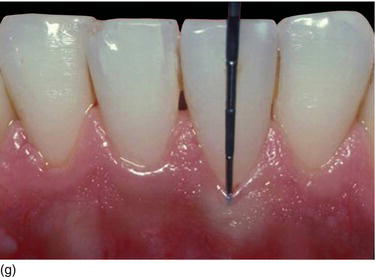
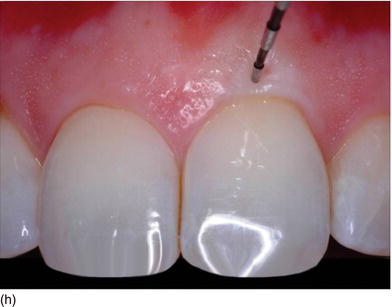
The thickness of the soft tissue around the teeth depends on the eruption on the ridge (see Figs 4.3b, c). However, in the maxillary anterior region with nonsubmerged immediate implant placement, mucosal recession between 3 and 4 years was significantly related to the buccal position implant placement rather than the tissue biotype (Chen et al. 2007).
In the maxillary and mandibulary anterior regions, between 19 and 50 months the thin tissue biotype showed greater peri-implant mucosal recession than the thick tissue biotype, though this was not statistically significant (Evans and Chen, 2008).
In maxillary incisors with flapless immediate implant placement post extraction, mucosal recession of more than 1 mm occurred in 24% of sites with the thin tissue biotype, compared to 10.5% of sites with the thick tissue biotype (Chen and Evans, 2009).
Therefore, the stability of the marginal gingiva depends on two anatomical parameters:
- the presence of hard tissue underlying the peri-implant gingiva, and
- the thickness of the peri-implant gingiva.
Bone resorption on thin biotypes is greater than on thick biotypes, and there is significantly more peri-implant bone loss at sites with thin tissue compared to those with thick tissue. Consequently, there is a direct correlation between the thickness of the peri-implant soft tissue and the peri-implant bone loss (Linkevicius et al., 2009).
Furthermore, Nisapakultorn et al. (2010) found that in the maxillary incisors, the facial marginal mucosal level was significantly associated with the peri-implant tissue biotype: a thin biotype was significantly associated with an increased risk of facial marginal mucosal recession.
The thickness of the soft tissue influences the crestal bone change around implants (Fig. 4.4i):
- If the tissue thickness is 2.0 mm or less, crestal bone loss of up to 1.45 mm can be expected despite a supracrestal position of the implant/abutment interface.
- If the tissue thickness is 2.5 mm or more, crestal bone loss of up to 0.26 mm can be expected.
- Significant marginal bone recession could be avoided if the implant/abutment junction was positioned approximately 2 mm above the bone level; a negligible amount of bone loss, of around 0.2 mm, would then occur.
Figure 4.4 (i) Evaluation of the gingival thickness after flap elevation.
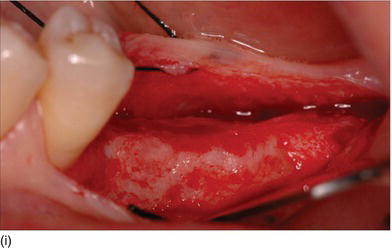
In the evaluation of marginal bone loss, the measurement of gingival thickness should be mandatory. Therefore, it is recommended that bone-level implant placement be avoided if a thin biotype is present at an implant site, or if it is necessary to change the biotype from thin to thick. In a study by Linkevicius et al. (2009), mean peri-implant bone loss at 1 year was found to be inversely correlated with peri-implant mucosal thickness:
- thin peri-implant mucosa, < 2 mm
- medium peri-implant mucosa, 2.1–3.0 mm
- thick peri-implant mucosa, > 3.1 mm.
Consequently, it is important to consider the influence of soft tissue thickness on crestal bone changes around implants along with the patient’s biotype before any implant placement, especially in the esthetic zone (Linkevicius et al., 2009).
The ideal esthetic is often difficult to achieve because the patient’s clinical parameters can vary considerably, depending on the remaining hard and soft tissue in the implant site (Garber, 2010). A thick biotype presents a low risk of marginal discoloration. This risk is higher with a thin biotype, and it becomes necessary to increase its thickness to prevent marginal coloration and/or gingival recession (Leziy and Miller, 2008b). There are favorable and unfavorable factors that should be taken into consideration, and these will be described in more detail in Chapter 6 (see “Peri-implant risk factors”).
The implant survival rate is not affected by the quality and quantity of the peri-implant keratinized tissue. However, a certain amount of height and thickness is important to maintain esthetic and soft tissue health around the implant – especially around an implant with a rough surface, where the adhesion of the connective tissue is fragile in comparison to the dental collagen fibers, which are inserted securely into the root cementum (Rompen, 2011).
The gingival smile
When smiling, a “gummy smile” is an excessive gum display with short clinical teeth, which reveals a more significant amount of the surface of the gingiva (Foley et al., 2003). In general, it is acceptable for up to 2–3 mm of gingival tissue to be displayed upon a full smile (Garber and Salama, 2000). However, such a situation is not systematically negative, as such people often produce very pretty smiles if certain rules of harmony are respected.
An exposition of more than 3 mm of gingival tissue leads to an excessive gingival display, or a “gummy smile.” However, there are other smiles with a gingival display of more than 3 mm that need to be corrected, because they are particularly unattractive (Kokich et al., 1999).
Patients with a significant display of gingiva as a result of genetic factors (i.e., a skeletal deformity such as a thick alveolar buccal bone, altered passive eruption, and a short upper lip) or due to medication (anti-epileptic, antihypertensive, or immunosuppressant drugs) represent a very important challenge for the dental clinician in terms of treatment planning, because of the multifactorial etiologic factors, as well as the psychological and human relations implications associated with the appearance of the face (Nowzari and Rich, 2008).
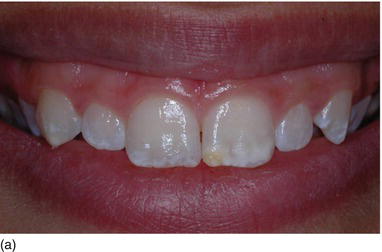
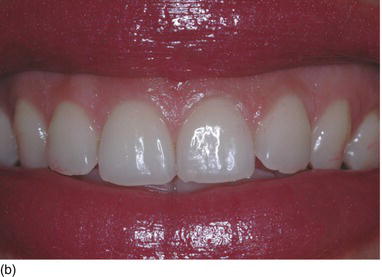
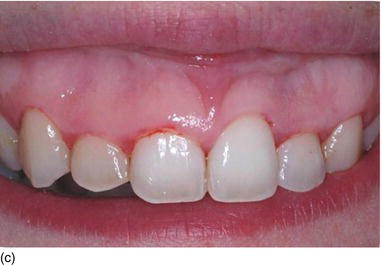
A classification of maxillary vertical gingival evaluation has been proposed by Garber and Salama (1996), on the basis of the height of the exposed gingiva:
- degree I, 2–4 mm of exposed gingiva (Fig. 4.5a)
- degree II, 4–8 mm of exposed gingiva (Fig. 4.5b)
- degree III, > 8 mm of exposed gingiva (Fig. 4.5c).
Figure 4.5 (a) A high lip line, with 2 mm of gingiva showing (Degree I). (b) A gingival smile, with > 5 mm of gingival display, but not unesthetic (Degree II). (c) An excessive gum display > 8 mm, giving an unesthetic gummy smile (Degree III).
Various therapeutic protocols, depending on the height of the exposed gingiva or the excessive amount of vertical bone, are described in Table 4.2.
Although gingival exposure is generally the result of several factors, four primary etiologies can be noted for the gingival smile (Foley et al., 2003; Barbant et al., 2011a).
Osseous etiology: basal, alveolar, or combined
The skeletal abnormality leads to vertical maxillary hypertrophy (up to 8 mm), which is sometimes aggravated by the upper pro alveoli. The main characteristics of this abnormality are as follows (Figs 4.6a–f):
- excessive maxillary vertical growth;
- excessive growth of the maxillary alveolar bone
- malocclusion, with dental maxillary disharmony and dental malpositions
- augmentation of the lower half of the face
- incompetence of the lips, and
- a convex profile and a Class II angle malocclusion.
In general, the patient’s profile is highly convex and presents an augmentation of the lower half of the face, an incompetence of the lips, and a Class II angle malocclusion at rest, and at different dynamic stages of the smile (Cheng-Yi et al., 2008; Toca et al., 2008). Orthodontic treatment with the extraction of all the first premolars, combined with orthognatic surgery if necessary, will resolve these problems.
Table 4.2 Different therapeutic protocols depending on the exposed height of the gingiva
Data from Attal and Tirlet (2011).
| Degree | Gingival exposure (mm) | Therapeutic protocol |
| I | 2–4 | Orthodontic ingression with mini-implants Orthodontic treatment and periodontal surgery Periodontal surgery and restorative treatment |
| II | 4–8 | Periodontal surgery and restorative treatment Orthognatic surgery (depending on the root length and the clinical crown: root ratio) |
| III | > 8 | Orthognatic surgery with or without periodontal surgery and restorative treatment |
Figure 4.6 (a) An increase in height of the lower third part of the patient ’ s face. (b) The patient ’ s smile, showing excessive gingiva and a maxillary overjet. (c) Class II, division 1, with a large open bite. (d) The patient ’ s face 3 years later, with a decrease in the height of the lower third part of the face after extraction of the four first premolars and orthodontic therapy. (e) The patient ’ s beautiful smile after therapy, with a normal amount of gingiva. (f) The correct occlusal relation and tooth position after orthodontic therapy. (Figures (a),(d),(e),(f) courtesy of Dr. P. Curiel.)

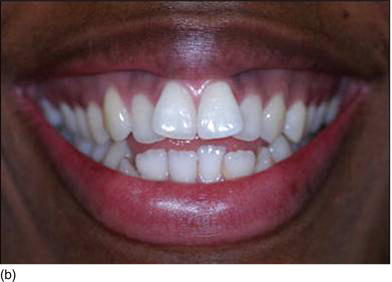
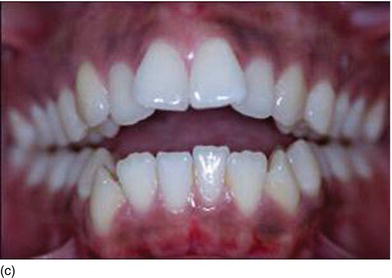

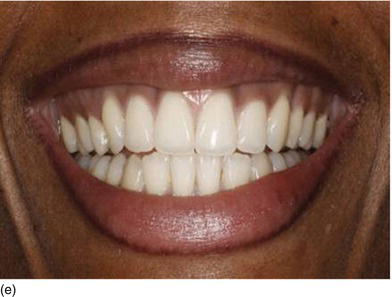
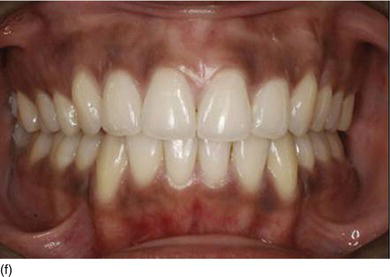
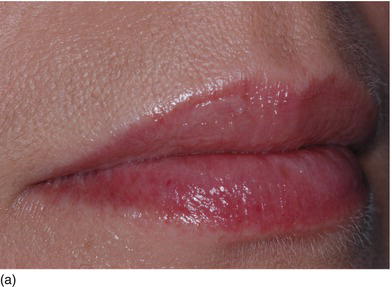
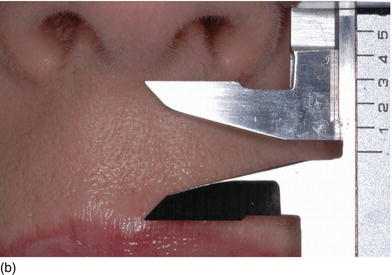
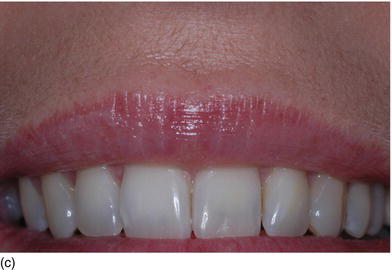
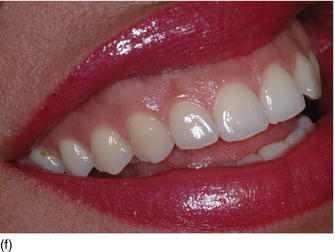
Figure 4.7 (a) Beautiful lips, with a normal upper lip height. (b) Measuring the vertical height between the nose and the tip of the upper lip. (c) The patient’s beautiful smile, without showing the gingival margin.
Muscular etiology
There is usually a short tonicity and/or a hypertonicity of the upper lip with lifting or “elevator” muscles, causing an exaggerated labial elevation. The characteristics of this abnormality are listed below:
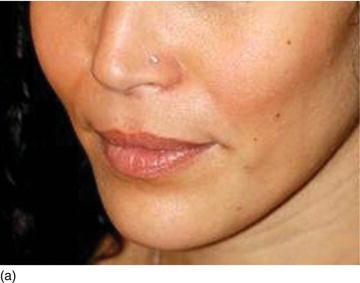
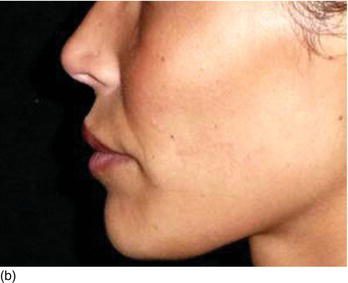
- Hypertonicity of the upper lip (Figs 4.7a–c):
- normal maxillary height
- a normal length of the upper lip, between 20 and 25 mm
- 2 mm of incisal edge showing at rest, with the full anterior teeth exposed on smiling (Figs 4.7a–c).
- Hyperfunction of the elevator muscle of the upper lip, showing teeth and gingiva on a forced smile (Vig and Brundo, 1978; Peck et al., 1992; Ezquerra et al., 1999; Van der Geld et al., 2008).
- Short upper lip (Figs 4.7d–f):
- the length of a normal upper lip is 20–25 mm
- the length of a short upper lip is less than 20 mm
- the upper teeth are visible with the lips at rest, and
- the lip length decreases by an average of 4 mm between the lips at rest and a spontaneous or forced smile showing the full teeth length and excess display of gingiva (Paris and Faucher, 2003).
This clinical situation is more evident on natural and forced smiles (Vig and Brundo, 1978).
Figure 4.7 (d) Beautiful lips but a short upper lipheight. (e) Measuring the vertical height between the nose and the tip of the upper lip. (f) More appearance of gingiva on a forced smile with a short upper lip. (g) A symmetrical smile, with a harmonious lip contour and level. (h) An asymmetrical smile, with a disharmonious lip level.


Various treatments for muscular etiology have been proposed, such as the following:
- Surgical techniques to reposition the upper lip in a more coronal position, limiting the retraction of the elevator muscle:
- an elliptic incision in the depth of the vestibule
- myectomy of the elevator muscle (Litton and Fournier, 1979; Miskinyar, 1983; Rosenblatt and Simon, 2006; Fairbairn, 2010), or
- rhinoplasty associated with resection of the lowering muscle of the nasal septum (Cachay and Velásquez, 1992).
- Injection of Type A botulism toxin (Botox®) and/or hyaluronic acid (Figs 4.8a, b):
- limits the hypertonicity of the elevator muscle of the upper lip
- the site of the injection is 10.4 mm from the lateral edge of the nose, and
- the site of the injection is 32.4 mm above the smile line at rest (Polo, 2005; Toca et al., 2008).
Figure 4.8 (a) A profile view of a young woman, showing the missing midline tip of the upper lip. (b) Restoration of the volume of the upper lip by means of an injection of hyaluronic acid. (Courtesy of Dr. C. Lepage, Paris, France.)
Dental etiology
Short clinical crowns are usually found because of size abnormalities with small teeth, crowns shortened by parafunctional wear (abrasion, bulimia, anorexia), or incomplete eruption of teeth. It is the most important indication of the crown lengthening procedure, combined with laminate veneering if necessary. Clinical situations in which an excess of gingiva combined with short teeth can be found are as follows (Coslet et al., 1977; Foley et al., 2003; Fradeani and Barducci, 2008; Gürel, 2008b):
- an unusually short natural tooth length (Figs 4.9a)
- shorter teeth due to bruxomania or anorexia and/or bulimia (Fig. 4.9b), or
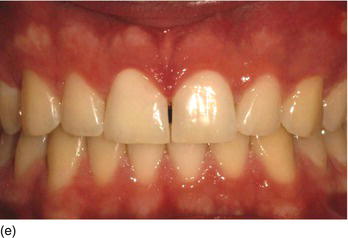
- shorter teeth due to delayed passive eruption (Figs 4.9c–e).
Delayed altered passive eruption
Natural teeth eruption refers to total teeth eruption with a normal relationship between the bone crest and the cemento-enamel junction (see the section on “Natural passive eruption”). Altered passive eruption refers to incomplete natural or delayed eruption, or their absence, in individuals for unknown reasons, and leads to a more coronally positioned gingival margin that may be associated with a normal or coronally displaced bone level (Figs 4.10a–d) (Coslet et al., 1977; Kurtzman and Silverstein, 2008). These individuals have square-shaped clinical crowns with no natural proportion, and tend to display excessive gingival tissue upon smiling. A classification of natural passive eruption and delayed passive eruption (Table 4.3) has been described by several authors (Gargiulo et al., 1961; McGuire and Levine, 1997; Gürel, 2008b; Kao et al., 2008; Monnet-Corti and Borghetti, 2008; Silderberg et al., 2009).
Figure 4.9 (a) Maxillary and mandibular short teeth because of abrasion, at the initial consultation. (b) The teeth are worn and short because of bulimia and anorexia.
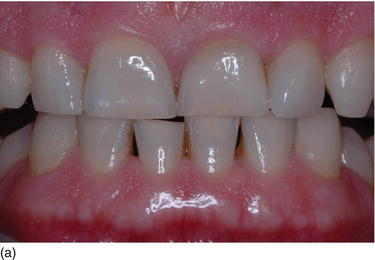
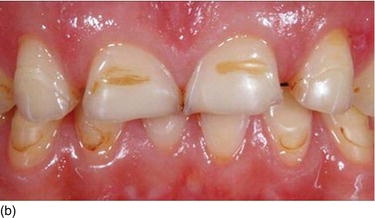
Figure 4.9 (c) Delayed passive eruption at an early stage of orthodontics. (d) Crown lengthening procedures to reestablish the normal relation between the BC and the CEJ. (e) The final result after periodontal and completion of orthodontic therapy.
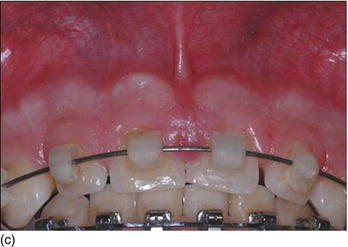
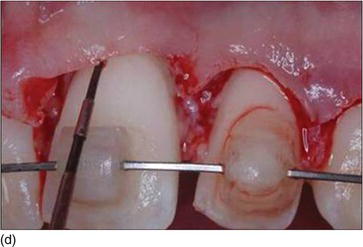
The following three clinical parameters will allow us to distinguish a natural teeth eruption Type I-A or Type II-A from delayed (altered) passive eruption Type I-B or Type II-B (Coslet et al., 1977; Goubron et al., 2011):
- The quantity of keratinized gingiva is measured from the free gingival margin (FGM) to the mucogingival line, and is present in excessive (Type I-B) or limited (Type II-B) quantities.
- The distance obtained by sounding from the bone crest to the gingival margin is less than 1 mm.
- The alveolar bone crest is located at the teeth’s cemento-enamel junction.
Summary
In conclusion, the gingival smile can result from various abnormalities (skeletal, muscular, dental, and physiological), and often a combination of several factors. Once the diagnosis of a gingival smile has been established on the smile at rest and the spontaneous smile, it is necessary to determine the cause. Since the gingival smile is of multifactorial origin, each particular etiology cannot be treated successfully until in-depth esthetic and etiologic diagnostics have been carried out (Nowzari and Rich, 2008). Once the correct treatment has begun, it will involve a multidisciplinary therapeutic plan – which is often quite complex – combining orthognatic orthodontics, osseous and perio-plastic surgery, and restorative treatments (Barbant et al., 2011a).
The gingival smile therefore constitutes a real therapeutic challenge: it demands a rigorous overall procedure in which the surgeon, after having evaluated the nature of the anatomical disorders and the often multiple etiologies, should plan the optimal therapy. The clinician should also be aware of the relationship between hard and soft tissues, in addition to the definitive restorative or natural parameters to be achieved (Bitter, 2007), and must generally propose an interdisciplinary approach to treatment aiming to rebalance the proportions of the three elements that compose the smile – the lips, the teeth, and the gums – in harmony with the face (Garber and Salama, 1996).
A detailed explanation of the dental treatment of gummy smile etiologies will be presented in Chapter 5 (see “Crown lengthening procedures”).
Figure 4.10 (a) Natural eruption of teeth: Type IA. (b) Natural eruption of teeth: Type IIA. (c) Delayed passive eruption: Type IB. (d) Delayed passive eruption: Type IIB. (Courtesy of Dr. G. Tirlet.)
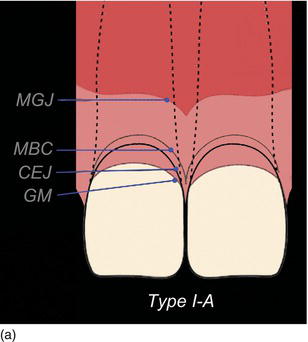
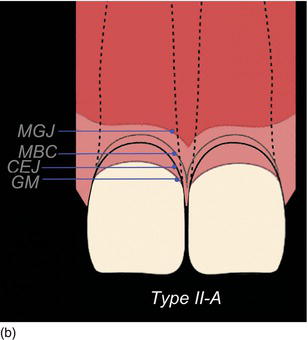
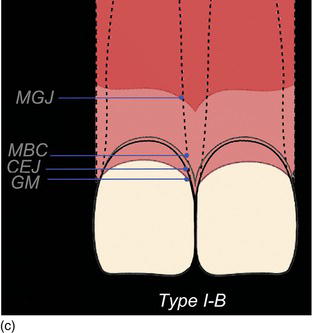
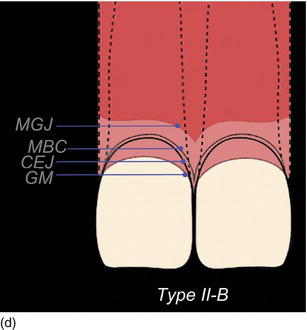
Table 4.3 The classification of natural eruption and altered passive eruption in relation to the qu/>
Stay updated, free dental videos. Join our Telegram channel

VIDEdental - Online dental courses


